Balancing Chemical Equations
... molecule. A coefficient pertains to every element in the compound, regardless of parenthesis. The example below is telling us that there are 2 oxygen molecules (O2) in the reactants. That is a total of 4 oxygen atoms. Ex. CH4 + 2O2 CO2 + 2H2O C=1 C=1 H=4 H=4 O=4 O=4 The equation is now balanced ...
... molecule. A coefficient pertains to every element in the compound, regardless of parenthesis. The example below is telling us that there are 2 oxygen molecules (O2) in the reactants. That is a total of 4 oxygen atoms. Ex. CH4 + 2O2 CO2 + 2H2O C=1 C=1 H=4 H=4 O=4 O=4 The equation is now balanced ...
Bonding. A. Ionic bonds form when anions and cations arise
... Covalent bonds form when it is not possible for electrons to be transferred and so must be shared between atoms. 1. Generally this is the case when two nonmetals bond. The tendency of nonmetals is to gain electrons according to the octet rule, a very easy thing to accomplish when they bond with meta ...
... Covalent bonds form when it is not possible for electrons to be transferred and so must be shared between atoms. 1. Generally this is the case when two nonmetals bond. The tendency of nonmetals is to gain electrons according to the octet rule, a very easy thing to accomplish when they bond with meta ...
The Chemical Basis of Life
... Chemical reactions involve making and breaking bonds. Making bonds requires the input of energy. Energy is stored in molecules in the chemical bonds. Breaking bonds usually releases energy. ...
... Chemical reactions involve making and breaking bonds. Making bonds requires the input of energy. Energy is stored in molecules in the chemical bonds. Breaking bonds usually releases energy. ...
Basics of Chemistry
... Matter is made of atoms Life requires ~25 chemical elements Atomic structure determines behavior of an element ...
... Matter is made of atoms Life requires ~25 chemical elements Atomic structure determines behavior of an element ...
Science notes on Atoms, Periodic table
... Lithium is a metal, it has 1 valence electron therefore it wants to give away 1 electron. All metals want to have 8 valence electrons. Carbon is a non-metal, it has 6 valence electrons & it wants to have 8 therefore it takes 2 valence electrons. Li1+ & C2- cross over rule & becomes Li2C (lithium c ...
... Lithium is a metal, it has 1 valence electron therefore it wants to give away 1 electron. All metals want to have 8 valence electrons. Carbon is a non-metal, it has 6 valence electrons & it wants to have 8 therefore it takes 2 valence electrons. Li1+ & C2- cross over rule & becomes Li2C (lithium c ...
Preliminary Course Atomic Structure 1 + 2
... So Calcium would have to react with TWO clorines – Calcium forms CaCl2, not CaCl ...
... So Calcium would have to react with TWO clorines – Calcium forms CaCl2, not CaCl ...
Ch 02.01-03: Atoms Molecules Ions
... 1. Each element is composed of tiny, indestructible particles called atoms. 2. All atoms of a given element have the same mass and other properties that distinguish them from the atoms of other elements. 3. Atoms combine in simple, whole-number ratios to form compounds. 4. Atoms of one element canno ...
... 1. Each element is composed of tiny, indestructible particles called atoms. 2. All atoms of a given element have the same mass and other properties that distinguish them from the atoms of other elements. 3. Atoms combine in simple, whole-number ratios to form compounds. 4. Atoms of one element canno ...
UC Chapter 6 Study Guide
... The test has 3 parts: Vocabulary, science concepts, and inquiry skills. The inquiry skills and/or critical thinking section is where the student has to answer short answer questions from the concepts in the chapter-these are not provided. There are 4 questions. Vocab: Atom – smallest possible partic ...
... The test has 3 parts: Vocabulary, science concepts, and inquiry skills. The inquiry skills and/or critical thinking section is where the student has to answer short answer questions from the concepts in the chapter-these are not provided. There are 4 questions. Vocab: Atom – smallest possible partic ...
Ch. 3 - Chemical Reactions
... Balancing Equation Steps 1. Write the unbalanced equation. 2. Count atoms on each side. 3. Add coefficients to make #s equal. Coefficient subscript = # of atoms 4. Reduce coefficients to lowest possible ratio, if necessary. ...
... Balancing Equation Steps 1. Write the unbalanced equation. 2. Count atoms on each side. 3. Add coefficients to make #s equal. Coefficient subscript = # of atoms 4. Reduce coefficients to lowest possible ratio, if necessary. ...
05 Chemistry Basics with Flips 2011
... Pair of electrons not shared equally by 2 atoms Water = O + H oxygen has stronger “attraction” for the shared electrons than hydrogen oxygen has higher ...
... Pair of electrons not shared equally by 2 atoms Water = O + H oxygen has stronger “attraction” for the shared electrons than hydrogen oxygen has higher ...
CHEMICAL REACTIONS
... way to balance O with a whole number, double all the coefficients and try again. (Because O is diatomic as an element) • (Shortcut) Polyatomic ions that appear on both sides of the equation should be balanced as independent units ...
... way to balance O with a whole number, double all the coefficients and try again. (Because O is diatomic as an element) • (Shortcut) Polyatomic ions that appear on both sides of the equation should be balanced as independent units ...
