Mole-mole factor
... – If special conditions such as heat are required – The number of molecules, formula units, or atoms of each type of molecule involved in the reaction • Number can be in terms of single atoms, or moles of atoms – The relative number of moles of each reactant and product ...
... – If special conditions such as heat are required – The number of molecules, formula units, or atoms of each type of molecule involved in the reaction • Number can be in terms of single atoms, or moles of atoms – The relative number of moles of each reactant and product ...
Chapter 4: Types of Chemical Reactions and Solution Stoichiometry
... Example: If a solution containing potassium chloride is added to a solution containing ammonium nitrate, will a precipitate form? KCl(aq) + NH4NO3(aq) → K+(aq) + Cl-(aq) + NH4+(aq) + NO3-(aq) Possible reaction products are KCl and NH4NO3, NH4Cl and KNO3. All are soluble, so there is no precipitate. ...
... Example: If a solution containing potassium chloride is added to a solution containing ammonium nitrate, will a precipitate form? KCl(aq) + NH4NO3(aq) → K+(aq) + Cl-(aq) + NH4+(aq) + NO3-(aq) Possible reaction products are KCl and NH4NO3, NH4Cl and KNO3. All are soluble, so there is no precipitate. ...
Removal of hydrogen fluoride from gas streams
... I would like to thank my supervisors, Professor N. J. Coville, University of Witwatersrand and Dr. P. A. B. Carstens, South African Nuclear Energy Corporation Limited (Necsa) for their support, suggestions and guidance. ...
... I would like to thank my supervisors, Professor N. J. Coville, University of Witwatersrand and Dr. P. A. B. Carstens, South African Nuclear Energy Corporation Limited (Necsa) for their support, suggestions and guidance. ...
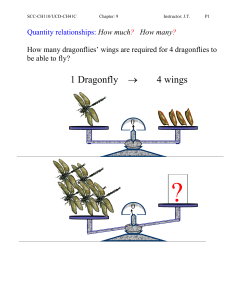
Quantity relationships: How much
... The calculated amount of product if is based on the assumption that all of the reactant is converted into product is called the theoretical yield. In laboratory or in industrial production, the actual amount of product isolated from a reaction is usually less than the theoretical yield, and it is ca ...
... The calculated amount of product if is based on the assumption that all of the reactant is converted into product is called the theoretical yield. In laboratory or in industrial production, the actual amount of product isolated from a reaction is usually less than the theoretical yield, and it is ca ...
Chemistry Science Notebook: Student Edition
... lesson. This column will help students find information and locate the references in their textbooks quickly. Students can also use this column to sketch drawings that help them visually remember the lesson’s information. In the column on the right side of the page, students will write detailed note ...
... lesson. This column will help students find information and locate the references in their textbooks quickly. Students can also use this column to sketch drawings that help them visually remember the lesson’s information. In the column on the right side of the page, students will write detailed note ...
Under Choice Based Credit System Proposed syllabus and Scheme of Examination
... 2. Estimation of oxalic acid by titrating it with KMnO4. 3. Estimation of water of crystallization in Mohr’s salt by titrating with KMnO4. 4. Estimation of Fe (II) ions by titrating it with K2Cr2O7 using internal indicator. 5. Estimation of Cu (II) ions iodometrically using Na2S2O3. Section B: Organ ...
... 2. Estimation of oxalic acid by titrating it with KMnO4. 3. Estimation of water of crystallization in Mohr’s salt by titrating with KMnO4. 4. Estimation of Fe (II) ions by titrating it with K2Cr2O7 using internal indicator. 5. Estimation of Cu (II) ions iodometrically using Na2S2O3. Section B: Organ ...
Cliffs Notes
... Copyright © 2001 Gary S. Thorpe. All rights reserved. No part of this book, including interior design, cover design, and icons, may be reproduced or transmitted in any form, by any means (electronic, photocopying, recording, or otherwise) without the prior written permission of the publisher. Librar ...
... Copyright © 2001 Gary S. Thorpe. All rights reserved. No part of this book, including interior design, cover design, and icons, may be reproduced or transmitted in any form, by any means (electronic, photocopying, recording, or otherwise) without the prior written permission of the publisher. Librar ...
Stoichiometric Calculations
... Remember: Do not use coefficients when converting between moles and mass. The molar mass is for only ONE mole! ...
... Remember: Do not use coefficients when converting between moles and mass. The molar mass is for only ONE mole! ...
Stoichiometry
... molecular, or formula mass of the substance – For elements, M = atomic mass (from per. table) – For molecular compounds and molecular elements, M = molecular mass – For ionic compounds, M = formula mass ⇒ For compounds and molecular elements, M equals the sum of the molar (atomic) masses of the elem ...
... molecular, or formula mass of the substance – For elements, M = atomic mass (from per. table) – For molecular compounds and molecular elements, M = molecular mass – For ionic compounds, M = formula mass ⇒ For compounds and molecular elements, M equals the sum of the molar (atomic) masses of the elem ...
STOICHIOMETRY via ChemLog - Small
... When carrying out a chemical reaction, we may use the exact amount of each reactant needed. Or, we may use an excess of some reactants and a limited amount of others. We may do this if one reactant is very expensive and others are inexpensive so that we can use all of the expensive compound. It can ...
