Chapter 1
... *Notes-The Law of Conservation of Mass dictates that chemical equations must be balanced because atoms are never _____lost__or _____gained__in a chemical reaction. ...
... *Notes-The Law of Conservation of Mass dictates that chemical equations must be balanced because atoms are never _____lost__or _____gained__in a chemical reaction. ...
Basic Chemistry
... Types of Bonds Covalent bonds formed when electrons are shared among elements in a compound. usually formed between two non metals or a non metal and hydrogen Models or diagrams can be drawn to show the shared electrons ...
... Types of Bonds Covalent bonds formed when electrons are shared among elements in a compound. usually formed between two non metals or a non metal and hydrogen Models or diagrams can be drawn to show the shared electrons ...
Balancing chemical equations notes
... chemicals are combined together and what chemicals are made when a reaction occurs. The law of conservation of mass says that matter can neither be created nor destroyed, and this requires that all chemical reactions be balanced. Consider the following balanced equation: Cu (s) + 4 HNO3 (aq) Cu(NO ...
... chemicals are combined together and what chemicals are made when a reaction occurs. The law of conservation of mass says that matter can neither be created nor destroyed, and this requires that all chemical reactions be balanced. Consider the following balanced equation: Cu (s) + 4 HNO3 (aq) Cu(NO ...
CHEMISTRY-1 CHAPTER 8 CHEMICAL REACTIONS
... HNO3(aq) + AgCl(s) 1. HCl(aq) + AgNO3(aq) 2. 3 CaCl2(aq) +2 Na3PO4(aq) Ca3(PO4)2(s) + 6 NaCl(aq) 3. Pb(NO3)2(aq) + BaCl2(aq) PbCl2(s) + Ba(NO3)2(aq) 4. FeCl3(aq) + 3 NaOH(aq) Fe(OH)3(s) +3 NaCl(aq) 5. H2SO4(aq) +2 NaOH(aq) 2 H2O(l) + Na2SO4(aq) ...
... HNO3(aq) + AgCl(s) 1. HCl(aq) + AgNO3(aq) 2. 3 CaCl2(aq) +2 Na3PO4(aq) Ca3(PO4)2(s) + 6 NaCl(aq) 3. Pb(NO3)2(aq) + BaCl2(aq) PbCl2(s) + Ba(NO3)2(aq) 4. FeCl3(aq) + 3 NaOH(aq) Fe(OH)3(s) +3 NaCl(aq) 5. H2SO4(aq) +2 NaOH(aq) 2 H2O(l) + Na2SO4(aq) ...
Chemical Reactions and Equations
... Here stannous chloride is a reducing agent. It reduces ferric chloride to ferrous chloride. ...
... Here stannous chloride is a reducing agent. It reduces ferric chloride to ferrous chloride. ...
HIGHER TIER CHEMISTRY MINI-MOCK UNIT 2
... Electrolysis can be used to remove unwanted hair from the skin. The hair is first coated with a layer of gel containing ions in solution. The positive electrode is connected by a patch to the skin.The negative electrode is connected to the hair. Electricity flows through the gel and causes electroly ...
... Electrolysis can be used to remove unwanted hair from the skin. The hair is first coated with a layer of gel containing ions in solution. The positive electrode is connected by a patch to the skin.The negative electrode is connected to the hair. Electricity flows through the gel and causes electroly ...
Name__________________________ Period_______ Word
... example of a reaction condition is the heat symbol (∆ ), which indicates that the reactants were heated. An equation can also indicate the physical state of the reactants and products. The physical states of matter symbols are listed below. ...
... example of a reaction condition is the heat symbol (∆ ), which indicates that the reactants were heated. An equation can also indicate the physical state of the reactants and products. The physical states of matter symbols are listed below. ...
Chapters 6, 8
... All other elements have electronic configuration that is more or less unstable. To attain the electronic configuration of the closest noble gas, the element produces compounds. An element can produce a compound by: ...
... All other elements have electronic configuration that is more or less unstable. To attain the electronic configuration of the closest noble gas, the element produces compounds. An element can produce a compound by: ...
The Atomic Theory of Matter
... • John Dalton’s theory says that atoms are the smallest particles that still retain chemical identity of an element. • With new technology we have found subatomic particles within atoms. • A lot of things are still considered correct with John Dalton’s theory even today. ...
... • John Dalton’s theory says that atoms are the smallest particles that still retain chemical identity of an element. • With new technology we have found subatomic particles within atoms. • A lot of things are still considered correct with John Dalton’s theory even today. ...
Ch. 7 & 8 Notes (Chemical Reactions) teacher
... side. The arrow means “________”, or “reacts to produce” when read aloud. ...
... side. The arrow means “________”, or “reacts to produce” when read aloud. ...
