Chemical Reactions Chemistry - is the study of matter, its properties
... Many chemicals can be hazardous to human health or the environment if they are not handled safely. There are a variety of symbols used to identify hazardous chemicals. Many household products are labeled with Hazardous Household Product Symbols (HHPS). Dangerous materials in the workplace are labele ...
... Many chemicals can be hazardous to human health or the environment if they are not handled safely. There are a variety of symbols used to identify hazardous chemicals. Many household products are labeled with Hazardous Household Product Symbols (HHPS). Dangerous materials in the workplace are labele ...
A.P. Chemistry Complexation Reactions
... (B) in a compound A single element must be more reactive to replace another element. ...
... (B) in a compound A single element must be more reactive to replace another element. ...
Limiting Reactant WS with Answers
... 8) The amino acid arginine is an essential component of all proteins. This compound contains 41.36% carbon, 8.10% hydrogen, and 32.17% nitrogen, with the remainder being oxygen. a) Determine the empirical formula of arginine. b) The molar mass of arginine is known to be between 100 and 200 g/mol. D ...
... 8) The amino acid arginine is an essential component of all proteins. This compound contains 41.36% carbon, 8.10% hydrogen, and 32.17% nitrogen, with the remainder being oxygen. a) Determine the empirical formula of arginine. b) The molar mass of arginine is known to be between 100 and 200 g/mol. D ...
Chapter 2 - Chemistry
... 4.) Add prefix, derived from the Greek, to each element name to denote subscript of element in formula. Note: generally, prefix mono- is not used, unless needed to distinguish two compounds of the same two elements Karen Hattenhauer (Fall 2007) ...
... 4.) Add prefix, derived from the Greek, to each element name to denote subscript of element in formula. Note: generally, prefix mono- is not used, unless needed to distinguish two compounds of the same two elements Karen Hattenhauer (Fall 2007) ...
- Catalyst
... Question 7: Fill in the blanks of the statements below with the words in the box. Note, you will only use each word once. 1. A positively charged ion is a(n) ____________________. 2. A proton has a +1 _______________________________. 3. Like charges _________________________ each other. ...
... Question 7: Fill in the blanks of the statements below with the words in the box. Note, you will only use each word once. 1. A positively charged ion is a(n) ____________________. 2. A proton has a +1 _______________________________. 3. Like charges _________________________ each other. ...
Chemistry a material science!
... Likewise the freezing and boiling temperature of both H2 and O2 is much lower than the corresponding freezing and boiling temperature of H2O. ...
... Likewise the freezing and boiling temperature of both H2 and O2 is much lower than the corresponding freezing and boiling temperature of H2O. ...
Chemical Formulas and Formula Weight Calculations
... When elements combine in different ratios, subscripts elements combine in different ratios subscripts are added, following the element symbol, to indicate that the number of atoms of that element in the compound if it is greater than one. • The subscript refers only to the element it immediatel ...
... When elements combine in different ratios, subscripts elements combine in different ratios subscripts are added, following the element symbol, to indicate that the number of atoms of that element in the compound if it is greater than one. • The subscript refers only to the element it immediatel ...
1st Semester Chem Final Exam Study Guide 2012-2013
... b. What is the percent composition of each of the elements in Iron (III) Iodide? ...
... b. What is the percent composition of each of the elements in Iron (III) Iodide? ...
Isotopes - Cloudfront.net
... What was happening to the air particles inside as you tried to add pressure? Why was it so hard to lift the plunger out of the syringe? Why did the marshmallow respond as it did to the changes in pressure? Why can’t you compress water? ...
... What was happening to the air particles inside as you tried to add pressure? Why was it so hard to lift the plunger out of the syringe? Why did the marshmallow respond as it did to the changes in pressure? Why can’t you compress water? ...
Science 1206 Unit 3 Part 1
... formula H2O and the empirical formula H2O since the atoms are already in the simplest form. Whereas hydrogen peroxide has the molecular formula H2O2 and the empirical formula HO. Polyatomic ion – an ion that consists of two or more different non-metal atoms that are joined by covalent bonds ...
... formula H2O and the empirical formula H2O since the atoms are already in the simplest form. Whereas hydrogen peroxide has the molecular formula H2O2 and the empirical formula HO. Polyatomic ion – an ion that consists of two or more different non-metal atoms that are joined by covalent bonds ...
Name
... 8th grade Semester 1 study guide (version B) Essential Standard 3a: The structure of the atom and how it is composed of protons, neutrons and electrons. ...
... 8th grade Semester 1 study guide (version B) Essential Standard 3a: The structure of the atom and how it is composed of protons, neutrons and electrons. ...
Atom (A) or Ion (I)
... 13. Whether atom or ion, the number of these is always the same as the atomic number. 14. In an atom, the number of these is equal to the number of protons; In an ion, the number of these is different from the number of protons. 15. Add the total of these two types of particles to get the atom's mas ...
... 13. Whether atom or ion, the number of these is always the same as the atomic number. 14. In an atom, the number of these is equal to the number of protons; In an ion, the number of these is different from the number of protons. 15. Add the total of these two types of particles to get the atom's mas ...