... When carrying out a chemical reaction, we may use the exact amount of each reactant needed. Or, we may use an excess of some reactants and a limited amount of others. We may do this if one reactant is very expensive and others are inexpensive so that we can use all of the expensive compound. It can ...
PC_Chemistry_Macomb_April08
... Nuclear stability is related to a decrease in potential energy when the nucleus forms from protons and neutrons. If the neutron/proton ratio is unstable, the element will undergo radioactive decay. The rate of decay is characteristic of each isotope; the time for half the parent nuclei to decay is c ...
... Nuclear stability is related to a decrease in potential energy when the nucleus forms from protons and neutrons. If the neutron/proton ratio is unstable, the element will undergo radioactive decay. The rate of decay is characteristic of each isotope; the time for half the parent nuclei to decay is c ...
Quantitative chemistry notes
... The mole is defined as the amount of substance that contains as many elementary particles as there are atoms in 12g of carbon-12 The average atomic mass (AR) of an atom of carbon-12 is 1.99252 x 10-23 g. So the number of atoms in 12g of carbon-12 is given by: 12 g / 1.999252 x 10-23 g = 6.02 x 1023 ...
... The mole is defined as the amount of substance that contains as many elementary particles as there are atoms in 12g of carbon-12 The average atomic mass (AR) of an atom of carbon-12 is 1.99252 x 10-23 g. So the number of atoms in 12g of carbon-12 is given by: 12 g / 1.999252 x 10-23 g = 6.02 x 1023 ...
Chemistry In action
... R. R. Donnelley/Jefferson City. The cover was printed by R. R. Donnelley/Jefferson City. The paper in this book was manufactured by a mill whose forest management programs include sustained yield—harvesting of its timberlands. Sustained yield harvesting principles ensure that the number of trees cut ...
... R. R. Donnelley/Jefferson City. The cover was printed by R. R. Donnelley/Jefferson City. The paper in this book was manufactured by a mill whose forest management programs include sustained yield—harvesting of its timberlands. Sustained yield harvesting principles ensure that the number of trees cut ...
Document
... Suppose Si (having 4 valence electrons is doped with Ga (which has 3 valence electrons), 3 valence electrons are involved in bond formation with neighboring Si atom. A vacancy is left which can be filled by the transfer of a valence electron from a neighboring Si atom. The movement of electron into ...
... Suppose Si (having 4 valence electrons is doped with Ga (which has 3 valence electrons), 3 valence electrons are involved in bond formation with neighboring Si atom. A vacancy is left which can be filled by the transfer of a valence electron from a neighboring Si atom. The movement of electron into ...
General and Inorganic Chemistry – Laboratory Techniques
... When the atomic theory developed to the point where it was possible to write specific formulae for the various oxides and their binary compounds, names reflecting composition more or less accurately then became common. As a number of inorganic compounds rapidly grew, the essential pattern of nomencl ...
... When the atomic theory developed to the point where it was possible to write specific formulae for the various oxides and their binary compounds, names reflecting composition more or less accurately then became common. As a number of inorganic compounds rapidly grew, the essential pattern of nomencl ...
CHAPTER 5 GASES
... in a dish of mercury so that no air enters the tube. Some of the mercury in the tube flows into the dish, creating a vacuum at the top, closed end of the tube. The weight of the mercury remaining in the tube is supported by atmospheric pressure. See Figure 5.2 of the text. A manometer works in a sim ...
... in a dish of mercury so that no air enters the tube. Some of the mercury in the tube flows into the dish, creating a vacuum at the top, closed end of the tube. The weight of the mercury remaining in the tube is supported by atmospheric pressure. See Figure 5.2 of the text. A manometer works in a sim ...
Devillez (ld2653) – Test 1 Review – Devillez – (99998)
... In Rutherford’s gold foil experiment in 1910, α (alpha) particles were fired at gold foil, and the resulting deflection of the particles were observed. Most of the α particles went through the sample undeflected, suggesting that much of the atom was empty space. But of the few α particles that were ...
... In Rutherford’s gold foil experiment in 1910, α (alpha) particles were fired at gold foil, and the resulting deflection of the particles were observed. Most of the α particles went through the sample undeflected, suggesting that much of the atom was empty space. But of the few α particles that were ...
Gas chromatography–mass spectrometry

Gas chromatography–mass spectrometry (GC-MS) is an analytical method that combines the features of gas-chromatography and mass spectrometry to identify different substances within a test sample. Applications of GC-MS include drug detection, fire investigation, environmental analysis, explosives investigation, and identification of unknown samples. GC-MS can also be used in airport security to detect substances in luggage or on human beings. Additionally, it can identify trace elements in materials that were previously thought to have disintegrated beyond identification.GC-MS has been widely heralded as a ""gold standard"" for forensic substance identification because it is used to perform a specific test. A specific test positively identifies the actual presence of a particular substance in a given sample. A non-specific test merely indicates that a substance falls into a category of substances. Although a non-specific test could statistically suggest the identity of the substance, this could lead to false positive identification.