
Express Scripts 2015 Drug Trend Report
... recent years. Compared to 2014, brand prices in 2015 were 16% higher. Brand medications have increased in price by 164% between 2008 and 2015. Consider the case of Gleevec: In 2015, Novartis, the exclusive manufacturer, engaged in the prevalent practice of increasing the price of a medication in the ...
... recent years. Compared to 2014, brand prices in 2015 were 16% higher. Brand medications have increased in price by 164% between 2008 and 2015. Consider the case of Gleevec: In 2015, Novartis, the exclusive manufacturer, engaged in the prevalent practice of increasing the price of a medication in the ...
CYP2D6 - PGXL Laboratories
... requirement (e.g., warfarin) due to decreased clearance, increased elimination half-life, and increased time to reach steady-state blood concentrations. VKORC1 Intermediate Warfarin Sensitivity: ‡The warfarin maintenance dose estimate was derived using a published formula that accounts for age, gend ...
... requirement (e.g., warfarin) due to decreased clearance, increased elimination half-life, and increased time to reach steady-state blood concentrations. VKORC1 Intermediate Warfarin Sensitivity: ‡The warfarin maintenance dose estimate was derived using a published formula that accounts for age, gend ...
The following information was generated from the Hazardous
... enlargement, occur commonly in females; these changes may not be reversible following discontinuance of the drug. [McEvoy, G.K. (ed.). American Hospital Formulary Service- Drug Information 2005. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 2005 (Plus Supplements)., p. 2953]**PEE ...
... enlargement, occur commonly in females; these changes may not be reversible following discontinuance of the drug. [McEvoy, G.K. (ed.). American Hospital Formulary Service- Drug Information 2005. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 2005 (Plus Supplements)., p. 2953]**PEE ...
The Potential of Cyclodextrins as Novel Active Pharmaceutical
... The deviation from linearity can be positive (AP ) or negative (AN ) (Figure 3). A positive or negative deviation form linearity implies that the molar ratio between ligand and substrate (CD and drug respectively) is not 1:1 [10]. In this case, it is very difficult to predict from these diagrams the ...
... The deviation from linearity can be positive (AP ) or negative (AN ) (Figure 3). A positive or negative deviation form linearity implies that the molar ratio between ligand and substrate (CD and drug respectively) is not 1:1 [10]. In this case, it is very difficult to predict from these diagrams the ...
Inhaled synthetic surfactant abolishes the early allergen-induced response in asthma K.S. Babu
... Subjects received both Pumactant (PT) and no treatment (NT), in a randomised order, separated by at least a 3-week interval. Pumactant was formulated as a novel, micronised dry powder. Two administrations of Pumactant were given prior to exposure to the allergen BPT. The first was administered 8 h i ...
... Subjects received both Pumactant (PT) and no treatment (NT), in a randomised order, separated by at least a 3-week interval. Pumactant was formulated as a novel, micronised dry powder. Two administrations of Pumactant were given prior to exposure to the allergen BPT. The first was administered 8 h i ...
Attachment: Product Information: Atazanavir
... with a meal high in calories, fat, and protein (721 kcal, 37.3 g fat, 29.4 g protein) resulted in a mean increase in AUC of 35% and no change in Cmax compared to administration in the fasting state. Administration of Reyataz with either a light meal or a high fat meal decreased the coefficient of va ...
... with a meal high in calories, fat, and protein (721 kcal, 37.3 g fat, 29.4 g protein) resulted in a mean increase in AUC of 35% and no change in Cmax compared to administration in the fasting state. Administration of Reyataz with either a light meal or a high fat meal decreased the coefficient of va ...
Appendix 10: Administering drugs via feeding tubes
... Box A10.D Drug interactions and preventive action when giving drugs by tubes Binding of drugs to tubes e.g. carbamazepine, diazepam, phenytoin.3 Dilute the drug with at least 30–60ml of water and flush well; monitor clinical response. Direct interaction of drug and feed causing coagulation in the t ...
... Box A10.D Drug interactions and preventive action when giving drugs by tubes Binding of drugs to tubes e.g. carbamazepine, diazepam, phenytoin.3 Dilute the drug with at least 30–60ml of water and flush well; monitor clinical response. Direct interaction of drug and feed causing coagulation in the t ...