How do chemists determine the formula of
... This will contain 60.80g of Na, 28.60 grams of B and 10.60 grams H 2) Determine the number of moles of each 3) Determine the simplest whole number ratio ...
... This will contain 60.80g of Na, 28.60 grams of B and 10.60 grams H 2) Determine the number of moles of each 3) Determine the simplest whole number ratio ...
Atomic Theory Practice Test
... ____ 18. The electrons involved in the formation of a chemical bond are called a. dipoles. c. Lewis electrons. b. s electrons. d. valence electrons. ____ 19. In a chemical bond, the link between atoms results from the attraction between electrons and a. Lewis structures. c. van der Waals forces. b. ...
... ____ 18. The electrons involved in the formation of a chemical bond are called a. dipoles. c. Lewis electrons. b. s electrons. d. valence electrons. ____ 19. In a chemical bond, the link between atoms results from the attraction between electrons and a. Lewis structures. c. van der Waals forces. b. ...
Chemical formulae Worksheet
... 2. Name the second element with an –ide: fluorine fluoride 3. Add a prefix to each name to indicate the subscript monoboron, trifluoride 4. Write the first element with prefix, then the second element with prefix 5. Drop prefix mono from first element: boron trifluoride 1 = mono-not used on first ...
... 2. Name the second element with an –ide: fluorine fluoride 3. Add a prefix to each name to indicate the subscript monoboron, trifluoride 4. Write the first element with prefix, then the second element with prefix 5. Drop prefix mono from first element: boron trifluoride 1 = mono-not used on first ...
CHEMICAL FORMULAE AND EQUATIONS Molecule Empirical
... 2. Name the second element with an –ide: fluorine fluoride 3. Add a prefix to each name to indicate the subscript monoboron, trifluoride 4. Write the first element with prefix, then the second element with prefix 5. Drop prefix mono from first element: boron trifluoride 1 = mono-not used on first ...
... 2. Name the second element with an –ide: fluorine fluoride 3. Add a prefix to each name to indicate the subscript monoboron, trifluoride 4. Write the first element with prefix, then the second element with prefix 5. Drop prefix mono from first element: boron trifluoride 1 = mono-not used on first ...
Mongar Higher Secondary School
... b) An atom X is in second period and group II A of the periodic table. i) What is the number of valence electrons in its atom? ...
... b) An atom X is in second period and group II A of the periodic table. i) What is the number of valence electrons in its atom? ...
General Chemistry First Semester Review General
... - aqueous (aq) is written if a solution is used - pure liquids (not a mixture of something) use (l) - solid: This could refer to a multitude of different substances: metals, flakes, crystals, and precipitates; use (s) - gas: Use (g), these are usually diatomic molecules such as O2, H2, Cl2, etc. - r ...
... - aqueous (aq) is written if a solution is used - pure liquids (not a mixture of something) use (l) - solid: This could refer to a multitude of different substances: metals, flakes, crystals, and precipitates; use (s) - gas: Use (g), these are usually diatomic molecules such as O2, H2, Cl2, etc. - r ...
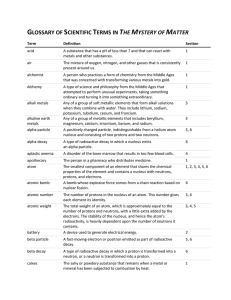
GLOSSARY OF SCIENTIFIC TERMS IN THE MYSTERY OF MATTER
... A group of two or more atoms linked together by sharing electrons in a chemical bond. A heavy, neutral particle in an atom’s nucleus that accounts for almost all of each atom’s mass, in addition to protons. Any of the six gases helium, neon, argon, krypton, xenon, and radon. Because the outermost el ...
... A group of two or more atoms linked together by sharing electrons in a chemical bond. A heavy, neutral particle in an atom’s nucleus that accounts for almost all of each atom’s mass, in addition to protons. Any of the six gases helium, neon, argon, krypton, xenon, and radon. Because the outermost el ...
Word Equations • a summary
... Reactions in which one element “displaces” or replaces another in a compound. The general formula is an element reacting with a compound to produce a new element and a new compound. A metal (cation) can displace another metal (cation) or hydrogen: X + YZ Y + XZ eg. 1 Mg + ZnCl2 Zn + MgCl2 ...
... Reactions in which one element “displaces” or replaces another in a compound. The general formula is an element reacting with a compound to produce a new element and a new compound. A metal (cation) can displace another metal (cation) or hydrogen: X + YZ Y + XZ eg. 1 Mg + ZnCl2 Zn + MgCl2 ...
File
... its own unique properties is formed. Good evidence is when a new gas is formed; heat or light is produced; a precipitate is formed; rusting/tarnishing 38. Was the zinc and hydrochloric acid combination a chemical reaction? What were some indicators? Yes because a new gas was formed 39. If I combined ...
... its own unique properties is formed. Good evidence is when a new gas is formed; heat or light is produced; a precipitate is formed; rusting/tarnishing 38. Was the zinc and hydrochloric acid combination a chemical reaction? What were some indicators? Yes because a new gas was formed 39. If I combined ...