Chapter 1: Chemistry and You
... 2015-2016 Chemistry Midterm Review This review sheet is a list of topics and sample practice problems only. The practice problems are good representation of what to expect on the midterm, but it is not enough to just study from the review. You need to look over your notes, old review sheets, tests a ...
... 2015-2016 Chemistry Midterm Review This review sheet is a list of topics and sample practice problems only. The practice problems are good representation of what to expect on the midterm, but it is not enough to just study from the review. You need to look over your notes, old review sheets, tests a ...
Honors Chemistry
... 2. In single and double replacement reactions, reactants that are compounds are always aqueous. 3. In single and double replacement reactions, products that are compounds should have their phases identified using a solubility chart (aqueous vs. precipitate) 4. In synthesis and decomposition reaction ...
... 2. In single and double replacement reactions, reactants that are compounds are always aqueous. 3. In single and double replacement reactions, products that are compounds should have their phases identified using a solubility chart (aqueous vs. precipitate) 4. In synthesis and decomposition reaction ...
SCIENCE 10: Chemical Reactions – Atomic Structure
... Metals ions lose electrons to form positive ions. Non-metals gain electrons to form negative ions. The resulting compound must be electrically neutral: the positive charges must equal the negative charges. Example: magnesium combines with chlorine to form the ionic compound magnesium chloride ...
... Metals ions lose electrons to form positive ions. Non-metals gain electrons to form negative ions. The resulting compound must be electrically neutral: the positive charges must equal the negative charges. Example: magnesium combines with chlorine to form the ionic compound magnesium chloride ...
Steady state
... needed to produce an electric current. 3) For a steady current to flow in a wire, the wire must be part of a closed circuit. 4) The electric field is constant along all parts of the circuit when a steady current is flowing. 5) The electric current in a wire is proportional to the drift velocity of t ...
... needed to produce an electric current. 3) For a steady current to flow in a wire, the wire must be part of a closed circuit. 4) The electric field is constant along all parts of the circuit when a steady current is flowing. 5) The electric current in a wire is proportional to the drift velocity of t ...
Jackson 4.10 Homework Problem Solution
... We could have guessed this form of the solution based on the symmetry of the problem, but it is often safer and more instructive to go through all the steps. The right hemisphere is a separate region and can be now solved separately. In the region between the shells, we can tell that there are no fr ...
... We could have guessed this form of the solution based on the symmetry of the problem, but it is often safer and more instructive to go through all the steps. The right hemisphere is a separate region and can be now solved separately. In the region between the shells, we can tell that there are no fr ...
AP Chapter Five Outline
... C. Net Ionic Equations: Equations that includes only the symbols or formulas of ions in solution or compounds that undergo change. Spectator ions, or ions that are present in the reaction but to not undergo any change are ignored. ...
... C. Net Ionic Equations: Equations that includes only the symbols or formulas of ions in solution or compounds that undergo change. Spectator ions, or ions that are present in the reaction but to not undergo any change are ignored. ...
Jean-Charles Matéo-Vélez - Institut de Mathématiques de Toulouse
... acceleration of positive and negative ions due to the strong electric field (Lorentz force), collisions with the neutral molecules of air, ionic wind effect, competition between positive and negative ionic wind. ...
... acceleration of positive and negative ions due to the strong electric field (Lorentz force), collisions with the neutral molecules of air, ionic wind effect, competition between positive and negative ionic wind. ...
Chemical and Molecular Formulas PPT
... The Law of Definite Proportions states that in samples of any chemical compound, the masses of the elements are always in the same proportions (consistent with Dalton’s Atomic Theoryatoms combine in simple whole-number ...
... The Law of Definite Proportions states that in samples of any chemical compound, the masses of the elements are always in the same proportions (consistent with Dalton’s Atomic Theoryatoms combine in simple whole-number ...
Chemical Potential
... The surfaces of large proteins, nucleic acids, cell membranes, and many other surfaces relevant to biology, are often charged. The charges are often important for solubulizing the proteins or membranes. Those charged surfaces, when immersed in solution where ions are present, will attract a thin `at ...
... The surfaces of large proteins, nucleic acids, cell membranes, and many other surfaces relevant to biology, are often charged. The charges are often important for solubulizing the proteins or membranes. Those charged surfaces, when immersed in solution where ions are present, will attract a thin `at ...
Does electrical double layer formation lead to salt exclusion or to
... the natures of the charge-forming processes. In the ideal case, in order to apply an external potential difference between two metal electrodes, these interfaces should be polarizable, implying that the applied potential does not leak away by Faradaic currents 共there are no interfacial redox reactio ...
... the natures of the charge-forming processes. In the ideal case, in order to apply an external potential difference between two metal electrodes, these interfaces should be polarizable, implying that the applied potential does not leak away by Faradaic currents 共there are no interfacial redox reactio ...
PRACTICE EXAM 1 for Midterm 1
... charged conductor A) is always parallel to the surface. B) is always zero because the electric field is zero inside conductors. C) is always perpendicular to the surface of the conductor. D) is perpendicular to the surface of the conductor only if it is a sphere, a cylinder, or a flat sheet. E) can ...
... charged conductor A) is always parallel to the surface. B) is always zero because the electric field is zero inside conductors. C) is always perpendicular to the surface of the conductor. D) is perpendicular to the surface of the conductor only if it is a sphere, a cylinder, or a flat sheet. E) can ...
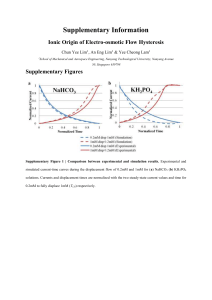
Supplementary Information
... Equation S9) is usually ignored and Stokes flow is assumed. Since the electrical double layer (EDL) thickness is small compared to the characteristic size of the experimental microchannel, flow inside the EDL is excluded in our numerical model. As such, we model only the flow of the bulk fluid (with ...
... Equation S9) is usually ignored and Stokes flow is assumed. Since the electrical double layer (EDL) thickness is small compared to the characteristic size of the experimental microchannel, flow inside the EDL is excluded in our numerical model. As such, we model only the flow of the bulk fluid (with ...
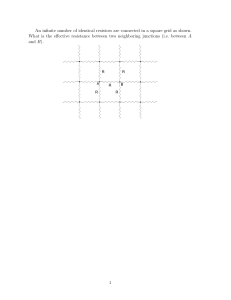
An infinite number of identical resistors are connected in a square
... If current +I0 is injected at point A and allowed to flow to infinity, each of the resistors connected directly to point A will, by symmetry, carry I0 /4 directed away from A. Similarly, if current −I0 is injected at point B, each resistor connected to B will carry current I0 /4 directed toward B. ...
... If current +I0 is injected at point A and allowed to flow to infinity, each of the resistors connected directly to point A will, by symmetry, carry I0 /4 directed away from A. Similarly, if current −I0 is injected at point B, each resistor connected to B will carry current I0 /4 directed toward B. ...
Nanofluidic circuitry
Nanofluidic circuitry is a nanotechnology aiming for control of fluids in nanometer scale. Due to the effect of an electrical double layer within the fluid channel, the behavior of nanofluid is observed to be significantly different compared with its microfluidic counterparts. Its typical characteristic dimensions fall within the range of 1–100 nm. At least one dimension of the structure is in nanoscopic scale. Phenomena of fluids in nano-scale structure are discovered to be of different properties in electrochemistry and fluid dynamics.