Students will review concepts from their quiz and then correct it at
... Two or more __elements___ or ____compounds__ NOT chemically combined. No reaction between substances. Mixtures can be uniform (called __homogeneous___) and are known as solutions. Mixtures can also be non-uniform (called ____heterogeneous____). Mixtures can be separated into their components by chem ...
... Two or more __elements___ or ____compounds__ NOT chemically combined. No reaction between substances. Mixtures can be uniform (called __homogeneous___) and are known as solutions. Mixtures can also be non-uniform (called ____heterogeneous____). Mixtures can be separated into their components by chem ...
AP Chemistry Ch. 3 Sections 3.7-3.8 Notes Chemical Equations
... All atoms present in the reactants must be accounted for among the products. In other words, there must be the same number of each type of atom on the product side and on the reactant side of the arrow. Known as balancing a chemical equation. CH4 + O2 → CO2 + H2O This equation is not balanced. Notic ...
... All atoms present in the reactants must be accounted for among the products. In other words, there must be the same number of each type of atom on the product side and on the reactant side of the arrow. Known as balancing a chemical equation. CH4 + O2 → CO2 + H2O This equation is not balanced. Notic ...
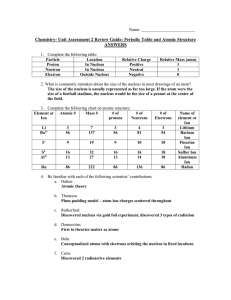
Chem Unit 2 Review Guide ANSWERS
... Anions are negatively charged particles that have gained electrons. Cations are positively charged particles that have lost electrons. 21.) Solve the following half life problems: a.- Sodium-24 has a half-life of 15 hours. How much sodium-24 will remain in an 18.0g sample after 60 hours? 4 half live ...
... Anions are negatively charged particles that have gained electrons. Cations are positively charged particles that have lost electrons. 21.) Solve the following half life problems: a.- Sodium-24 has a half-life of 15 hours. How much sodium-24 will remain in an 18.0g sample after 60 hours? 4 half live ...
Notes matter energy
... Exercise: What are the main group metals? Solution: The main group is group A. Main group metals includes the elements Li and below in group IA, the elements Be and below in group IIA, the elements of Al and below in group IIIA, the elements Sn and below in group IVA, the elements Bi and below in gr ...
... Exercise: What are the main group metals? Solution: The main group is group A. Main group metals includes the elements Li and below in group IA, the elements Be and below in group IIA, the elements of Al and below in group IIIA, the elements Sn and below in group IVA, the elements Bi and below in gr ...
Atom (A) or Ion
... 82. What is pH and how is it measured? 83. What factors affect solubility? 84. What is molarity? 85. If I have 2.5 mol of calcium carbonate in .3 L of solution, what is the molarity? 86. If I have 700 mL of a 5 M NaOH solution, how many grams of NaOH were used to make the solution? 87. What are coll ...
... 82. What is pH and how is it measured? 83. What factors affect solubility? 84. What is molarity? 85. If I have 2.5 mol of calcium carbonate in .3 L of solution, what is the molarity? 86. If I have 700 mL of a 5 M NaOH solution, how many grams of NaOH were used to make the solution? 87. What are coll ...
Atom (A) or Ion (I)
... 82. What is pH and how is it measured? 83. What factors affect solubility? 84. What is molarity? 85. If I have 2.5 mol of calcium carbonate in .3 L of solution, what is the molarity? 86. If I have 700 mL of a 5 M NaOH solution, how many grams of NaOH were used to make the solution? 87. What are coll ...
... 82. What is pH and how is it measured? 83. What factors affect solubility? 84. What is molarity? 85. If I have 2.5 mol of calcium carbonate in .3 L of solution, what is the molarity? 86. If I have 700 mL of a 5 M NaOH solution, how many grams of NaOH were used to make the solution? 87. What are coll ...
