Chemical Bonds Study Guide Answer Key
... 9. oxidation number- the charge on an ion 10. metallic bonds- describes how metal atoms are held together in a solid 11. alloy- a combination of 2 or more elements, at least one is a metal 12. covalent bonds- bonds formed when nonmetals share electrons 13. polar bonds- covalent bonds that have a pos ...
... 9. oxidation number- the charge on an ion 10. metallic bonds- describes how metal atoms are held together in a solid 11. alloy- a combination of 2 or more elements, at least one is a metal 12. covalent bonds- bonds formed when nonmetals share electrons 13. polar bonds- covalent bonds that have a pos ...
chapter
... • Substances that can’t be broken down into simpler substances by ordinary chemical reactions • Each element has a chemical symbol (Example: C for carbon) • Four elements (oxygen, carbon, hydrogen, and nitrogen) make up more than 96% of the mass of most organisms • Calcium, phosphorus, potassium, an ...
... • Substances that can’t be broken down into simpler substances by ordinary chemical reactions • Each element has a chemical symbol (Example: C for carbon) • Four elements (oxygen, carbon, hydrogen, and nitrogen) make up more than 96% of the mass of most organisms • Calcium, phosphorus, potassium, an ...
Phy 211: General Physics I
... 1. the chemical symbol for each of the elements present 2. The # of atoms of each element present in the molecule Examples of chemical formulas: – Elemental oxygen: O2 (2 O atoms per molecule) – Water: H2O (2 H atoms & 1 O atom) – Aluminum sulfate: Al2(SO4)3 (2 Al, 3 S & 12 O atoms) ...
... 1. the chemical symbol for each of the elements present 2. The # of atoms of each element present in the molecule Examples of chemical formulas: – Elemental oxygen: O2 (2 O atoms per molecule) – Water: H2O (2 H atoms & 1 O atom) – Aluminum sulfate: Al2(SO4)3 (2 Al, 3 S & 12 O atoms) ...
Intermolecular Attractions
... Draw the electron dot formula. Then state how many bonding and unbonding pairs are present. A) NBr3 B) Water C) Chlorite ion (ClO2- ) D) CF2Cl2 ...
... Draw the electron dot formula. Then state how many bonding and unbonding pairs are present. A) NBr3 B) Water C) Chlorite ion (ClO2- ) D) CF2Cl2 ...
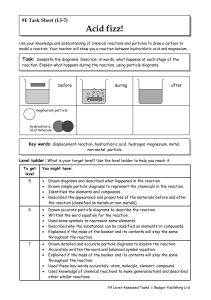
Year 9 Homework Task 9E-5 Reactions 5-7
... Drawn detailed and accurate particle diagrams to explain the reaction. Accurately written the word and balanced symbol equation. Explained if the mass of the beaker and its contents will stay the same throughout the reaction. Used these key words accurately: atom, molecule, element, compound. Used k ...
... Drawn detailed and accurate particle diagrams to explain the reaction. Accurately written the word and balanced symbol equation. Explained if the mass of the beaker and its contents will stay the same throughout the reaction. Used these key words accurately: atom, molecule, element, compound. Used k ...
Unit 1 Lecture PPT
... – has a constant, invariable composition – It’s identified by a formula or symbol – Examples water- H2O – always has 2 hydrogens and 1 oxygen Helium - He ...
... – has a constant, invariable composition – It’s identified by a formula or symbol – Examples water- H2O – always has 2 hydrogens and 1 oxygen Helium - He ...
Types of Changes in Matter
... Update ALL atom counts after adding a coefficient. If an element appears more than once per side, balance it last. Balance polyatomic ions as single units. “1 SO4” instead of “1 S” and “4 O” ...
... Update ALL atom counts after adding a coefficient. If an element appears more than once per side, balance it last. Balance polyatomic ions as single units. “1 SO4” instead of “1 S” and “4 O” ...
Balancing Chemical Equations
... Because of the law of conservation of matter, these equations must be balanced. In other words, the number of atoms of each element must be conserved. For example, look at the reaction of hydrogen and oxygen to produce water: H2 + O2 → H2O. The reactant side of the equation contains two atoms of hyd ...
... Because of the law of conservation of matter, these equations must be balanced. In other words, the number of atoms of each element must be conserved. For example, look at the reaction of hydrogen and oxygen to produce water: H2 + O2 → H2O. The reactant side of the equation contains two atoms of hyd ...
Chemical Equations and Reaction Types Lab
... *Note: The seven diatomic elements, when uncombined, are written with subscripts of 2 (H2; O2; etc.) 3) Determine the products and write the correct formula for each product. Once the correct formula is written it must not be changed during the subsequent balancing operation. 4) Balance the chemical ...
... *Note: The seven diatomic elements, when uncombined, are written with subscripts of 2 (H2; O2; etc.) 3) Determine the products and write the correct formula for each product. Once the correct formula is written it must not be changed during the subsequent balancing operation. 4) Balance the chemical ...
... • Yes, but it is the empirical formula, not the molecular formula. • An empirical formula only gives the smallest wholenumber ratio of each type of atom in a compound, not the specific number of each type of atom in a molecule. • The molecular formula is always a whole-number multiple of the empiric ...
Name ……………………………..………...… …….. Index No
... 1 (a)The electron arrangement of ions A3+ and B2- are 2.8 and 2.8.8 respectively,Write down the electron arrangement of the elements A and B.(2mks) ...
