Answers to Problem-Solving Practice Problems
... (4) Check your answer: Because the density is a little less than 1.00 g/mL, the volume in milliliters should be a little larger than the mass in grams. The calculated answer, 4.92 mL, is a little larger than the mass, 4.33 g. 1.2 Substance A must be a mixture since some of it dissolves and some, sub ...
... (4) Check your answer: Because the density is a little less than 1.00 g/mL, the volume in milliliters should be a little larger than the mass in grams. The calculated answer, 4.92 mL, is a little larger than the mass, 4.33 g. 1.2 Substance A must be a mixture since some of it dissolves and some, sub ...
Chapter 3 Solutions - Bremerton School District
... There are three peaks in the mass spectrum, each 2 mass units apart. This is consistent with two isotopes, differing in mass by two mass units. The peak at 157.84 corresponds to a Br2 molecule composed of two atoms of the lighter isotope. This isotope has mass equal to 157.84/2 or 78.92. This corres ...
... There are three peaks in the mass spectrum, each 2 mass units apart. This is consistent with two isotopes, differing in mass by two mass units. The peak at 157.84 corresponds to a Br2 molecule composed of two atoms of the lighter isotope. This isotope has mass equal to 157.84/2 or 78.92. This corres ...
Low Temperature Precursors for SnOx Thin Films
... of tin and materials chemistry and Dr. Andrew Johnson providing the opportunity for numerous discussions and also sorting out the crystals that were investigated by X-ray diffraction - including all of the duds. Thanks go to the both EPSRC for funding me through the DTC in sustainable chemical techn ...
... of tin and materials chemistry and Dr. Andrew Johnson providing the opportunity for numerous discussions and also sorting out the crystals that were investigated by X-ray diffraction - including all of the duds. Thanks go to the both EPSRC for funding me through the DTC in sustainable chemical techn ...
b - Gordon State College
... 2) Find the moles of each reactant: moles = mass in gram / molar mass 3) Pick up any reactant, say A, and use the stoichiometry to calculate the required amount of the other reactant B. 4) Compare the required amount of B with the available amount of B. a) If required > available, then B is the limi ...
... 2) Find the moles of each reactant: moles = mass in gram / molar mass 3) Pick up any reactant, say A, and use the stoichiometry to calculate the required amount of the other reactant B. 4) Compare the required amount of B with the available amount of B. a) If required > available, then B is the limi ...
Composition and Evolution of Interstellar Clouds
... Widespread warm H I gas (WNM at 6,000–10,000 K) exists in the space between cold H I diffuse clouds (CNM at 50– 100 K), as observed by H I 21-cm emission and absorption lines, respectively (cf. Ferrière, 2001). The density ratio between the WNM and diffuse clouds is approximately the inverse of the ...
... Widespread warm H I gas (WNM at 6,000–10,000 K) exists in the space between cold H I diffuse clouds (CNM at 50– 100 K), as observed by H I 21-cm emission and absorption lines, respectively (cf. Ferrière, 2001). The density ratio between the WNM and diffuse clouds is approximately the inverse of the ...
CHAPTER 3 STOICHIOMETRY
... Strategy: We can look up the molar mass of arsenic (As) on the periodic table (74.92 g/mol). We want to find the mass of a single atom of arsenic (unit of g/atom). Therefore, we need to convert from the unit mole in the denominator to the unit atom in the denominator. What conversion factor is neede ...
... Strategy: We can look up the molar mass of arsenic (As) on the periodic table (74.92 g/mol). We want to find the mass of a single atom of arsenic (unit of g/atom). Therefore, we need to convert from the unit mole in the denominator to the unit atom in the denominator. What conversion factor is neede ...
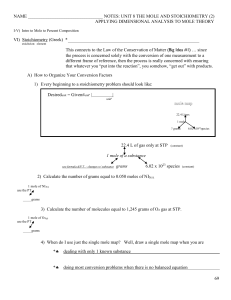
NAME NOTES: UNIT 8 THE MOLE AND STOICHIOMETRY (2
... There are many types of homogeneous solutions. The "aqueous solution" is just one type. It is however, the most important to the understanding of the material contained in this Chemistry syllabus. ...
... There are many types of homogeneous solutions. The "aqueous solution" is just one type. It is however, the most important to the understanding of the material contained in this Chemistry syllabus. ...
Topic 1 Quantitative Chemistry Answers - slider-dpchemistry-11
... A substance that cannot be divided into simpler, smaller substances. In an element, all the atoms have the same number of protons or electrons, but the number of neutrons may vary (more about this Topic 2) b) atom The smallest part of an element that can exist. An atom consists of an extremely tiny ...
... A substance that cannot be divided into simpler, smaller substances. In an element, all the atoms have the same number of protons or electrons, but the number of neutrons may vary (more about this Topic 2) b) atom The smallest part of an element that can exist. An atom consists of an extremely tiny ...
