Velicky17.10.BECBrno07L3
... • the mean field component of the interactions determines most of the deviations from the non-interacting case • beyond the mean field, the interactions change the quasi-particles and result into superfluidity even in these dilute systems ...
... • the mean field component of the interactions determines most of the deviations from the non-interacting case • beyond the mean field, the interactions change the quasi-particles and result into superfluidity even in these dilute systems ...
here
... Emergent quantum phases in correlated spin-orbit coupled materials. Abbreviations are as follows: TME = topological magnetoelectric effect, (F)QHE = (fractional) quantum Hall effect. Correlations are W-I = weak-intermediate, I = intermediate (requiring magnetic order, say, but mean field-like), and ...
... Emergent quantum phases in correlated spin-orbit coupled materials. Abbreviations are as follows: TME = topological magnetoelectric effect, (F)QHE = (fractional) quantum Hall effect. Correlations are W-I = weak-intermediate, I = intermediate (requiring magnetic order, say, but mean field-like), and ...
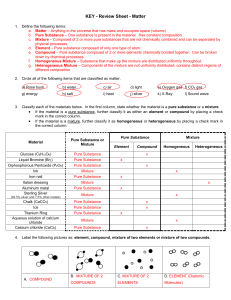
Matter Test Review Sheet
... a. Matter – Anything in the universe that has mass and occupies space (volume) b. Pure Substance – One substance is present in the material. Has constant composition. c. Mixture – Composed of 2 or more pure substances that are not chemically combined and can be separated by physical processes. d. El ...
... a. Matter – Anything in the universe that has mass and occupies space (volume) b. Pure Substance – One substance is present in the material. Has constant composition. c. Mixture – Composed of 2 or more pure substances that are not chemically combined and can be separated by physical processes. d. El ...
"Pentaquarks: Theory & Experiment"(ppt 1.59M)
... For baryons with spin ½ and + parity, there is no N around, and a weak evidence for S(1770), so Diakonov and Petro (hep-ph/0310212) suggeted a missing N around 1650-1690 MeV; the position of N(1710) is not at M=1710 MeV, but some where around M=1650 to 1690 MeV, suggested by Arndt et al(nucl-th/0312 ...
... For baryons with spin ½ and + parity, there is no N around, and a weak evidence for S(1770), so Diakonov and Petro (hep-ph/0310212) suggeted a missing N around 1650-1690 MeV; the position of N(1710) is not at M=1710 MeV, but some where around M=1650 to 1690 MeV, suggested by Arndt et al(nucl-th/0312 ...
Impact and Momentum - definition and units
... Often impulse is found indirectly by considering the change in momentum of a body, which is especially useful when the force and time are unknown, but both the momentum before and after can be found, as for example in a collision. Worked Example 2. A child kicks a stationary football, of mass 0.5 kg ...
... Often impulse is found indirectly by considering the change in momentum of a body, which is especially useful when the force and time are unknown, but both the momentum before and after can be found, as for example in a collision. Worked Example 2. A child kicks a stationary football, of mass 0.5 kg ...
Wolfgang Ketterle Department of Physics Superfluidity and Coherence in Bose-Einstein Condensates
... 6. Transport of Bose-Einstein Condensates with Optical Tweezers Conventional condensate production techniques severely limit optical and mechanical access to experiments due to the many laser beams and magnetic coils needed to create BECs. This conflict between cooling infrastructure and accessibili ...
... 6. Transport of Bose-Einstein Condensates with Optical Tweezers Conventional condensate production techniques severely limit optical and mechanical access to experiments due to the many laser beams and magnetic coils needed to create BECs. This conflict between cooling infrastructure and accessibili ...
New Title
... a. H2 + O2 H2O b. Mg + O2 MgO c. Na + O2 Na2O d. 2 H2O2 2 H2O + O2 17. A number placed in front of a chemical formula in a chemical equation is called a(n) 18. What does a coefficient tell you? ...
... a. H2 + O2 H2O b. Mg + O2 MgO c. Na + O2 Na2O d. 2 H2O2 2 H2O + O2 17. A number placed in front of a chemical formula in a chemical equation is called a(n) 18. What does a coefficient tell you? ...
Diamagnetism and de Haas-van Alphen oscillations in the electronic
... Here we outline the structure of this article. A general introduction is given, with a short summary of the historical development of the theory and the experimental advancements related to the de Haas-van Alphen (dHvA) effect (§1.2). We are interested in deriving magnetic quantities, as susceptibil ...
... Here we outline the structure of this article. A general introduction is given, with a short summary of the historical development of the theory and the experimental advancements related to the de Haas-van Alphen (dHvA) effect (§1.2). We are interested in deriving magnetic quantities, as susceptibil ...
Fysiikan seminaarit -haku Oulun yliopisto | Fysiikan seminaarit
... indicate that some of the complex physics of galaxy formation are not yet fully understood? 17.02.2011 MSc. Timo Virtanen (University of Oulu) Quasiparticle dynamics in Fermi liquids Abstract: Fermi liquid theory is a fundamental theory of interacting many body systems, a standard against which othe ...
... indicate that some of the complex physics of galaxy formation are not yet fully understood? 17.02.2011 MSc. Timo Virtanen (University of Oulu) Quasiparticle dynamics in Fermi liquids Abstract: Fermi liquid theory is a fundamental theory of interacting many body systems, a standard against which othe ...
