Chapter22_LEC
... • the most abundant elements of the Earth’s crust are O and Si • silicates are covalent atomic solids of Si and O and minor amounts of other elements found in rocks, soils, and clays silicates have variable structures – leading to the variety of properties found in rocks, clays, and soils Tro, Ch ...
... • the most abundant elements of the Earth’s crust are O and Si • silicates are covalent atomic solids of Si and O and minor amounts of other elements found in rocks, soils, and clays silicates have variable structures – leading to the variety of properties found in rocks, clays, and soils Tro, Ch ...
Chapter - WTPS.org
... • the most abundant elements of the Earth’s crust are O and Si • silicates are covalent atomic solids of Si and O and minor amounts of other elements found in rocks, soils, and clays silicates have variable structures – leading to the variety of properties found in rocks, clays, and soils Tro, Ch ...
... • the most abundant elements of the Earth’s crust are O and Si • silicates are covalent atomic solids of Si and O and minor amounts of other elements found in rocks, soils, and clays silicates have variable structures – leading to the variety of properties found in rocks, clays, and soils Tro, Ch ...
Minimum electrophilicity principle in Lewis acid–base complexes of
... weak Lewis bases, CO, HCN, CH3CN and CH3F molecules are selected. It is remarked that for the weakly bound molecule, the stability of the BBr3 adduct is lower than the other acid adducts. Therefore, BBr3 is the weaker Lewis acid than BF3 for the weakly bound adducts. According to the Table 2, in LA– ...
... weak Lewis bases, CO, HCN, CH3CN and CH3F molecules are selected. It is remarked that for the weakly bound molecule, the stability of the BBr3 adduct is lower than the other acid adducts. Therefore, BBr3 is the weaker Lewis acid than BF3 for the weakly bound adducts. According to the Table 2, in LA– ...
input
... * * * semiempirical basis sets * * * The elements for which these exist can be found in the 'further information' section of this manual. If you pick one of these, all other data in this group is ignored. GBASIS = MNDO - selects MNDO model hamiltonian = AM1 ...
... * * * semiempirical basis sets * * * The elements for which these exist can be found in the 'further information' section of this manual. If you pick one of these, all other data in this group is ignored. GBASIS = MNDO - selects MNDO model hamiltonian = AM1 ...
Regents Chemistry Review - New York Science Teacher
... The table gives information about the nucleus of each of four atoms. • How many different elements are represented by the nuclei in the table? ...
... The table gives information about the nucleus of each of four atoms. • How many different elements are represented by the nuclei in the table? ...
CHAPTER 20 METALLURGY AND THE CHEMISTRY OF METALS
... The trick in this process centers on the fact that TiCl4 is a liquid with a boiling point (136.4°C), a little higher than that of water. The tetrachloride can be formed by treating the oxide (rutile) with chlorine gas at high temperature. The balanced equation is: TiO2(s) + 2Cl2(g) → TiCl4(l) + O2(g ...
... The trick in this process centers on the fact that TiCl4 is a liquid with a boiling point (136.4°C), a little higher than that of water. The tetrachloride can be formed by treating the oxide (rutile) with chlorine gas at high temperature. The balanced equation is: TiO2(s) + 2Cl2(g) → TiCl4(l) + O2(g ...
Chemistry 133 Problem Set Introduction
... 12 K [(12/24) × 100% = 50 %]. What is the purity of a double eagle in terms of karats? (1 troy ounce = 31.103 g) 1.81 Antifreeze contains the compound ethylene glycol. This compound not only lowers the freezing point of water but also increases the boiling point of water. The density of ethylene gly ...
... 12 K [(12/24) × 100% = 50 %]. What is the purity of a double eagle in terms of karats? (1 troy ounce = 31.103 g) 1.81 Antifreeze contains the compound ethylene glycol. This compound not only lowers the freezing point of water but also increases the boiling point of water. The density of ethylene gly ...