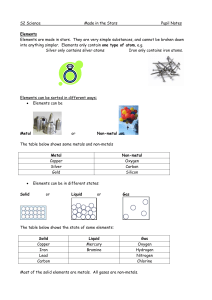
Made in the Stars Notes
... Elements can join together to form compounds. They have to be chemically joined, not just mixed together. A compound is a substance that has more than one kind of atom joined together. For example, if iron joins with oxygen from the air, it forms the compound iron oxide (rust). ...
... Elements can join together to form compounds. They have to be chemically joined, not just mixed together. A compound is a substance that has more than one kind of atom joined together. For example, if iron joins with oxygen from the air, it forms the compound iron oxide (rust). ...
Ionic Equations
... Predict formation of precipitates by application of solubility rules Write total and net ionic equations from balanced molecular equations ...
... Predict formation of precipitates by application of solubility rules Write total and net ionic equations from balanced molecular equations ...
PPT: Chemical Reactions Review
... Ox: C-2H4O → C0H2O (C is going from -2 to 0) Red: (Cr26+O7)2- → Cr3+ (Cr is being reduced from +6 to +3) ...
... Ox: C-2H4O → C0H2O (C is going from -2 to 0) Red: (Cr26+O7)2- → Cr3+ (Cr is being reduced from +6 to +3) ...
Chapter 6A Chemical Reactions CHAPTER OUTLINE
... q In these reactions, oxidation involves addition of oxygen or loss of hydrogen, and reduction involves Oxidation loss of oxygen or gain of hydrogen. (loss of q For example, poisonous methyl alcohol is ...
... q In these reactions, oxidation involves addition of oxygen or loss of hydrogen, and reduction involves Oxidation loss of oxygen or gain of hydrogen. (loss of q For example, poisonous methyl alcohol is ...
Common Chemical Formula List
... Chemical Formula Definition: An expression which states the number and type of atoms present in a molecule of a substance. Chemical formulas such as HClO4 can be divided into empirical formula, molecular formula, and structural formula. Chemical symbols of elements in the chemical formula represent ...
... Chemical Formula Definition: An expression which states the number and type of atoms present in a molecule of a substance. Chemical formulas such as HClO4 can be divided into empirical formula, molecular formula, and structural formula. Chemical symbols of elements in the chemical formula represent ...
Metals and non-metals III IMPORTANT POINTS Non-metals
... 1. a. Magnesium, chromium and sodium are all metals, hence, they react with oxygen to form basic oxides b. Chromium, as it is a transition metal. Metals have high density and coloured compounds are formed by transition metals. c. Bromine - the formula is Br2, that is, two atoms of bromine. d. Bromin ...
... 1. a. Magnesium, chromium and sodium are all metals, hence, they react with oxygen to form basic oxides b. Chromium, as it is a transition metal. Metals have high density and coloured compounds are formed by transition metals. c. Bromine - the formula is Br2, that is, two atoms of bromine. d. Bromin ...
Re-typed from The Ultimate Chemical Equations Handbook by
... 2. Mentally count or tally how many of each type of atom is present on each side of the equation. 3. Begin by balancing elements that are only found in one substance on each side. 4. Balance oxygen and hydrogen LAST – they usually balance out at the end or perhaps only the number of water molecules ...
... 2. Mentally count or tally how many of each type of atom is present on each side of the equation. 3. Begin by balancing elements that are only found in one substance on each side. 4. Balance oxygen and hydrogen LAST – they usually balance out at the end or perhaps only the number of water molecules ...
Chemical Reactions: Helpful Hints
... Reaction 10 involves a solid metal (Zn in the 0 oxidation state) and an aqueous metal ion (Ag+ in the 1+ oxidation state). Did you observe a band of shiny metal being formed at the interface of the solid and the solution (Hmm, what could that be? What was in solution that would give such luster?) R ...
... Reaction 10 involves a solid metal (Zn in the 0 oxidation state) and an aqueous metal ion (Ag+ in the 1+ oxidation state). Did you observe a band of shiny metal being formed at the interface of the solid and the solution (Hmm, what could that be? What was in solution that would give such luster?) R ...
Electrochemistry
Electrochemistry is the branch of physical chemistry that studies chemical reactions which take place at the interface of an electrode, usually a solid metal or a semiconductor, and an ionic conductor, the electrolyte. These reactions involve electric charges moving between the electrodes and the electrolyte (or ionic species in a solution). Thus electrochemistry deals with the interaction between electrical energy and chemical change.When a chemical reaction is caused by an externally supplied current, as in electrolysis, or if an electric current is produced by a spontaneous chemical reaction as in a battery, it is called an electrochemical reaction. Chemical reactions where electrons are transferred directly between molecules and/or atoms are called oxidation-reduction or (redox) reactions. In general, electrochemistry describes the overall reactions when individual redox reactions are separate but connected by an external electric circuit and an intervening electrolyte.