File
... The colour before as well as after the test must be given in the answer. For example, bromine water changes from brown to colourless, not ‘is decolorised’, and acidified potassium dichromate(vi) goes from orange to green, not ‘goes green’. Gas evolved is not an observation (it is a deduction). Bubbl ...
... The colour before as well as after the test must be given in the answer. For example, bromine water changes from brown to colourless, not ‘is decolorised’, and acidified potassium dichromate(vi) goes from orange to green, not ‘goes green’. Gas evolved is not an observation (it is a deduction). Bubbl ...
pdfCfE Higher - Unit 3 - Pupil Booklet 2 MB
... zero but the aim is to learn from mistakes and reduce the rate to a minimum. It is essential at this stage to revise all of your National 5 calculations that were based on moles and equations. ...
... zero but the aim is to learn from mistakes and reduce the rate to a minimum. It is essential at this stage to revise all of your National 5 calculations that were based on moles and equations. ...
1.6 Energy changes in chemical reactions
... Chemists deal with matter on a macroscopic scale in the laboratory, but explain its behaviour in terms of atoms and molecules. This requires a wide range of distances (see Figure 1.4). You will need to become familiar with the multiplication prefixes in Table 1.3 used to describe lengths on atomic a ...
... Chemists deal with matter on a macroscopic scale in the laboratory, but explain its behaviour in terms of atoms and molecules. This requires a wide range of distances (see Figure 1.4). You will need to become familiar with the multiplication prefixes in Table 1.3 used to describe lengths on atomic a ...
Testing Wavefunction Collapse
... clearly consistent way - the eventual diffusion of the packet, or the possibility of splitting it into disjoint parts, means the ‘particle’ does not remain localized, for instance. The other option is that the projection (2) does not take place. Rather, the correct wavefunction remains (1) so that a ...
... clearly consistent way - the eventual diffusion of the packet, or the possibility of splitting it into disjoint parts, means the ‘particle’ does not remain localized, for instance. The other option is that the projection (2) does not take place. Rather, the correct wavefunction remains (1) so that a ...
1 Introduction and Review of Electronic Technology - Wiley-VCH
... have only incrementally changed since the introduction of the automatic transmission and air conditioning, and a similar outcome is not unreasonable for electronics. It is unlikely that there will be a change away from silicon electronics as dramatic and complete as the recent tipping point in which ...
... have only incrementally changed since the introduction of the automatic transmission and air conditioning, and a similar outcome is not unreasonable for electronics. It is unlikely that there will be a change away from silicon electronics as dramatic and complete as the recent tipping point in which ...
Audit Schedule
... 1. It is imperative that you learn how to convert moles to grams and grams to moles. This simple conversion is a basic step in a great many problems that will be encountered throughout chemistry. 2. Remember that a single chemical symbol can have several different meanings. For instance, Fe can mean ...
... 1. It is imperative that you learn how to convert moles to grams and grams to moles. This simple conversion is a basic step in a great many problems that will be encountered throughout chemistry. 2. Remember that a single chemical symbol can have several different meanings. For instance, Fe can mean ...
Macroscopic electric field and osmotic pressure in ultracentrifugal
... dispersion which form a system of three interpenetrating fluids, or a mixture of three species, namely colloidal particles with number density ρ each carrying z elementary charges, monovalent cations with concentration c+, and monovalent anions with concentration c-. The dispersion is considered in ...
... dispersion which form a system of three interpenetrating fluids, or a mixture of three species, namely colloidal particles with number density ρ each carrying z elementary charges, monovalent cations with concentration c+, and monovalent anions with concentration c-. The dispersion is considered in ...
Are the Past and the Future Really Out There?
... advanced was that inertial forces should arise as a consequence of their interaction with the material contents of the universe via the action of a field. This should not be confused with the claim that the fermions acquire their masses as a result of interactions with Higgs fields, for the field in ...
... advanced was that inertial forces should arise as a consequence of their interaction with the material contents of the universe via the action of a field. This should not be confused with the claim that the fermions acquire their masses as a result of interactions with Higgs fields, for the field in ...
Chemical Bonding II
... called the electron sea model. The structure if viewed as metal ions sitting in a sea of valence electrons. Since the electrons are not associated with a particular nucleus or atom, they are free to move and conduct an electrical current. ...
... called the electron sea model. The structure if viewed as metal ions sitting in a sea of valence electrons. Since the electrons are not associated with a particular nucleus or atom, they are free to move and conduct an electrical current. ...
Atomic theory
In chemistry and physics, atomic theory is a scientific theory of the nature of matter, which states that matter is composed of discrete units called atoms. It began as a philosophical concept in ancient Greece and entered the scientific mainstream in the early 19th century when discoveries in the field of chemistry showed that matter did indeed behave as if it were made up of atoms.The word atom comes from the Ancient Greek adjective atomos, meaning ""uncuttable"". 19th century chemists began using the term in connection with the growing number of irreducible chemical elements. While seemingly apropos, around the turn of the 20th century, through various experiments with electromagnetism and radioactivity, physicists discovered that the so-called ""uncuttable atom"" was actually a conglomerate of various subatomic particles (chiefly, electrons, protons and neutrons) which can exist separately from each other. In fact, in certain extreme environments, such as neutron stars, extreme temperature and pressure prevents atoms from existing at all. Since atoms were found to be divisible, physicists later invented the term ""elementary particles"" to describe the ""uncuttable"", though not indestructible, parts of an atom. The field of science which studies subatomic particles is particle physics, and it is in this field that physicists hope to discover the true fundamental nature of matter.