Electrical Energy Calculations (Higher). - School
... A 12 V battery is connected to a lamp in series with a resistor and an ammeter. A voltmeter is connected across the lamp, as shown below. ...
... A 12 V battery is connected to a lamp in series with a resistor and an ammeter. A voltmeter is connected across the lamp, as shown below. ...
Chem 1A Final Exam – Fall 2005
... 2) For each of the following covalent compounds and polyatomic ions, draw a valid Lewis structure, indicating geometry around central atoms, hybridization, bond angles, and whether the species is polar or not. (20 pts) geometry Lewis structure around each each central atom chemical formula (include ...
... 2) For each of the following covalent compounds and polyatomic ions, draw a valid Lewis structure, indicating geometry around central atoms, hybridization, bond angles, and whether the species is polar or not. (20 pts) geometry Lewis structure around each each central atom chemical formula (include ...
What is the current running through each resistor in the circuit?
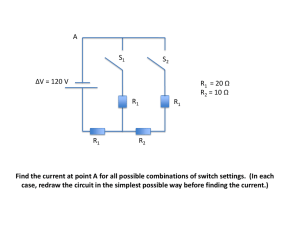
... Find the current at point A for all possible combinations of switch settings. (In each case, redraw the circuit in the simplest possible way before finding the current.) ...
... Find the current at point A for all possible combinations of switch settings. (In each case, redraw the circuit in the simplest possible way before finding the current.) ...
Nanosystems Engineering on Microfluidic Platform: Potential
... India (OLP 101512). 2. “Design and Development of Gold-Iron Oxide Based Smart Magnetic Nanosensor for Detection and Separation of Heavy Metal Ions” funded by Department of Biotechnology, India (GAP 101612). 3. Nano patterning of metallic and polymeric thin films funded by CSIR India (ESC0112) ...
... India (OLP 101512). 2. “Design and Development of Gold-Iron Oxide Based Smart Magnetic Nanosensor for Detection and Separation of Heavy Metal Ions” funded by Department of Biotechnology, India (GAP 101612). 3. Nano patterning of metallic and polymeric thin films funded by CSIR India (ESC0112) ...
Chem Bonding Notes
... (1)H2O (3)H2Se (2)H2S (4)H2Te 8. Which compound contains ionic bonds? (1) NO (3) CaO (2) NO 2 (4) CO 2 9. Metallic bonding occurs between atoms of (1) sulfur (3) fluorine (2) copper (4) carbon 10. Covalent bonds are formed when electrons are (1) transferred from one atom to another (2) captured by t ...
... (1)H2O (3)H2Se (2)H2S (4)H2Te 8. Which compound contains ionic bonds? (1) NO (3) CaO (2) NO 2 (4) CO 2 9. Metallic bonding occurs between atoms of (1) sulfur (3) fluorine (2) copper (4) carbon 10. Covalent bonds are formed when electrons are (1) transferred from one atom to another (2) captured by t ...
Department of Natural Sciences
... A rubber object receives a negative charge by rubbing it with a fur. In the rubbing process, have electrons been added to the object or have protons been removed from it? A. Electrons have been added. ...
... A rubber object receives a negative charge by rubbing it with a fur. In the rubbing process, have electrons been added to the object or have protons been removed from it? A. Electrons have been added. ...
Review of Definitions
... 13.Ionization. Any process by which an electrically neutral atom or molecule is converted into an electrically charged atom or molecule 14. Dielectric material. An insulating material or a very poor conductor of electric current. When dielectrics are placed in an electric field, practically no curre ...
... 13.Ionization. Any process by which an electrically neutral atom or molecule is converted into an electrically charged atom or molecule 14. Dielectric material. An insulating material or a very poor conductor of electric current. When dielectrics are placed in an electric field, practically no curre ...
Review of Definitions
... another atom in a chemical bond. The electronegativity of an atom depends on two factors : (1) the number of positive charges in the nucleus (the more protons, the more electronegative) and (2) the distance of the outer electrons from the nucleus (the greater the distance, the less electronegative) ...
... another atom in a chemical bond. The electronegativity of an atom depends on two factors : (1) the number of positive charges in the nucleus (the more protons, the more electronegative) and (2) the distance of the outer electrons from the nucleus (the greater the distance, the less electronegative) ...
Science 9 electricity powerpoint Topic 1
... Objects are positive when they have more protons than ...
... Objects are positive when they have more protons than ...
22A The effect of current on cell potential
... and the degree of polarization is given by an overvoltage, or overpotential, which is symbolized by . Eapplied = Ecell – IR – Factors that influence polarization are (1) electrode size, shape, and composition; (2) composition of the electrolyte solution; (3) temperature and stirring rate; (4) cur ...
... and the degree of polarization is given by an overvoltage, or overpotential, which is symbolized by . Eapplied = Ecell – IR – Factors that influence polarization are (1) electrode size, shape, and composition; (2) composition of the electrolyte solution; (3) temperature and stirring rate; (4) cur ...
Nanofluidic circuitry
Nanofluidic circuitry is a nanotechnology aiming for control of fluids in nanometer scale. Due to the effect of an electrical double layer within the fluid channel, the behavior of nanofluid is observed to be significantly different compared with its microfluidic counterparts. Its typical characteristic dimensions fall within the range of 1–100 nm. At least one dimension of the structure is in nanoscopic scale. Phenomena of fluids in nano-scale structure are discovered to be of different properties in electrochemistry and fluid dynamics.