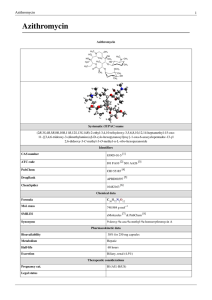
Azithromycin - Rebound Health
... treatment for four or five additional days. With the "Z-Pak," this means two 250 mg tablets (a total of 500 mg) on the first day and one 250 mg tablet once daily for the next four days. Pfizer brand-name (e.g., Zithromax) azithromycin tablets are mottled pink, unscored, film-coated, modified-oval-sh ...
... treatment for four or five additional days. With the "Z-Pak," this means two 250 mg tablets (a total of 500 mg) on the first day and one 250 mg tablet once daily for the next four days. Pfizer brand-name (e.g., Zithromax) azithromycin tablets are mottled pink, unscored, film-coated, modified-oval-sh ...
Ofloxacin as a Reference Marker in Hair of Various Colors
... The goal of this study was to determine if an oral dose of the antibiotic ofloxacin (OFLX) could be used as a "marker" substance to establish reference points with respect to time in hair of various colors. Male and female subjects (n = 32) between 18 and 40 years of age received 800 mg of OFLX as a ...
... The goal of this study was to determine if an oral dose of the antibiotic ofloxacin (OFLX) could be used as a "marker" substance to establish reference points with respect to time in hair of various colors. Male and female subjects (n = 32) between 18 and 40 years of age received 800 mg of OFLX as a ...
View the 2016 Prior Authorization Criteria
... All FDA-approved indications not otherwise excluded from Part D. CYP2D6 extensive metabolizers and intermediate metabolizers taking a strong or moderate CYP2D6 inhibitor (e.g., paroxetine, terbinafine) concomitantly with a strong or moderate CYP3A inhibitor (e.g., ketoconazole, fluconazole). CYP2D6 ...
... All FDA-approved indications not otherwise excluded from Part D. CYP2D6 extensive metabolizers and intermediate metabolizers taking a strong or moderate CYP2D6 inhibitor (e.g., paroxetine, terbinafine) concomitantly with a strong or moderate CYP3A inhibitor (e.g., ketoconazole, fluconazole). CYP2D6 ...
Spiractin - Medicines.org.au
... In animal studies, spironolactone was devoid of teratogenic effects in mice and rabbits at oral doses up to 20mg/kg/day, and in rats at dietary doses up to 50 mg/kg/day. However, increased resorption rate was seen at 20 mg/kg/day in rabbits, and the incidence of stillbirths was increased in rats dos ...
... In animal studies, spironolactone was devoid of teratogenic effects in mice and rabbits at oral doses up to 20mg/kg/day, and in rats at dietary doses up to 50 mg/kg/day. However, increased resorption rate was seen at 20 mg/kg/day in rabbits, and the incidence of stillbirths was increased in rats dos ...
Wellington ICU Drug Manual - Wellington Intensive Care Unit
... This second edition was updated in 2013 with revisions made reflecting the changes in our unit’s Intensive Care practice. ...
... This second edition was updated in 2013 with revisions made reflecting the changes in our unit’s Intensive Care practice. ...
Epinefrina. Adrenalina. Monografia del farmaco.
... Adrenalin has been shown to be teratogenic in rats when given in doses about 25 times the human dose. There are no adequate and well-controlled studies in pregnant women. Adrenalin should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus. EpiPen and Epi ...
... Adrenalin has been shown to be teratogenic in rats when given in doses about 25 times the human dose. There are no adequate and well-controlled studies in pregnant women. Adrenalin should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus. EpiPen and Epi ...
Macrocycles in new drug discovery
... highlighting the favorable changes in biological and physicochemical properties that macrocyclization can afford. Natural product macrocycles and their synthetic derivatives have long been clinically useful and attention is now being focused on the wider use of macrocyclic scaffolds in medicinal che ...
... highlighting the favorable changes in biological and physicochemical properties that macrocyclization can afford. Natural product macrocycles and their synthetic derivatives have long been clinically useful and attention is now being focused on the wider use of macrocyclic scaffolds in medicinal che ...
Decreased Serum Concentrations of Tamoxifen
... tamoxifen, but because of more frequent side effects aminoglu tethimide is generally used after tamoxifen as a second line endocrine treatment (12). Combination therapy with tamoxifen plus aminoglutethi mide should afford both estrogen receptor blockade and reduced plasma estrogen levels, and becaus ...