Introduction to Chemistry
... count their teeth. Had he done so, he would have found that men and women have exactly the same number of teeth. In terms of physical science, Aristotle thought about dropping two balls of exactly the same size and shape but of different masses to see which one would strike the ground first. In his ...
... count their teeth. Had he done so, he would have found that men and women have exactly the same number of teeth. In terms of physical science, Aristotle thought about dropping two balls of exactly the same size and shape but of different masses to see which one would strike the ground first. In his ...
AP Chemistry Notes and Worksheets 2014
... o It stated that when two elements form a series of compounds, the ratios of the masses of the second element that combine with 1 gram of the first element can always be reduced to small whole numbers. Data for compounds of nitrogen and oxygen: Compound Percent N Percent O Grams of N Reacting With 1 ...
... o It stated that when two elements form a series of compounds, the ratios of the masses of the second element that combine with 1 gram of the first element can always be reduced to small whole numbers. Data for compounds of nitrogen and oxygen: Compound Percent N Percent O Grams of N Reacting With 1 ...
Quarter 1
... the mass of the reactants and products was less than 100 g sodium sulfate (Na2SO4) is lighter than air some of the water molecules turned into gas ...
... the mass of the reactants and products was less than 100 g sodium sulfate (Na2SO4) is lighter than air some of the water molecules turned into gas ...
2 Atoms and Molecules
... In Chapter 1, we defined elements as homogeneous pure substances made up of identical atoms. At least 115 different elements are known to exist. This leads to the conclusion that a minimum of 115 different kinds of atoms exist. Eighty-eight of the elements are naturally occurring and therefore are f ...
... In Chapter 1, we defined elements as homogeneous pure substances made up of identical atoms. At least 115 different elements are known to exist. This leads to the conclusion that a minimum of 115 different kinds of atoms exist. Eighty-eight of the elements are naturally occurring and therefore are f ...
Chemistry Essentials For Dummies
... No part of this publication may be reproduced, stored in a retrieval system or transmitted in any form or by any means, electronic, mechanical, photocopying, recording, scanning or otherwise, except as permitted under Sections 107 or 108 of the 1976 United States Copyright Act, without either the pr ...
... No part of this publication may be reproduced, stored in a retrieval system or transmitted in any form or by any means, electronic, mechanical, photocopying, recording, scanning or otherwise, except as permitted under Sections 107 or 108 of the 1976 United States Copyright Act, without either the pr ...
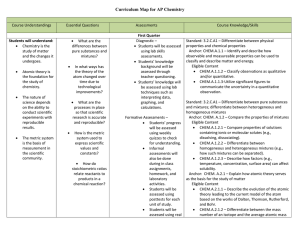
AP Chemistry Curriculum Map - Belle Vernon Area School District
... orbitals with electrons, distribution of electrons in orbitals, shapes of orbitals). Anchor: CHEM.A.2.3 – Explain how periodic trends in the properties of atoms allow for the prediction of physical and chemical properties. Eligible Content CHEM.A.2.3.1 – Explain how the periodicity of chemical pro ...
... orbitals with electrons, distribution of electrons in orbitals, shapes of orbitals). Anchor: CHEM.A.2.3 – Explain how periodic trends in the properties of atoms allow for the prediction of physical and chemical properties. Eligible Content CHEM.A.2.3.1 – Explain how the periodicity of chemical pro ...
Final Exam - KFUPM Faculty List
... Q19. Arrange the following ions: K+, S2-, Ca2+, Cl-, in order of increasing ionic radius. A) Ca2+ < K+ < Cl- < S2B) K+ < Ca2+ < Cl- < S2C) S2- < Cl- < K+ < Ca2+ D) Cl- < S2- < Ca2+ < K+ E) Ca2+ < K+ < S2- < ClIf the ions are isoelectronic then the cations are smaller and the smaller, the larger the ...
