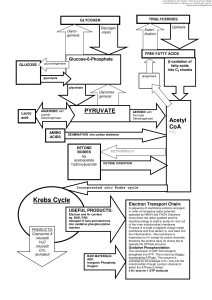
Photosynth-Cellular Respiration
... High-energy electrons move through the electron transport chain from photosystem II to photosystem I. As electrons pass from chlorophyll to NADP+, more hydrogen ions are pumped across the membrane. ATP synthase in the membrane allows H+ ions to pass through it. The enzyme binds ADP and a phosphate g ...
... High-energy electrons move through the electron transport chain from photosystem II to photosystem I. As electrons pass from chlorophyll to NADP+, more hydrogen ions are pumped across the membrane. ATP synthase in the membrane allows H+ ions to pass through it. The enzyme binds ADP and a phosphate g ...
Krebs Cycle - Deranged Physiology
... operated by NADH and FADH. Electrons move down the redox gradient and the resulting energy is used to pump H+ ions out of the inner mitochondrial membrane. Purpose is to build a negative charge inside membrane and thus attract H+ ions back into the mitochondrion. (the membrane is impervious to H+ ex ...
... operated by NADH and FADH. Electrons move down the redox gradient and the resulting energy is used to pump H+ ions out of the inner mitochondrial membrane. Purpose is to build a negative charge inside membrane and thus attract H+ ions back into the mitochondrion. (the membrane is impervious to H+ ex ...
Aerobic Respiration
... then enters the Kreb’s cycle in mitochondria. • During the Kreb’s cycle, citrate is reconverted to oxaloacetate in a series of small steps. • These processes involve decarboxylation and dehydrogenation, using NAD and FAD as hydrogen carriers. • For each turn of the cycle 2 CO2 molecules are produced ...
... then enters the Kreb’s cycle in mitochondria. • During the Kreb’s cycle, citrate is reconverted to oxaloacetate in a series of small steps. • These processes involve decarboxylation and dehydrogenation, using NAD and FAD as hydrogen carriers. • For each turn of the cycle 2 CO2 molecules are produced ...
Study Guide Chapter 10 Photosynthesis
... form; without plagiarizing) that explains the differences between C3, C4, and CAM plants. Also explain why these different evolutionary paths were essential in overcoming photorespiration. (Turn this in for a grade). Study Guide: Page 73 Read starting with Alternative mechanisms of carbon fixation h ...
... form; without plagiarizing) that explains the differences between C3, C4, and CAM plants. Also explain why these different evolutionary paths were essential in overcoming photorespiration. (Turn this in for a grade). Study Guide: Page 73 Read starting with Alternative mechanisms of carbon fixation h ...
the Four Stages of Biochemical Energy Production
... Citric acid cycle – For every glucose, two acetyl groups enter the citric acid cycle (Krebs cycle) • Each two-carbon acetyl group combines with a fourcarbon compound • Two CO2 molecules are removed (why is this important?) • Energy captured as 1 ATP, 3 NADH, and 1 FADH2 form from each acetyl group ...
... Citric acid cycle – For every glucose, two acetyl groups enter the citric acid cycle (Krebs cycle) • Each two-carbon acetyl group combines with a fourcarbon compound • Two CO2 molecules are removed (why is this important?) • Energy captured as 1 ATP, 3 NADH, and 1 FADH2 form from each acetyl group ...
CHEMISTRY
... Smallest unit nucleus: center/core is most of the mass of the atom a. protons: + charge ...
... Smallest unit nucleus: center/core is most of the mass of the atom a. protons: + charge ...
Photosynthesis
... 24(OH) ¾¾¾12H2O + 6O2. 2. Dark Reaction (in stroma) : 6CO2 + 18ATP + 12NADPH ¾¾¾C6H12O6 + 6H2O + 18 ADP + 18P (i) +12NADP+. Q3. Where does non-cyclic photophosphorylation occur? Why is it known as non cyclic? Ans3. Non-cyclic photophosphorylation occurs normally in green plants. Noncyclic photo ...
... 24(OH) ¾¾¾12H2O + 6O2. 2. Dark Reaction (in stroma) : 6CO2 + 18ATP + 12NADPH ¾¾¾C6H12O6 + 6H2O + 18 ADP + 18P (i) +12NADP+. Q3. Where does non-cyclic photophosphorylation occur? Why is it known as non cyclic? Ans3. Non-cyclic photophosphorylation occurs normally in green plants. Noncyclic photo ...
Cell Energy - Denton ISD
... • Process that releases chemical energy from sugars and other carbon-based molecules to make ATP when oxygen is present. • Aerobic process- it requires oxygen • Takes place in the mitochondria ...
... • Process that releases chemical energy from sugars and other carbon-based molecules to make ATP when oxygen is present. • Aerobic process- it requires oxygen • Takes place in the mitochondria ...
UNIT 4 STUDY GUIDE: Energetics
... 10) Where do the light reactions take place? Where does the Calvin cycle take place? 11) Summarize the “main events” of the light reactions. (HINT: your summary should include 7-10 details or steps!...in other words more explanation than: “light goes in, ATP and NADPH come out”) ...
... 10) Where do the light reactions take place? Where does the Calvin cycle take place? 11) Summarize the “main events” of the light reactions. (HINT: your summary should include 7-10 details or steps!...in other words more explanation than: “light goes in, ATP and NADPH come out”) ...
