Chemical Reactions Notes-1a-1
... Both KI(aq) + Pb(NO3)2(aq) are colorless solutions. When mixed, they form a bright yellow precipitate of PbI2 and a solution of KNO3. ...
... Both KI(aq) + Pb(NO3)2(aq) are colorless solutions. When mixed, they form a bright yellow precipitate of PbI2 and a solution of KNO3. ...
MYP 10 PeriodicityWS
... 5(a) Draw a diagram to show the structure of sodium chloride. Explain, in terms of bonding, why sodium chloride has a high melting point. (b) Lithium reacts with water. Write an equation for the reaction and state two observations that could be made during the reaction. [SL paper 2, Nov 05] 6 (a) Fo ...
... 5(a) Draw a diagram to show the structure of sodium chloride. Explain, in terms of bonding, why sodium chloride has a high melting point. (b) Lithium reacts with water. Write an equation for the reaction and state two observations that could be made during the reaction. [SL paper 2, Nov 05] 6 (a) Fo ...
Worksheet 20.2
... 1- Atoms can achieve a noble gas structure by gaining, losing or sharing electrons with other atoms. 2- The rule states that, except for hydrogen , an atom combines with other atoms to form bonds in order to have 8 electrons in its valence energy level ( like noble gases). Lewis dot symbols are repr ...
... 1- Atoms can achieve a noble gas structure by gaining, losing or sharing electrons with other atoms. 2- The rule states that, except for hydrogen , an atom combines with other atoms to form bonds in order to have 8 electrons in its valence energy level ( like noble gases). Lewis dot symbols are repr ...
lecture19
... Insulating materials between capacitor plates are known as dielectrics. In the circuits we have dealt with, that material is air. It could be other insulators (glass, rubber, etc.). Dielectric materials are characterized by a dielectric constant k such that when placed between the plates of a capaci ...
... Insulating materials between capacitor plates are known as dielectrics. In the circuits we have dealt with, that material is air. It could be other insulators (glass, rubber, etc.). Dielectric materials are characterized by a dielectric constant k such that when placed between the plates of a capaci ...
Electrohydrodynamics
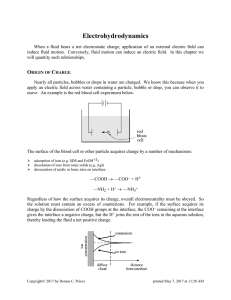
... Counterions (ions having opposite sign to the charge on the surface) would experience an electrostatic attraction for the surface which tends to pile up the ions next to the interface. On the other hand, these same ions undergo diffusion (Brownian motion) which tends to disperse them uniformly throu ...
... Counterions (ions having opposite sign to the charge on the surface) would experience an electrostatic attraction for the surface which tends to pile up the ions next to the interface. On the other hand, these same ions undergo diffusion (Brownian motion) which tends to disperse them uniformly throu ...
Electric Charge
... Every 2.0 seconds, 10 C of charge flow through each of these areas. The current in this wire is ____ A. Current is the ratio of charge to time. The quantity of charge passing through a cross section in 2 seconds is 10 C. The ratio of charge to time is I = Q / t = ( 10 C) / ( 2 s) = 5 C/s = 5 Ampere ...
... Every 2.0 seconds, 10 C of charge flow through each of these areas. The current in this wire is ____ A. Current is the ratio of charge to time. The quantity of charge passing through a cross section in 2 seconds is 10 C. The ratio of charge to time is I = Q / t = ( 10 C) / ( 2 s) = 5 C/s = 5 Ampere ...
Current-Voltage Relationship
... the ammeter is measuring a flow of charges and these charges must actually flow through the ammeter for it to be able to make the measurement. The voltmeter, on the other hand, measures the difference of potentials between two points. Hence, its ends must be brought in contact with the two points in ...
... the ammeter is measuring a flow of charges and these charges must actually flow through the ammeter for it to be able to make the measurement. The voltmeter, on the other hand, measures the difference of potentials between two points. Hence, its ends must be brought in contact with the two points in ...
Quiz 9.1, 9.2 Study Guide
... Materials through which charge can flow easily are called conductors. (Ex: most metals) Materials through which charge cannot flow easily are called insulators. (Ex: paper, cotton, rubber) The ampere is a unit of electric current. The unit of measure for resistance is called an ohm. Electric charges ...
... Materials through which charge can flow easily are called conductors. (Ex: most metals) Materials through which charge cannot flow easily are called insulators. (Ex: paper, cotton, rubber) The ampere is a unit of electric current. The unit of measure for resistance is called an ohm. Electric charges ...
E:\My Documents\sch3u\SCH3Ureview.wpd
... h) most reactive non-metal element i) most reactive metal element 12) a) Draw a Lewis dot diagram for a typical atom in the Oxygen family of elements. b) What is the typical ionic charge of these atoms. c) Explain why all the atoms in this family form stable ions with this charge. 13) The Alkali Met ...
... h) most reactive non-metal element i) most reactive metal element 12) a) Draw a Lewis dot diagram for a typical atom in the Oxygen family of elements. b) What is the typical ionic charge of these atoms. c) Explain why all the atoms in this family form stable ions with this charge. 13) The Alkali Met ...
Chapter 8
... matter can neither be created nor destroyed, but it can change forms chemical equations must show that matter was conserved ...
... matter can neither be created nor destroyed, but it can change forms chemical equations must show that matter was conserved ...
Caffeine 200 mg Tablet Structure: Molecular Formula and Mass
... standard, dissolve it in: (23.2 mg)/(0.230 mg/mL) = 101 mL of methanol. This makes the high standard solution concentration equal to 0.230 mg/mL. Low Standard: The low limit is 85%; therefore the concentration of the low standard = (0.200 mg/mL) X 0.85 = 0.170 mg/mL. Dilute 1.00 mL of high standard ...
... standard, dissolve it in: (23.2 mg)/(0.230 mg/mL) = 101 mL of methanol. This makes the high standard solution concentration equal to 0.230 mg/mL. Low Standard: The low limit is 85%; therefore the concentration of the low standard = (0.200 mg/mL) X 0.85 = 0.170 mg/mL. Dilute 1.00 mL of high standard ...
Document
... opposed to passive control which uses the momentum of free stream of fluid. The following will be addressed only plasma actuators. All actuators based on nonthermal plasma have many advantages. For example, they have no mechanical parts, they are not sources of noise and vibration, they have small v ...
... opposed to passive control which uses the momentum of free stream of fluid. The following will be addressed only plasma actuators. All actuators based on nonthermal plasma have many advantages. For example, they have no mechanical parts, they are not sources of noise and vibration, they have small v ...
Ohm`s Law Quiz Key Equations
... 9. A typical wall outlet in the United States provides a voltage of 120 V to your appliances. What current would be necessary for the electricity to pass through a 17-ohm resistor? ...
... 9. A typical wall outlet in the United States provides a voltage of 120 V to your appliances. What current would be necessary for the electricity to pass through a 17-ohm resistor? ...
Nanofluidic circuitry
Nanofluidic circuitry is a nanotechnology aiming for control of fluids in nanometer scale. Due to the effect of an electrical double layer within the fluid channel, the behavior of nanofluid is observed to be significantly different compared with its microfluidic counterparts. Its typical characteristic dimensions fall within the range of 1–100 nm. At least one dimension of the structure is in nanoscopic scale. Phenomena of fluids in nano-scale structure are discovered to be of different properties in electrochemistry and fluid dynamics.