Structure and Reactivity
... Contents in Brief . . . . . . . . . . . . . . . . . . . . . Introduction . . . . . . . . . . . . . . . . . . . . . . . . Electronegativity and Bond Polarity . . . . . . . . . . . Molecular Polarity and Dipole Moment . . . . . . . . . Polarity of Polyatomic Ions and Ionic Compounds . . . Non-covalent ...
... Contents in Brief . . . . . . . . . . . . . . . . . . . . . Introduction . . . . . . . . . . . . . . . . . . . . . . . . Electronegativity and Bond Polarity . . . . . . . . . . . Molecular Polarity and Dipole Moment . . . . . . . . . Polarity of Polyatomic Ions and Ionic Compounds . . . Non-covalent ...
Chemical Reactivity as Described by Quantum Chemical Methods
... aspects can be included when further linking reactivity to the property vertex. In the field of organic chemistry, the ab initio calculation of functional group properties and their use in studies on acidity and basicity is discussed together with the use of DFT descriptors to study the kinetics of ...
... aspects can be included when further linking reactivity to the property vertex. In the field of organic chemistry, the ab initio calculation of functional group properties and their use in studies on acidity and basicity is discussed together with the use of DFT descriptors to study the kinetics of ...
Chapter 18.2
... Based on this simple idea, we can follow a set of rules to assign oxidation numbers . We go through the set of rules until we find the FIRST rule that applies to our specific atom in the compound or ion of interest. Oxidation number rules (Rule 1) Atoms of pure elemental compounds (e.g. metals, soli ...
... Based on this simple idea, we can follow a set of rules to assign oxidation numbers . We go through the set of rules until we find the FIRST rule that applies to our specific atom in the compound or ion of interest. Oxidation number rules (Rule 1) Atoms of pure elemental compounds (e.g. metals, soli ...
Chem101 - Lecture 2 Elements Elements
... characteristic number of isotopes and relative abundance of each. • For example ...
... characteristic number of isotopes and relative abundance of each. • For example ...
Complete the following equations
... While ionization energy generally increases from left to right across period in the periodic table, a certain anomaly is observed in this trend. For example, in the second period, ionization energy decreases from Be to B and from N to O; in the third period, ionization energy decreases from Mg to Al ...
... While ionization energy generally increases from left to right across period in the periodic table, a certain anomaly is observed in this trend. For example, in the second period, ionization energy decreases from Be to B and from N to O; in the third period, ionization energy decreases from Mg to Al ...
Chapter 3 - Higher Education | Kendall Hunt Publishing
... exactly like every other part. The salt can be separated from the water by physical means. Seawater and air are also examples of homogeneous mixtures. We know that there are two types of homogeneous matter: pure substances and homogeneous mixtures. According to this classification scheme, matter can ...
... exactly like every other part. The salt can be separated from the water by physical means. Seawater and air are also examples of homogeneous mixtures. We know that there are two types of homogeneous matter: pure substances and homogeneous mixtures. According to this classification scheme, matter can ...
General Safety
... The nature of matter, atomic structure, electromagnetic radiation and the electromagnetic spectrum, the atomic spectrum of Hydrogen and the Bohr model, the quantum mechanical model of the atom, the Photoelectric Effect, quantum numbers, orbital shapes and energies, electron spin (Pauli Exclusion Pri ...
... The nature of matter, atomic structure, electromagnetic radiation and the electromagnetic spectrum, the atomic spectrum of Hydrogen and the Bohr model, the quantum mechanical model of the atom, the Photoelectric Effect, quantum numbers, orbital shapes and energies, electron spin (Pauli Exclusion Pri ...
Shell Structure of Nuclei and Cold Atomic Gases in Traps
... II. Fluctuations and shell structure of BCS gaps in nuclei well described by periodic orbit theory. Non-universal corrections to BCS fluctuations important (beyond RMT). III. Cold Fermi-gas in 2D traps - Detailed shell structure: Hund’s rule for repulsive int.; Pairing type for attractive int. ...
... II. Fluctuations and shell structure of BCS gaps in nuclei well described by periodic orbit theory. Non-universal corrections to BCS fluctuations important (beyond RMT). III. Cold Fermi-gas in 2D traps - Detailed shell structure: Hund’s rule for repulsive int.; Pairing type for attractive int. ...
