chemistry - My Study materials – Kumar
... KVS RO Bangalore and respected sir Shri M.K. Kulshreshtha, Retd-AC, KVS RO Chandigarh for their blessings, motivation and encouragement in bringing out this project in such an excellent form. I also extend my special thanks to respected sir Shri. P. S. Raju, Principal, KV Gachibowli, respected madam ...
... KVS RO Bangalore and respected sir Shri M.K. Kulshreshtha, Retd-AC, KVS RO Chandigarh for their blessings, motivation and encouragement in bringing out this project in such an excellent form. I also extend my special thanks to respected sir Shri. P. S. Raju, Principal, KV Gachibowli, respected madam ...
Principles of Chemistry 1 and 2 Notes
... has a dipole moment or not. Example 1: CO2 ::O = C = O:: (linear, according to the VSEPR model) The CO2 compound does have two electronegative atoms (oxygen) and is expected to have two dipole moments. However, these two dipole moments are identical (same magnitude) and are in opposite directions so ...
... has a dipole moment or not. Example 1: CO2 ::O = C = O:: (linear, according to the VSEPR model) The CO2 compound does have two electronegative atoms (oxygen) and is expected to have two dipole moments. However, these two dipole moments are identical (same magnitude) and are in opposite directions so ...
4) What is the term for the procedure of collecting data and recording
... finding an electron with a given energy? A) electron shell B) electron subshell C) orbital D) quantum level E) none of the above ...
... finding an electron with a given energy? A) electron shell B) electron subshell C) orbital D) quantum level E) none of the above ...
East Meck Chemistry
... any chemical change involves only a rearrangement of the atoms. Atoms do not just appear. Atoms do not just disappear. This is an example of the law of conservation of mass (or matter), which says that in a chemical change, matter is neither ________________ nor destroyed. All chemical changes also ...
... any chemical change involves only a rearrangement of the atoms. Atoms do not just appear. Atoms do not just disappear. This is an example of the law of conservation of mass (or matter), which says that in a chemical change, matter is neither ________________ nor destroyed. All chemical changes also ...
Order date : 24-07-2010
... Coordination chemistry formation and stability of complexes, chelate and macrocyclic effect. The crystal field and ligand field theories, orbital splitting in octahedral, tetrahedral and square planar fields, spectrochemical- and nephelauxetic series, Jahn-Teller effect. M.O. theory- composition of ...
... Coordination chemistry formation and stability of complexes, chelate and macrocyclic effect. The crystal field and ligand field theories, orbital splitting in octahedral, tetrahedral and square planar fields, spectrochemical- and nephelauxetic series, Jahn-Teller effect. M.O. theory- composition of ...
Deuterium fractionation of methylamine through atomic grain
... amino acid in space [2]. Laboratory studies revealed that methylamine can be formed by various reactions both in the gas phase and the solid phase in molecular clouds (MCs) [3,4]. In either case, it is likely that the CH3NH2 is retained on icy grains at as low as 10 K and is subjected to various pro ...
... amino acid in space [2]. Laboratory studies revealed that methylamine can be formed by various reactions both in the gas phase and the solid phase in molecular clouds (MCs) [3,4]. In either case, it is likely that the CH3NH2 is retained on icy grains at as low as 10 K and is subjected to various pro ...
Li K-edge XANES and Li(1s) XPS Spectra of Lithium Compounds
... obtained. Li K-edge XANES spectra are shown in Fig. 2. They have four or five peaks with a variety of peak intensities. The shapes and positions of their peaks may depend on the electron densities of lithium ions and covalent bond characteristics between lithium ions and donor atoms. Li(1s) XPS spec ...
... obtained. Li K-edge XANES spectra are shown in Fig. 2. They have four or five peaks with a variety of peak intensities. The shapes and positions of their peaks may depend on the electron densities of lithium ions and covalent bond characteristics between lithium ions and donor atoms. Li(1s) XPS spec ...
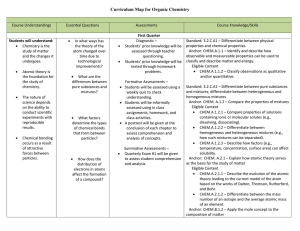
Organic Chemistry Curriculum Map - Belle Vernon Area School District
... ionization energy, electro-negativity, atomic size, and classification of elements. Anchor: CHEM.A.2.1 – Explain how atomic theory serves as the basis for the study of matter. Eligible Content CHEM.A.2.1.2 – Differentiate between the mass number of an isotope and the average atomic mass of an elem ...