... tamoxifen, but because of more frequent side effects aminoglu tethimide is generally used after tamoxifen as a second line endocrine treatment (12). Combination therapy with tamoxifen plus aminoglutethi mide should afford both estrogen receptor blockade and reduced plasma estrogen levels, and becaus ...
[27] Kumar, RMNV- Eds. Handbook of Particulate Drug Delivery,2008
... Table-2: Different silks and their use ...
... Table-2: Different silks and their use ...
500ct. 7.5-500 mg HCb and APAP Tablets[Watson[Base[rev2
... determined to be 3.8 ± 0.3 hours. Hydrocodone exhibits a complex pattern of metabolism including O-demethylation, N-demethylation and 6-keto reduction to the corresponding 6-α- and 6-β-hydroxy-metabolites. See OVERDOSAGE for toxicity information. Acetaminophen: Acetaminophen is rapidly absorbed from ...
... determined to be 3.8 ± 0.3 hours. Hydrocodone exhibits a complex pattern of metabolism including O-demethylation, N-demethylation and 6-keto reduction to the corresponding 6-α- and 6-β-hydroxy-metabolites. See OVERDOSAGE for toxicity information. Acetaminophen: Acetaminophen is rapidly absorbed from ...
DEXDOMITOR 0.5 mg/mL
... DEXDOMITOR was used in the presence of anesthetics and/or other preanesthetics. It is not always possible to reliably estimate the frequency of an adverse event or to establish a causal relationship to the drug, especially when multiple drugs are administered. The following reported adverse events a ...
... DEXDOMITOR was used in the presence of anesthetics and/or other preanesthetics. It is not always possible to reliably estimate the frequency of an adverse event or to establish a causal relationship to the drug, especially when multiple drugs are administered. The following reported adverse events a ...
Pharmacologic Identification of the Lower Esophageal Sphincter
... identify at least the first part of the dose-response curve for each level of the esophagus. The definition of the curve for most drugs was coarse because, within each decade of concentration, doses were made only at two points. The actual concentrations used are apparent from the illustrations show ...
... identify at least the first part of the dose-response curve for each level of the esophagus. The definition of the curve for most drugs was coarse because, within each decade of concentration, doses were made only at two points. The actual concentrations used are apparent from the illustrations show ...
Acetaminophen: Old drug, new warnings
... doses of acetaminophen or in malnourished people, the toxic metabolite accumulates, resulting in liver damage (Figure 1).6 The liver is damaged by two mechanisms. In one, NAPQI binds to hepatic cell macromolecules, causing dysfunction of the enzymatic systems, structural and metabolic disarray, an ...
... doses of acetaminophen or in malnourished people, the toxic metabolite accumulates, resulting in liver damage (Figure 1).6 The liver is damaged by two mechanisms. In one, NAPQI binds to hepatic cell macromolecules, causing dysfunction of the enzymatic systems, structural and metabolic disarray, an ...
the scientific value of non-clinical animal studies in drug development
... of animal studies, which is that it is a living model system that is being used to extrapolate results from. Animal model systems have high intrinsic variability, immutable differences within species and are also mechanistically not completely understood(17). Study design can influence all three fac ...
... of animal studies, which is that it is a living model system that is being used to extrapolate results from. Animal model systems have high intrinsic variability, immutable differences within species and are also mechanistically not completely understood(17). Study design can influence all three fac ...
Billing with National Drug Codes (NDCs)
... F2 (International Unit) – Products described as IU/vial or micrograms Note: ME is also a recognized billing qualifier that may be used to identify milligrams as the NDC unit of measure; however, drug costs are generally created at the UN or ML level. If a drug product is billed using milligrams, i ...
... F2 (International Unit) – Products described as IU/vial or micrograms Note: ME is also a recognized billing qualifier that may be used to identify milligrams as the NDC unit of measure; however, drug costs are generally created at the UN or ML level. If a drug product is billed using milligrams, i ...
ACMD Advisory Council on the Misuse of Drugs ACMD consideration of tramadol
... 3.1. Tramadol – (±)-trans-2-((dimethylamino)methyl)-1-(3methoxyphenyl)cyclohexanol – is a synthetic opioid with effects similar to those of codeine. It is an analgesic with weak agonist activity at µ-opioid receptors in the brain (Ki = 2400 nM; Gillen et al., 2000). The affinity of tramadol for the ...
... 3.1. Tramadol – (±)-trans-2-((dimethylamino)methyl)-1-(3methoxyphenyl)cyclohexanol – is a synthetic opioid with effects similar to those of codeine. It is an analgesic with weak agonist activity at µ-opioid receptors in the brain (Ki = 2400 nM; Gillen et al., 2000). The affinity of tramadol for the ...