7R CHEMISTRY 1 REVIEW
... _____________ 1. Made up of more than one type of molecule. _____________ 2. Can be separated by physical means. _____________ 3. Made up of only one type of atom. ...
... _____________ 1. Made up of more than one type of molecule. _____________ 2. Can be separated by physical means. _____________ 3. Made up of only one type of atom. ...
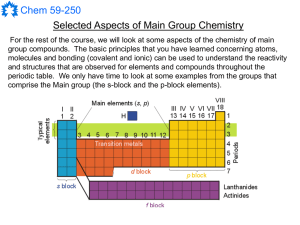
Main Group Notes 1
... These reactions are very exothermic and increase in violence from the lightest to the heaviest elements in the group (enough to ignite the H2 for the heavier elements). The non-reversible nature of this reaction means that such metals are very useful for drying many kinds of solvents. ...
... These reactions are very exothermic and increase in violence from the lightest to the heaviest elements in the group (enough to ignite the H2 for the heavier elements). The non-reversible nature of this reaction means that such metals are very useful for drying many kinds of solvents. ...
Summer - Honors Chemistry
... slide over and around each other, so a liquid is formed. A liquid has no definite shape and takes the shape of its container, but it does have a definite volume. In a solid, the attractions are so great that the particles are kept in rigidly fixed positions, able only to vibrate about these position ...
... slide over and around each other, so a liquid is formed. A liquid has no definite shape and takes the shape of its container, but it does have a definite volume. In a solid, the attractions are so great that the particles are kept in rigidly fixed positions, able only to vibrate about these position ...
Aim # 8: How do we write and balance a chemical equation?
... Does this expression, in words, convey the fact that mass has been conserved in this chemical reaction? Pb(NO3)2(aq) + 2KI(aq) → PbI2(s) + 2KNO3(aq) ...
... Does this expression, in words, convey the fact that mass has been conserved in this chemical reaction? Pb(NO3)2(aq) + 2KI(aq) → PbI2(s) + 2KNO3(aq) ...
What is Chemistry? Chemistry
... o Atoms that gain electrons to form compounds are called anions. Anions have a _________________________________. o Naming Anions: Drop the last few letters of the element name and add “ide”. o E.g. Group 17 (Halogens) gain electrons easily and release lots of energy in the process highly reactive ...
... o Atoms that gain electrons to form compounds are called anions. Anions have a _________________________________. o Naming Anions: Drop the last few letters of the element name and add “ide”. o E.g. Group 17 (Halogens) gain electrons easily and release lots of energy in the process highly reactive ...
Honors Chemistry
... 5. What is an isotope? 6. How can the number of protons be determined? Neutrons? Electrons? 7. Determine the # of p+, no and e- in the following: oxygen-14, 356210XY34- and Astatine. 8. Draw a wave. Label the wavelength and amplitude on your drawing. 9. What are the colors that make up white light? ...
... 5. What is an isotope? 6. How can the number of protons be determined? Neutrons? Electrons? 7. Determine the # of p+, no and e- in the following: oxygen-14, 356210XY34- and Astatine. 8. Draw a wave. Label the wavelength and amplitude on your drawing. 9. What are the colors that make up white light? ...
UNIT 1 - MATTER AND CHEMICAL BONDING
... f) ferrous iodide l) cobalt(III) sulphate 5. Classify each of the following reactions as synthesis, single displacement, double displacement, combustion or decomposition. a) iron + copper(I) nitrate iron(II) nitrate + copper b) phosphorus + oxygen diphosphorus pentoxide c) calcium carbonate ca ...
... f) ferrous iodide l) cobalt(III) sulphate 5. Classify each of the following reactions as synthesis, single displacement, double displacement, combustion or decomposition. a) iron + copper(I) nitrate iron(II) nitrate + copper b) phosphorus + oxygen diphosphorus pentoxide c) calcium carbonate ca ...
The Chemical Earth
... equal the number of positive charges. Ex: NaCl – the numbers of sodium and chloride ions is equal. If the charges on the ions are not equal then there will be more ions with the smaller charge. Ex: the compound formed between Ca2+ and Clis CaCl2 (there are 2 Cl ions for each Ca ion) ...
... equal the number of positive charges. Ex: NaCl – the numbers of sodium and chloride ions is equal. If the charges on the ions are not equal then there will be more ions with the smaller charge. Ex: the compound formed between Ca2+ and Clis CaCl2 (there are 2 Cl ions for each Ca ion) ...
- Catalyst
... Question 7: Fill in the blanks of the statements below with the words in the box. Note, you will only use each word once. A. atom ...
... Question 7: Fill in the blanks of the statements below with the words in the box. Note, you will only use each word once. A. atom ...
Elements, Compounds and Chemical Reactions
... The Earth is Made of Elements • From these last two pie charts, you should be able to see that our air is mostly Nitrogen, then oxygen, our crust is mostly oxygen and then silicon, and our bodies are oxygen and ...
... The Earth is Made of Elements • From these last two pie charts, you should be able to see that our air is mostly Nitrogen, then oxygen, our crust is mostly oxygen and then silicon, and our bodies are oxygen and ...