... ionization energy, electro-negativity, atomic size, and classification of elements. Anchor: CHEM.A.2.1 – Explain how atomic theory serves as the basis for the study of matter. Eligible Content CHEM.A.2.1.2 – Differentiate between the mass number of an isotope and the average atomic mass of an elem ...
Appendices and Glossary
... this is a trivial example, we will use this same conversion factor or factor label approach for nearly all of the stoichiometric calculations in this book. A.3 MOLAR MASSES AND ATOMIC WEIGHTS OF THE ELEMENTS The number under the symbol of an element on the periodic table is the element’s atomic weig ...
... this is a trivial example, we will use this same conversion factor or factor label approach for nearly all of the stoichiometric calculations in this book. A.3 MOLAR MASSES AND ATOMIC WEIGHTS OF THE ELEMENTS The number under the symbol of an element on the periodic table is the element’s atomic weig ...
Chapter One
... tried to divide a sample of water into infin itesimally small portions, we would eventually end up with a s ingle molecule of water containing two hydrogen atoms and one oxygen atom. If we tried to break this molecule into its individual atom~, we would no longer have water. A molecule is therefore ...
... tried to divide a sample of water into infin itesimally small portions, we would eventually end up with a s ingle molecule of water containing two hydrogen atoms and one oxygen atom. If we tried to break this molecule into its individual atom~, we would no longer have water. A molecule is therefore ...
Table of Contents - slccscience`s Home Page
... elements, it often seems odd that an entire branch of chemistry is devoted to a single element and its compounds while the other 116 elements and their compounds are all lumped together in a separate discipline, but there is a very good reason for this. There are about 1.5 million known inorganic co ...
... elements, it often seems odd that an entire branch of chemistry is devoted to a single element and its compounds while the other 116 elements and their compounds are all lumped together in a separate discipline, but there is a very good reason for this. There are about 1.5 million known inorganic co ...
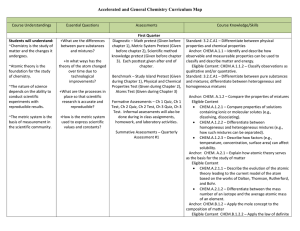
Chemistry Curriculum Map - Belle Vernon Area School District
... for the first twenty elements of the periodic table. Anchor: CHEM.A.2.2 – Describe the behavior of electrons in atoms. Eligible Content: CHEM.A.2.2.1 – Predict the ground state electronic configurations and/or orbital diagram for a given atom or ion. Standard: 3.2.C.A2 – Relate the position of an el ...
... for the first twenty elements of the periodic table. Anchor: CHEM.A.2.2 – Describe the behavior of electrons in atoms. Eligible Content: CHEM.A.2.2.1 – Predict the ground state electronic configurations and/or orbital diagram for a given atom or ion. Standard: 3.2.C.A2 – Relate the position of an el ...
Chapter One
... again and again and again. In theory, we should eventually end up with a single gold atom. If we tried to split this atom in half, we would end up with something that no longer retains any of the characteristics of the element. An atom is therefore the smallest particle that can be used to identify ...
... again and again and again. In theory, we should eventually end up with a single gold atom. If we tried to split this atom in half, we would end up with something that no longer retains any of the characteristics of the element. An atom is therefore the smallest particle that can be used to identify ...
theodore l. brown h. eugene lemay, jr. bruce e. bursten catherine j
... system, or transmission in any form or by any means, electronic, mechanical, photocopying, recording, or likewise. To obtain permission(s) to use material from this work, please submit a written request to Pearson Education, Inc., Permissions Department, 1900 E. Lake Ave., Glenview, IL 60025. Many o ...
... system, or transmission in any form or by any means, electronic, mechanical, photocopying, recording, or likewise. To obtain permission(s) to use material from this work, please submit a written request to Pearson Education, Inc., Permissions Department, 1900 E. Lake Ave., Glenview, IL 60025. Many o ...
CLUE - virtual laboratories
... Chemistry, and particularly introductory general chemistry, is simply not changing that much and people learn pretty much the same way they always did, at least if we restrict ourselves to the last fe thousand years.1 On the other hand, there is compelling evidence that the way chemistry is commonly ...