... 1 (a)The electron arrangement of ions A3+ and B2- are 2.8 and 2.8.8 respectively,Write down the electron arrangement of the elements A and B.(2mks) ...
Organic Molecules
... structures containing thousands of atoms! Although carbon is present in all organic compounds, other elements such as hydrogen (H), oxygen (O), nitrogen (N), sulfur (S) and phosphorus (P) are also common in these molecules. Until the early nineteenth century, chemists had managed to make many simple ...
... structures containing thousands of atoms! Although carbon is present in all organic compounds, other elements such as hydrogen (H), oxygen (O), nitrogen (N), sulfur (S) and phosphorus (P) are also common in these molecules. Until the early nineteenth century, chemists had managed to make many simple ...
Electrochemistry Lecture
... exception are O, which never has an Ox# of +6 and F, which never has an Ox# of +7.) For nonmetals, (brown) and metalloids (green) the A group number minus 8 gives the lowest possible oxidation number ...
... exception are O, which never has an Ox# of +6 and F, which never has an Ox# of +7.) For nonmetals, (brown) and metalloids (green) the A group number minus 8 gives the lowest possible oxidation number ...
MIDTERM REVIEW UNIT 1: Mass/Measurement
... 4. What is the atomic mass of Hafnium if out of every 200 atoms, 10 have mass 176.00 g/mol, 38 have mass 177.00 g/mol, 54 have mass 178.00 g/mol, 28 have mass 179.00 g/mol, and 70 have mass 180.00 g/mol? ...
... 4. What is the atomic mass of Hafnium if out of every 200 atoms, 10 have mass 176.00 g/mol, 38 have mass 177.00 g/mol, 54 have mass 178.00 g/mol, 28 have mass 179.00 g/mol, and 70 have mass 180.00 g/mol? ...
eBook AQA GCSE Chemistry Unit C2 Part 1
... made up of two or more atoms chemically bonded together. In ammonia, each molecule consists of one atom of nitrogen joined to three atoms of hydrogen. The atoms are held together by covalent bonds. A covalent bond is a shared pair of electrons. Covalent bonds form so that atoms can achieve stable el ...
... made up of two or more atoms chemically bonded together. In ammonia, each molecule consists of one atom of nitrogen joined to three atoms of hydrogen. The atoms are held together by covalent bonds. A covalent bond is a shared pair of electrons. Covalent bonds form so that atoms can achieve stable el ...
Summer_Assignment_AP_Chemistry_TW 2015
... Strong Acids and Bases Strong acids dissociate completely in water, so the reaction goes to completion and they never reach equilibrium with their conjugate bases. Because there is no equilibrium, there is no equilibrium constant, so there is no dissociation constant for strong acids and bases. Stro ...
... Strong Acids and Bases Strong acids dissociate completely in water, so the reaction goes to completion and they never reach equilibrium with their conjugate bases. Because there is no equilibrium, there is no equilibrium constant, so there is no dissociation constant for strong acids and bases. Stro ...
Practice EXAM I
... negative charge indicates there are more electrons than protons. This is because neutron does not carry charge, a proton carries one positive charge, and an electron carries one negative charge. The ion in this question carries one negative charge indicating that the electron is one more than the pr ...
... negative charge indicates there are more electrons than protons. This is because neutron does not carry charge, a proton carries one positive charge, and an electron carries one negative charge. The ion in this question carries one negative charge indicating that the electron is one more than the pr ...
AP Chemistry Summer Assignment
... 42. You have 6 moles of silver ions and 150 g of copper (Cu). How many grams of silver (Ag) can you recover? Is this enough copper to react with all 6 moles of silver ions? ...
... 42. You have 6 moles of silver ions and 150 g of copper (Cu). How many grams of silver (Ag) can you recover? Is this enough copper to react with all 6 moles of silver ions? ...
GCSE_C2_Revision_+_Exam_Questions
... well in your science GCSE you will need to know all of the stuff on this revision booklet. ...
... well in your science GCSE you will need to know all of the stuff on this revision booklet. ...
File
... A) Ca(s) B) Hg( ) C) C12(g) D) CaC1 2(aq) 97. The burning of magnesium involves a conversion of A) chemical energy to mechanical energy B) chemical energy to heat energy C) heat energy to chemical energy D) heat energy to mechanical energy 98. Which term is defined as a measure of the average kineti ...
... A) Ca(s) B) Hg( ) C) C12(g) D) CaC1 2(aq) 97. The burning of magnesium involves a conversion of A) chemical energy to mechanical energy B) chemical energy to heat energy C) heat energy to chemical energy D) heat energy to mechanical energy 98. Which term is defined as a measure of the average kineti ...
Chapter 2
... • The periodic table of the elements shows the electron distribution for each element ...
... • The periodic table of the elements shows the electron distribution for each element ...
The representative Elements: Groups 1A – 4A
... 1. Pure CaCO3 are used as fillers in paint, toothpaste, paper, plastics, etc.; 2. the source for calcium metal and quicklime: Calcination: CaCO3(s) CaO(s) + CO2(g) ...
... 1. Pure CaCO3 are used as fillers in paint, toothpaste, paper, plastics, etc.; 2. the source for calcium metal and quicklime: Calcination: CaCO3(s) CaO(s) + CO2(g) ...