Atom (A) or Ion (I)
... 82. What is pH and how is it measured? 83. What factors affect solubility? 84. What is molarity? 85. If I have 2.5 mol of calcium carbonate in .3 L of solution, what is the molarity? 86. If I have 700 mL of a 5 M NaOH solution, how many grams of NaOH were used to make the solution? 87. What are coll ...
... 82. What is pH and how is it measured? 83. What factors affect solubility? 84. What is molarity? 85. If I have 2.5 mol of calcium carbonate in .3 L of solution, what is the molarity? 86. If I have 700 mL of a 5 M NaOH solution, how many grams of NaOH were used to make the solution? 87. What are coll ...
Sample % Sulfate Absolute Deviation A 44.02 B 44.11 C 43.98 D
... follow different rules. Additionally, some compounds (H2O, NH3, CH4, etc.) simply have common names that must be memorized. The two types of compounds we will focus on first are ionic compounds (formed from positive and negative ions) and binary nonmetal compounds (molecular compounds). Later we wil ...
... follow different rules. Additionally, some compounds (H2O, NH3, CH4, etc.) simply have common names that must be memorized. The two types of compounds we will focus on first are ionic compounds (formed from positive and negative ions) and binary nonmetal compounds (molecular compounds). Later we wil ...
1 - shawnschmitt
... 17. What is the total number of valence electrons of an atom of iodine in the ground state? there are 7 valence electrons in an atom of iodine 18. Draw the Lewis dot structure of arsenic? draw As surrounded by 5 dots (there should only be 1 pair) 19. How many valence electrons are in an atom with th ...
... 17. What is the total number of valence electrons of an atom of iodine in the ground state? there are 7 valence electrons in an atom of iodine 18. Draw the Lewis dot structure of arsenic? draw As surrounded by 5 dots (there should only be 1 pair) 19. How many valence electrons are in an atom with th ...
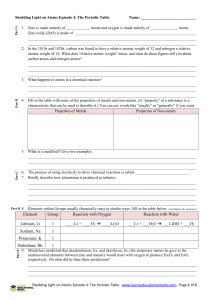
Element Group Reaction with Oxygen Reaction with Water Lithium
... 12. Trends in the Periodic Table are apparent both across each Period and down each Group. Using two lines (one for boiling point and one for melting point), graph the data in the table on the right onto the graph below. Use a colour code. Use the graph to estimate the melting point and boiling poin ...
... 12. Trends in the Periodic Table are apparent both across each Period and down each Group. Using two lines (one for boiling point and one for melting point), graph the data in the table on the right onto the graph below. Use a colour code. Use the graph to estimate the melting point and boiling poin ...
ap chemistry unit two notes
... Alkaline Earth Metals - Group IIA • Elements also react with water to form a base. •Forms +2 ions (Cations). •“s” sublevel electrons. Ca(s) + 2H2O(l) → Ca(OH)2(aq) + H2(g) ...
... Alkaline Earth Metals - Group IIA • Elements also react with water to form a base. •Forms +2 ions (Cations). •“s” sublevel electrons. Ca(s) + 2H2O(l) → Ca(OH)2(aq) + H2(g) ...
NM Strand
... 1. The label on a popular brand of “one-a-day” vitamins says there are 15mg of zinc per tablet. How many grams of zinc are in one tablet? 2. If an atom loses an electron, it becomes a(n) 3. What elements exist as diatomic molecules? 4. What is 1,230 in scientific notation? 5. A salt solution has a c ...
... 1. The label on a popular brand of “one-a-day” vitamins says there are 15mg of zinc per tablet. How many grams of zinc are in one tablet? 2. If an atom loses an electron, it becomes a(n) 3. What elements exist as diatomic molecules? 4. What is 1,230 in scientific notation? 5. A salt solution has a c ...
rp oc4
... 19. Another name for a homogeneous mixture is a(n) 20. What are four indicators of a chemical reaction? ...
... 19. Another name for a homogeneous mixture is a(n) 20. What are four indicators of a chemical reaction? ...
