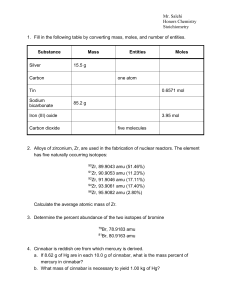
Stoichiometry - Cloudfront.net
... only carbon, hydrogen, and oxygen, determine the empirical formula of the unknown compound. 7. A 105.5 mg sample of a white substance is suspected to be cocaine, C 17H21NO4. The substance formed 279.3 mg of CO2 and 66.46 mg H2O on combustion. The compound contains 4.680% N by mass. Is the white soli ...
... only carbon, hydrogen, and oxygen, determine the empirical formula of the unknown compound. 7. A 105.5 mg sample of a white substance is suspected to be cocaine, C 17H21NO4. The substance formed 279.3 mg of CO2 and 66.46 mg H2O on combustion. The compound contains 4.680% N by mass. Is the white soli ...
Review Packet
... b. are used to make other elements c. are used to make compounds d. are never found in the periodic table of elements 30. Physical means can be used to separate a. elements b. pure substances b. mixtures d. compounds ...
... b. are used to make other elements c. are used to make compounds d. are never found in the periodic table of elements 30. Physical means can be used to separate a. elements b. pure substances b. mixtures d. compounds ...
Chemistry Semester Test Study Guide Chapters
... Be able to use the rules for sig figs for division and subtraction as well. ...
... Be able to use the rules for sig figs for division and subtraction as well. ...
Practice questions
... K, Ag, Po, Cl, S b) P, Po, Ag, Cl, S c) Ph, K, Ag, S, Cl d) P, K, Ag, Cl, S e) Ph, Po, Ag, Cl, S ...
... K, Ag, Po, Cl, S b) P, Po, Ag, Cl, S c) Ph, K, Ag, S, Cl d) P, K, Ag, Cl, S e) Ph, Po, Ag, Cl, S ...
Chapter 8: Chemical Reactions and Physical Changes
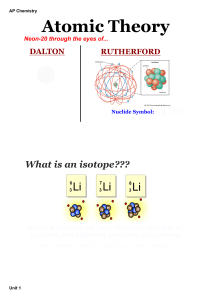
... • Mass number: total protons and neutrons in an atom’s nucleus • Atomic mass: the average mass of a sample of atoms of that element found in nature • Periodic table: chart that arranges elements by atomic number into rows and columns according to similarities in their properties ...
... • Mass number: total protons and neutrons in an atom’s nucleus • Atomic mass: the average mass of a sample of atoms of that element found in nature • Periodic table: chart that arranges elements by atomic number into rows and columns according to similarities in their properties ...
CHM 50 Exam 1 Review Name Due Tuesday 9/29/09 Exam 1 will
... a. Sodium metal plus water yields hydrogen gas and an aqueous sodium hydroxide solution. b. Potassium chlorate when heated yields potassium chlorate plus oxygen gas. c. An aqueous phosphoric acid solution plus an aqueous calcium hydroxide solution yields water and solid calcium phosphate. 5. A carta ...
... a. Sodium metal plus water yields hydrogen gas and an aqueous sodium hydroxide solution. b. Potassium chlorate when heated yields potassium chlorate plus oxygen gas. c. An aqueous phosphoric acid solution plus an aqueous calcium hydroxide solution yields water and solid calcium phosphate. 5. A carta ...
formula mass.
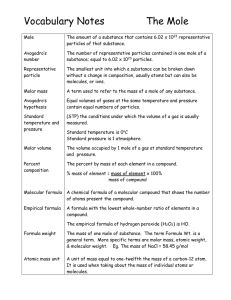
... The Avogadro number was named in honor of Amedeo Avogadro who discovered that a mole of any gas under the same conditions has the same number of molecules. Johann Josef Loschmidt, a German physicist, named and discovered the Avogadro number. Loschmidt realized that a mole of any substance—be i ...
... The Avogadro number was named in honor of Amedeo Avogadro who discovered that a mole of any gas under the same conditions has the same number of molecules. Johann Josef Loschmidt, a German physicist, named and discovered the Avogadro number. Loschmidt realized that a mole of any substance—be i ...
PowerPoint Template
... the total mass of substances does not change during a chemical reaction - Antoine Lavoisier (1 743-1 794) The number of substances may change, but the total amount of matter remains constant. ...
... the total mass of substances does not change during a chemical reaction - Antoine Lavoisier (1 743-1 794) The number of substances may change, but the total amount of matter remains constant. ...
Review Packet
... b. are used to make other elements c. are used to make compounds d. are never found in the periodic table of elements 30. Physical means can be used to separate a. elements b. pure substances b. mixtures d. compounds ...
... b. are used to make other elements c. are used to make compounds d. are never found in the periodic table of elements 30. Physical means can be used to separate a. elements b. pure substances b. mixtures d. compounds ...
Packet
... b. are used to make other elements c. are used to make compounds d. are never found in the periodic table of elements 30. Physical means can be used to separate a. elements b. pure substances b. mixtures d. compounds 31. Anything that takes up space and has mass is called a. matter b. mass c. volume ...
... b. are used to make other elements c. are used to make compounds d. are never found in the periodic table of elements 30. Physical means can be used to separate a. elements b. pure substances b. mixtures d. compounds 31. Anything that takes up space and has mass is called a. matter b. mass c. volume ...
Answers pg 233 #4,5
... products is 85 g, what was the mass of the solution? solid + solution = products 35g + ?? = 85g ?? = 85 g 35g ?? = 50g ...
... products is 85 g, what was the mass of the solution? solid + solution = products 35g + ?? = 85g ?? = 85 g 35g ?? = 50g ...
Vocabulary Notes
... without a change in composition, usually atoms but can also be molecules, or ions. ...
... without a change in composition, usually atoms but can also be molecules, or ions. ...
heats of reaction
... 2.) Write balanced heat of formation reactions for each of the following. Do not forget to include the ∆Hf values from the table for the phases listed. Use your appendix (∆H values) to write balanced formation equations determine ...
... 2.) Write balanced heat of formation reactions for each of the following. Do not forget to include the ∆Hf values from the table for the phases listed. Use your appendix (∆H values) to write balanced formation equations determine ...
Periodic Table Puzzle
... The relative atomic mass of C is greater than the relative atomic mass of N but less than that of E. ...
... The relative atomic mass of C is greater than the relative atomic mass of N but less than that of E. ...
Gas chromatography–mass spectrometry

Gas chromatography–mass spectrometry (GC-MS) is an analytical method that combines the features of gas-chromatography and mass spectrometry to identify different substances within a test sample. Applications of GC-MS include drug detection, fire investigation, environmental analysis, explosives investigation, and identification of unknown samples. GC-MS can also be used in airport security to detect substances in luggage or on human beings. Additionally, it can identify trace elements in materials that were previously thought to have disintegrated beyond identification.GC-MS has been widely heralded as a ""gold standard"" for forensic substance identification because it is used to perform a specific test. A specific test positively identifies the actual presence of a particular substance in a given sample. A non-specific test merely indicates that a substance falls into a category of substances. Although a non-specific test could statistically suggest the identity of the substance, this could lead to false positive identification.