... Chemistry, and particularly introductory general chemistry, is simply not changing that much and people learn pretty much the same way they always did, at least if we restrict ourselves to the last fe thousand years.1 On the other hand, there is compelling evidence that the way chemistry is commonly ...
UILChemistryProblemsPart2
... 28. What is [Cl-] in the solution formed by mixing 50 mL of 0.20 M hydrochloric acid (aq) with 50 mL of 0.30 M sodium hydroxide (aq)? Not really a neutralization, but looks like one. 2. Write the formula of hydrochloric acid. 3. Calculate moles of HCl, Write dissociation reaction for HCl and covert ...
... 28. What is [Cl-] in the solution formed by mixing 50 mL of 0.20 M hydrochloric acid (aq) with 50 mL of 0.30 M sodium hydroxide (aq)? Not really a neutralization, but looks like one. 2. Write the formula of hydrochloric acid. 3. Calculate moles of HCl, Write dissociation reaction for HCl and covert ...
Scientific Jury of the 30th International
... about these exercises, we will provide an opportunity to discuss these exercises (and other matters) with their fellow students from all over the world, even before they come together in Melbourne. We have set up a web-based chat forum so that they can get to know one another (after all - isn’t that ...
... about these exercises, we will provide an opportunity to discuss these exercises (and other matters) with their fellow students from all over the world, even before they come together in Melbourne. We have set up a web-based chat forum so that they can get to know one another (after all - isn’t that ...
Study guide for final
... 4) Li : is the proper Lewis structure (dot structure) for lithium. 5) Lewis theory predicts that the formula for a compound made of aluminum and phosphorus is AlP. 6) The correct Lewis structure for CO2 shows that the carbon atom has two sets of lone pair electrons. 7) The VSEPR theory predicts that ...
... 4) Li : is the proper Lewis structure (dot structure) for lithium. 5) Lewis theory predicts that the formula for a compound made of aluminum and phosphorus is AlP. 6) The correct Lewis structure for CO2 shows that the carbon atom has two sets of lone pair electrons. 7) The VSEPR theory predicts that ...
ChemistryReview
... 56. Neutrons and ____________________ have almost the same mass. 57. When an atom gains or loses energy, some of its ____________________ may move between energy levels. 58. The region in which an electron is most likely to be found is called a(an) ____________________. 59. When all the electrons i ...
... 56. Neutrons and ____________________ have almost the same mass. 57. When an atom gains or loses energy, some of its ____________________ may move between energy levels. 58. The region in which an electron is most likely to be found is called a(an) ____________________. 59. When all the electrons i ...
PDF - mockies – Mockiesgateacademy
... Eg. Consider the following reaction 2 H2 + O2 → 2H2O In this reaction one molecule of oxygen reacts with two molecules of hydrogen. So it would be desirable to take the molecules of H2 and oxygen in the ratio 2:1, so that the reactants are completely consumed during the reaction. But atoms and mole ...
... Eg. Consider the following reaction 2 H2 + O2 → 2H2O In this reaction one molecule of oxygen reacts with two molecules of hydrogen. So it would be desirable to take the molecules of H2 and oxygen in the ratio 2:1, so that the reactants are completely consumed during the reaction. But atoms and mole ...
Answers - Pearson
... compounds of Group 1 and 2 elements. These compounds were later broken down into their component elements by electrolysis. 3 The Schrödinger model: ...
... compounds of Group 1 and 2 elements. These compounds were later broken down into their component elements by electrolysis. 3 The Schrödinger model: ...
Molecular orbital diagram
A molecular orbital diagram, or MO diagram, is a qualitative descriptive tool explaining chemical bonding in molecules in terms of molecular orbital theory in general and the linear combination of atomic orbitals (LCAO) molecular orbital method in particular. A fundamental principle of these theories is that as atoms bond to form molecules, a certain number of atomic orbitals combine to form the same number of molecular orbitals, although the electrons involved may be redistributed among the orbitals. This tool is very well suited for simple diatomic molecules such as dihydrogen, dioxygen, and carbon monoxide but becomes more complex when discussing even comparatively simple polyatomic molecules, such as methane. MO diagrams can explain why some molecules exist and others do not. They can also predict bond strength, as well as the electronic transitions that can take place.