Safety - Wando High School
... 6. What is an elements oxidation number(s)? 7. What rule does bonding (typically) fulfill to create stability? 8. Ionic compounds form what type of structure? 9. Metallic, Covalent, and Ionic bonds have what properties? 10. What are the 3 types of intermolecular forces from strongest to weakest? 11. ...
... 6. What is an elements oxidation number(s)? 7. What rule does bonding (typically) fulfill to create stability? 8. Ionic compounds form what type of structure? 9. Metallic, Covalent, and Ionic bonds have what properties? 10. What are the 3 types of intermolecular forces from strongest to weakest? 11. ...
Midterm Review Date
... 44. Which substance contains bonds that involved the transfer of electrons from one atom to another? A) CO 2 B) NH 3 C) KBr D) Cl 2 45. Based on bond type, which compound has the highest melting point? A) CH 3OH C) CaCl2 ...
... 44. Which substance contains bonds that involved the transfer of electrons from one atom to another? A) CO 2 B) NH 3 C) KBr D) Cl 2 45. Based on bond type, which compound has the highest melting point? A) CH 3OH C) CaCl2 ...
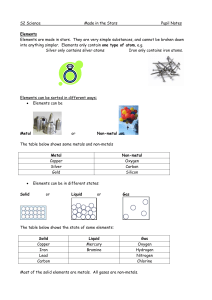
Properties of Metals vs. Nonmetals vs. Metalloids
... Use electronegativity values to determine if the individual bonds in the molecules above are polar. Look at the polarity of the bonds and the symmetry of the molecules above to determine if the molecules are polar (if one side of the molecule will be more negative than another). ...
... Use electronegativity values to determine if the individual bonds in the molecules above are polar. Look at the polarity of the bonds and the symmetry of the molecules above to determine if the molecules are polar (if one side of the molecule will be more negative than another). ...
Metals
... Non‐metal with a non‐ metal with a non‐metal When non‐metals combine, they form molecules. They may do so in multiple forms: CO ...
... Non‐metal with a non‐ metal with a non‐metal When non‐metals combine, they form molecules. They may do so in multiple forms: CO ...
Writing Chemical Formulas and Chemical Reactions
... compounds can be named using the regular naming system for binary molecular compounds if they are gases. But, binary acids are usually found as clear, viscous liquids at room temperature and a different naming system is used when they are in this state. If the binary acid is in aqueous state, the pr ...
... compounds can be named using the regular naming system for binary molecular compounds if they are gases. But, binary acids are usually found as clear, viscous liquids at room temperature and a different naming system is used when they are in this state. If the binary acid is in aqueous state, the pr ...
document
... ___T_____16. If an atom’s charge is positive, it has lost electrons. ___F______17. If an atom’s charge is negative, it has lost protons. ___F______18. You balance an equation by changing subscripts. ___T______19. You can only put a coefficient in front of a compound. ___T______20. The number of atom ...
... ___T_____16. If an atom’s charge is positive, it has lost electrons. ___F______17. If an atom’s charge is negative, it has lost protons. ___F______18. You balance an equation by changing subscripts. ___T______19. You can only put a coefficient in front of a compound. ___T______20. The number of atom ...
Ch 4 Review
... ____ 22. Physical properties of matter are characteristics that a. can be observed without changing the composition of substances. b. describe reactions between substances. c. describe reactions between unreactive substances. d. can be observed only after changing the composition of substances. ____ ...
... ____ 22. Physical properties of matter are characteristics that a. can be observed without changing the composition of substances. b. describe reactions between substances. c. describe reactions between unreactive substances. d. can be observed only after changing the composition of substances. ____ ...
Made in the Stars Notes
... at room temperature except for mercury, which is a liquid. Non-metal solids are usually brittle (they break easily). Non-metals can be solids, liquids or gases at room temperature. Non-metals usually have low melting and boiling points. They are poor conductors of electricity. The exception is graph ...
... at room temperature except for mercury, which is a liquid. Non-metal solids are usually brittle (they break easily). Non-metals can be solids, liquids or gases at room temperature. Non-metals usually have low melting and boiling points. They are poor conductors of electricity. The exception is graph ...