2nd Semester Practice Chemistry Final 2009
... a. less effective in solids than in liquids. c. equally effective in gases and in liquids. b. more effective in gases than in solids. d. more effective in liquids than in gases. 44. Which term best describes the process by which particles escape from the surface of a nonboiling liquid and enter the ...
... a. less effective in solids than in liquids. c. equally effective in gases and in liquids. b. more effective in gases than in solids. d. more effective in liquids than in gases. 44. Which term best describes the process by which particles escape from the surface of a nonboiling liquid and enter the ...
Week 3 July 22, 2016 Worksheet Review III 1 mol = 6.022 × 1023 1
... Another way to think about this problem (without actually doing any calculations) is to think about the conversion. In all cases we start with 10.0 g of an element, and then we divide by the mass of each element (blue numbers), and then multiply by Avogadro’s number. So the only difference between e ...
... Another way to think about this problem (without actually doing any calculations) is to think about the conversion. In all cases we start with 10.0 g of an element, and then we divide by the mass of each element (blue numbers), and then multiply by Avogadro’s number. So the only difference between e ...
Stoichiometric Calculations
... A series of steps can be used to determine the limiting reactant in any reaction: Step 1: Convert the given quantities into moles. These are your initial amounts of each reactant. Step 2: Divide each by its stoichiometrical coefficient from the balanced chemical equation. This factors in how much is ...
... A series of steps can be used to determine the limiting reactant in any reaction: Step 1: Convert the given quantities into moles. These are your initial amounts of each reactant. Step 2: Divide each by its stoichiometrical coefficient from the balanced chemical equation. This factors in how much is ...
Stoichiometric Calculations
... A series of steps can be used to determine the limiting reactant in any reaction: Step 1: Convert the given quantities into moles. These are your initial amounts of each reactant. Step 2: Divide each by its stoichiometrical coefficient from the balanced chemical equation. This factors in how much is ...
... A series of steps can be used to determine the limiting reactant in any reaction: Step 1: Convert the given quantities into moles. These are your initial amounts of each reactant. Step 2: Divide each by its stoichiometrical coefficient from the balanced chemical equation. This factors in how much is ...
synthesis and properties of v3+ analogues of jarosite-group
... of the lower solubility of Tl2SO4, a solution containing 0.23 M Tl2SO4 was used, and Li2SO4 was added to give an overall sulfate concentration of 0.4 M SO4. The resulting precipitate, however, had an excessive Tl content, and X-ray powder-diffraction analysis indicated the presence of TlCl. Reducing ...
... of the lower solubility of Tl2SO4, a solution containing 0.23 M Tl2SO4 was used, and Li2SO4 was added to give an overall sulfate concentration of 0.4 M SO4. The resulting precipitate, however, had an excessive Tl content, and X-ray powder-diffraction analysis indicated the presence of TlCl. Reducing ...
Chemistry Appendixes
... Scientists ask questions and seek concepts to answer these questions by applying consistent, logical reasoning to describe, explain, and predict observations, and by doing experiments to test hypotheses or predictions from these hypotheses. In this way science progresses using a general model for so ...
... Scientists ask questions and seek concepts to answer these questions by applying consistent, logical reasoning to describe, explain, and predict observations, and by doing experiments to test hypotheses or predictions from these hypotheses. In this way science progresses using a general model for so ...
File
... Alkaline earth metals have ns2 for valence electron configurations. They are all very reactive, losing their two valence electrons to nonmetals to form ionic compounds containing M 2+ cations. Alkaline earth metals, like alkali metals, are easily oxidized. Their preparation as pure metals must be do ...
... Alkaline earth metals have ns2 for valence electron configurations. They are all very reactive, losing their two valence electrons to nonmetals to form ionic compounds containing M 2+ cations. Alkaline earth metals, like alkali metals, are easily oxidized. Their preparation as pure metals must be do ...
GCSE Chemistry Sample Assessment Materials
... Comprehensive description of the formation of acid rain, including the presence of sulfur impurities in fossil fuels; sulfur combustion equation; at least three effects on the environment, including one effect on a material and one effect on a living organism There is a sustained line of reasoning w ...
... Comprehensive description of the formation of acid rain, including the presence of sulfur impurities in fossil fuels; sulfur combustion equation; at least three effects on the environment, including one effect on a material and one effect on a living organism There is a sustained line of reasoning w ...
Gas chromatography–mass spectrometry

Gas chromatography–mass spectrometry (GC-MS) is an analytical method that combines the features of gas-chromatography and mass spectrometry to identify different substances within a test sample. Applications of GC-MS include drug detection, fire investigation, environmental analysis, explosives investigation, and identification of unknown samples. GC-MS can also be used in airport security to detect substances in luggage or on human beings. Additionally, it can identify trace elements in materials that were previously thought to have disintegrated beyond identification.GC-MS has been widely heralded as a ""gold standard"" for forensic substance identification because it is used to perform a specific test. A specific test positively identifies the actual presence of a particular substance in a given sample. A non-specific test merely indicates that a substance falls into a category of substances. Although a non-specific test could statistically suggest the identity of the substance, this could lead to false positive identification.