International Journal of Chemical and Natural Science
... injecting blank solution( without any sample) and then a drug solution of 10µl injected into the column, under optimized chromatographic conditions, to demonstrate the separation of both ketorolac and phenylephrine from any of the impurities, if present. As there was no interference of impurities an ...
... injecting blank solution( without any sample) and then a drug solution of 10µl injected into the column, under optimized chromatographic conditions, to demonstrate the separation of both ketorolac and phenylephrine from any of the impurities, if present. As there was no interference of impurities an ...
RM-Ii3201 B DIS SPECIFICATION
... the needle tip. This solution should be replaced each time the heparin Iock is used. Aspirate before administering any so[utionvia the lock in orderto confirm patency and location of needle or catheter tlp. If the drug to be admlnlstered is incompatible wlth heparin, the entire heparin lock set shou ...
... the needle tip. This solution should be replaced each time the heparin Iock is used. Aspirate before administering any so[utionvia the lock in orderto confirm patency and location of needle or catheter tlp. If the drug to be admlnlstered is incompatible wlth heparin, the entire heparin lock set shou ...
Pharmacokinetics

Pharmacokinetics, sometimes abbreviated as PK (from Ancient Greek pharmakon ""drug"" and kinetikos ""moving, putting in motion""; see chemical kinetics), is a branch of pharmacology dedicated to determining the fate of substances administered externally to a living organism. The substances of interest include pharmaceutical agents, hormones, nutrients, and toxins. It attempts to discover the fate of a drug from the moment that it is administered up to the point at which it is completely eliminated from the body.Pharmacokinetics describes how the body affects a specific drug after administration through the mechanisms of absorption and distribution, as well as the chemical changes of the substance in the body (e.g. by metabolic enzymes such as cytochrome P450 or glucuronosyltransferase enzymes), and the effects and routes of excretion of the metabolites of the drug. Pharmacokinetic properties of drugs may be affected by elements such as the site of administration and the dose of administered drug. These may affect the absorption rate. Pharmacokinetics is often studied in conjunction with pharmacodynamics, the study of a drug's pharmacological effect on the body.A number of different models have been developed in order to simplify conceptualization of the many processes that take place in the interaction between an organism and a drug. One of these models, the multi-compartment model, gives the best approximation to reality; however, the complexity involved in using this type of model means that monocompartmental models and above all two compartmental models are the most-frequently used. The various compartments that the model is divided into are commonly referred to as the ADME scheme (also referred to as LADME if liberation is included as a separate step from absorption): Liberation - the process of release of a drug from the pharmaceutical formulation. See also IVIVC. Absorption - the process of a substance entering the blood circulation. Distribution - the dispersion or dissemination of substances throughout the fluids and tissues of the body. Metabolization (or biotransformation, or inactivation) – the recognition by the organism that a foreign substance is present and the irreversible transformation of parent compounds into daughter metabolites. Excretion - the removal of the substances from the body. In rare cases, some drugs irreversibly accumulate in body tissue.The two phases of metabolism and excretion can also be grouped together under the title elimination.The study of these distinct phases involves the use and manipulation of basic concepts in order to understand the process dynamics. For this reason in order to fully comprehend the kinetics of a drug it is necessary to have detailed knowledge of a number of factors such as: the properties of the substances that act as excipients, the characteristics of the appropriate biological membranes and the way that substances can cross them, or the characteristics of the enzyme reactions that inactivate the drug.All these concepts can be represented through mathematical formulas that have a corresponding graphical representation. The use of these models allows an understanding of the characteristics of a molecule, as well as how a particular drug will behave given information regarding some of its basic characteristics. Such as its acid dissociation constant (pKa), bioavailability and solubility, absorption capacity and distribution in the organism.The model outputs for a drug can be used in industry (for example, in calculating bioequivalence when designing generic drugs) or in the clinical application of pharmacokinetic concepts. Clinical pharmacokinetics provides many performance guidelines for effective and efficient use of drugs for human-health professionals and in veterinary medicine.













![[27] Kumar, RMNV- Eds. Handbook of Particulate Drug Delivery,2008](http://s1.studyres.com/store/data/002367987_1-7fa2f44d01cdb2370574c079aacf4d42-300x300.png)