... Q19. Arrange the following ions: K+, S2-, Ca2+, Cl-, in order of increasing ionic radius. A) Ca2+ < K+ < Cl- < S2B) K+ < Ca2+ < Cl- < S2C) S2- < Cl- < K+ < Ca2+ D) Cl- < S2- < Ca2+ < K+ E) Ca2+ < K+ < S2- < ClIf the ions are isoelectronic then the cations are smaller and the smaller, the larger the ...
Relativistic coupled cluster theory - in molecular properties and in
... of this effect on a different problem, namely the simulation of X-ray spectroscopy for actinides. As X-ray spectroscopy probes the core-region of a molecule, relativity has to be considered from the outset. Besides, one needs to carefully treat open-shells present in such problems since they lead to ...
... of this effect on a different problem, namely the simulation of X-ray spectroscopy for actinides. As X-ray spectroscopy probes the core-region of a molecule, relativity has to be considered from the outset. Besides, one needs to carefully treat open-shells present in such problems since they lead to ...
Theories of the constitution of gases in the early nineteenth century
... which made it unlikely that identical atoms could unite chemically. The decline of the simple atomic theory and its replacement by Avogadro’s hypothesis as the systematising principle of chemistry is one of the best documented episodes in the history of chemistry (second only to the study of its ori ...
... which made it unlikely that identical atoms could unite chemically. The decline of the simple atomic theory and its replacement by Avogadro’s hypothesis as the systematising principle of chemistry is one of the best documented episodes in the history of chemistry (second only to the study of its ori ...
BASIC CONCEPTS OF CHEMISTRY
... For the thermodynamic description of the system, the state functions are used. These are equations that can be uniquely identified by the parameters P, V and T. The values of these functions are independent of the nature of the process, resulting in a system of this state . The functions of the sta ...
... For the thermodynamic description of the system, the state functions are used. These are equations that can be uniquely identified by the parameters P, V and T. The values of these functions are independent of the nature of the process, resulting in a system of this state . The functions of the sta ...
Review - Discount Flies
... Size: Increases going down a group, decreases going left to right Ionization Energy – the energy required to remove an electron from an atom. decreases going down a group, increases going left to right across a period. ...
... Size: Increases going down a group, decreases going left to right Ionization Energy – the energy required to remove an electron from an atom. decreases going down a group, increases going left to right across a period. ...
File
... 8 Positive ions are formed when the vaporised atom or molecule is bombarded with fast-moving electrons. The kinetic energy of these electrons is great enough to cause the removal of an electron from the outermost orbital of the atom or of one of the bonding electrons in the molecule. e ...
... 8 Positive ions are formed when the vaporised atom or molecule is bombarded with fast-moving electrons. The kinetic energy of these electrons is great enough to cause the removal of an electron from the outermost orbital of the atom or of one of the bonding electrons in the molecule. e ...
Review Study Guide for the Final
... To what volume, in liters, must you dilute a solution containing 4 liters of 0.100 M of Ca(OH)2 to obtain a 0.00100 M solution as calcium hydroxide (Ca(OH)2)? ...
... To what volume, in liters, must you dilute a solution containing 4 liters of 0.100 M of Ca(OH)2 to obtain a 0.00100 M solution as calcium hydroxide (Ca(OH)2)? ...
Molecular orbital diagram
A molecular orbital diagram, or MO diagram, is a qualitative descriptive tool explaining chemical bonding in molecules in terms of molecular orbital theory in general and the linear combination of atomic orbitals (LCAO) molecular orbital method in particular. A fundamental principle of these theories is that as atoms bond to form molecules, a certain number of atomic orbitals combine to form the same number of molecular orbitals, although the electrons involved may be redistributed among the orbitals. This tool is very well suited for simple diatomic molecules such as dihydrogen, dioxygen, and carbon monoxide but becomes more complex when discussing even comparatively simple polyatomic molecules, such as methane. MO diagrams can explain why some molecules exist and others do not. They can also predict bond strength, as well as the electronic transitions that can take place.