Fall Exam 3
... Orbital energies increase in the order 3s < 3p < 3d because orbital penetration decreases in the order 3s > 3p > 3d. Orbital energies increase in the order 3s < 3p < 3d because the Schrödinger equation predicts that orbital energy depends only on the angular momentum quantum number, l. Orbital energ ...
... Orbital energies increase in the order 3s < 3p < 3d because orbital penetration decreases in the order 3s > 3p > 3d. Orbital energies increase in the order 3s < 3p < 3d because the Schrödinger equation predicts that orbital energy depends only on the angular momentum quantum number, l. Orbital energ ...
5. Nucleotides are covalently linked to form nucleic acids by the
... Chemical bond energy derived from exergonic reactions (e.g. the catabolic breakdown of food) is often temporarily stored in molecules called ...
... Chemical bond energy derived from exergonic reactions (e.g. the catabolic breakdown of food) is often temporarily stored in molecules called ...
Properties of Light λ
... = photons of light from 400-700nm = unit is microEinsteins Æ uE m-2 s-1 Einstein = 1 mole of photons microEinstein = 10-6 mole photons ...
... = photons of light from 400-700nm = unit is microEinsteins Æ uE m-2 s-1 Einstein = 1 mole of photons microEinstein = 10-6 mole photons ...
Chapter 9 - web.biosci.utexas.edu
... – energy source oxidized and degraded using molecules other than oxygen as exogenous electron acceptors – can yield large amount of energy (depending on reduction potential of energy source and electron acceptor), primarily by electron transport activity ...
... – energy source oxidized and degraded using molecules other than oxygen as exogenous electron acceptors – can yield large amount of energy (depending on reduction potential of energy source and electron acceptor), primarily by electron transport activity ...
Mitochondria: Energy Conversion
... mitochondria have allowed the above electrochemical reactions to be carried out effectively ...
... mitochondria have allowed the above electrochemical reactions to be carried out effectively ...
Chapter 2-1 The Nature of Matter
... B. Energy in Reactions – released or absorbed 1. RXNS (reactions) that release energy are spontaneous – produce heat light and sound 2. RXNS that absorb energy must have a source of energy – plants (sunlight) and animals (food) 3. Activation Energy – energy needed to get reaction started ...
... B. Energy in Reactions – released or absorbed 1. RXNS (reactions) that release energy are spontaneous – produce heat light and sound 2. RXNS that absorb energy must have a source of energy – plants (sunlight) and animals (food) 3. Activation Energy – energy needed to get reaction started ...
Biochemistry Test Review Cards
... 36. Enzymes are a type of __protein__ that is required for chemical__ reactions to Red is with an enzyme and black occur. is without ...
... 36. Enzymes are a type of __protein__ that is required for chemical__ reactions to Red is with an enzyme and black occur. is without ...
Aerobic Metabolism ii: electron transport chain
... During the oxidation of NADH there are 3 steps in which the change in reduction potential is sufficient for ATP synthesis. This steps occurs in complexes I, III and IV. The resulting transmembrane proton gradient is used to make ATP via ATP synthase. Recent experimental evidence indicates that appro ...
... During the oxidation of NADH there are 3 steps in which the change in reduction potential is sufficient for ATP synthesis. This steps occurs in complexes I, III and IV. The resulting transmembrane proton gradient is used to make ATP via ATP synthase. Recent experimental evidence indicates that appro ...
Cellular Respiration & Photosynthesis notes
... needed and oxygen is produced from photosynthesis. In summary, photophosphorylation take the energy in light and electrons in water to make the energy rich molecules ATP and NADPH: Water + ADP + phosphate + NADP + light--> ATP + NADPH + oxygen ...
... needed and oxygen is produced from photosynthesis. In summary, photophosphorylation take the energy in light and electrons in water to make the energy rich molecules ATP and NADPH: Water + ADP + phosphate + NADP + light--> ATP + NADPH + oxygen ...
國立屏東教育大學95學年度研究所碩士班入學考試
... 1. If matter is uniform throughout, cannot be separated into other substances by physical processes, but can be decomposed into other substances by chemical processes, it is called a (an) __________. (A) heterogeneous mixture (B) element (C) homogeneous mixture (D) compound (E) mixture of elements 2 ...
... 1. If matter is uniform throughout, cannot be separated into other substances by physical processes, but can be decomposed into other substances by chemical processes, it is called a (an) __________. (A) heterogeneous mixture (B) element (C) homogeneous mixture (D) compound (E) mixture of elements 2 ...
Quiz #3 - San Diego Mesa College
... A) is released in a sudden combustion reaction B) is used to manufacture ATP by coupling its build up to exergonic chemical reactions C) is released a little bit at a time in a series of chemical reactions D) is released after fermentation E) both, b and c Q. 16: Which of the following is NOT true o ...
... A) is released in a sudden combustion reaction B) is used to manufacture ATP by coupling its build up to exergonic chemical reactions C) is released a little bit at a time in a series of chemical reactions D) is released after fermentation E) both, b and c Q. 16: Which of the following is NOT true o ...
photosynthesis
... by absorbing certain wavelengths (blue-420 nm and red-660 nm are most important). • Plants are green because the green wavelength is reflected, not absorbed. ...
... by absorbing certain wavelengths (blue-420 nm and red-660 nm are most important). • Plants are green because the green wavelength is reflected, not absorbed. ...