Chapter 1-3 Exam Review
... 2. I can determine the number of protons, neutrons and electrons in isotopes and in ions. 3. describe the works of John Dalton, J.J. Thomson (cathode ray tube), Robert Millikan (Oil Drop Experiment) and Ernst Rutherford (Gold Foil Experiment). 4. use the periodic table to predict the charges of mona ...
... 2. I can determine the number of protons, neutrons and electrons in isotopes and in ions. 3. describe the works of John Dalton, J.J. Thomson (cathode ray tube), Robert Millikan (Oil Drop Experiment) and Ernst Rutherford (Gold Foil Experiment). 4. use the periodic table to predict the charges of mona ...
01 introduction to quantum physics
... In quantum theory, what you know is what you measure (or what some physical system “records”). The acts of measurement and observation can create the resulting state. A system does not have a definite value for a quantity until it is observed. Thus an electron is given a specific spin by an observat ...
... In quantum theory, what you know is what you measure (or what some physical system “records”). The acts of measurement and observation can create the resulting state. A system does not have a definite value for a quantity until it is observed. Thus an electron is given a specific spin by an observat ...
Resonances in three-body systems S U L
... mechanics whose approximate solution provides some insight into the more complex analysis associated with few-body problems. We have investigated resonance states composed of three particles interacting via Coulombic and more general potentials in non-relativistic quantum mechanics, using the comple ...
... mechanics whose approximate solution provides some insight into the more complex analysis associated with few-body problems. We have investigated resonance states composed of three particles interacting via Coulombic and more general potentials in non-relativistic quantum mechanics, using the comple ...
Effects of antioxidants for the degradation of flame
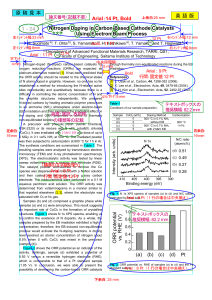
... 6 MGy in 0.1 vol% NH3 at 500 °C. The irradiated powder was then subjected to carbonization at 800 °C for 1 h in Ar. The synthesis conditions are summarized in Table 1. The resulting samples were analyzed by transmission electron microscopy (TEM) and X-ray photoelectron spectroscopy (XPS). The electr ...
... 6 MGy in 0.1 vol% NH3 at 500 °C. The irradiated powder was then subjected to carbonization at 800 °C for 1 h in Ar. The synthesis conditions are summarized in Table 1. The resulting samples were analyzed by transmission electron microscopy (TEM) and X-ray photoelectron spectroscopy (XPS). The electr ...
Phys. Rev. Lett. 104, 043002 (2010)
... small ensembles confined to a few micrometers, a single Rydberg atom can entirely block any further excitation. This, recently observed, ‘‘dipole blockade’’ [1–4], enables the production of highly entangled collective states with potential applications for fast quantum information processing [2,5,6] ...
... small ensembles confined to a few micrometers, a single Rydberg atom can entirely block any further excitation. This, recently observed, ‘‘dipole blockade’’ [1–4], enables the production of highly entangled collective states with potential applications for fast quantum information processing [2,5,6] ...
DEVELOPMENT, IMPLEMENTATION AND APPLICATION OF ELECTRONIC STRUCTURAL DESCRIPTORS TO THE
... Roughly and generally speaking, theoretical chemistry may be defined as the use of non-experimental reasoning to explain or predict chemical phenomena. Therefore, a theoretical chemist uses chemical, physical, mathematical and computing skills to study chemical systems. In theoretical chemistry, che ...
... Roughly and generally speaking, theoretical chemistry may be defined as the use of non-experimental reasoning to explain or predict chemical phenomena. Therefore, a theoretical chemist uses chemical, physical, mathematical and computing skills to study chemical systems. In theoretical chemistry, che ...
Electron configuration
In atomic physics and quantum chemistry, the electron configuration is the distribution of electrons of an atom or molecule (or other physical structure) in atomic or molecular orbitals. For example, the electron configuration of the neon atom is 1s2 2s2 2p6.Electronic configurations describe electrons as each moving independently in an orbital, in an average field created by all other orbitals. Mathematically, configurations are described by Slater determinants or configuration state functions.According to the laws of quantum mechanics, for systems with only one electron, an energy is associated with each electron configuration and, upon certain conditions, electrons are able to move from one configuration to another by the emission or absorption of a quantum of energy, in the form of a photon.Knowledge of the electron configuration of different atoms is useful in understanding the structure of the periodic table of elements. The concept is also useful for describing the chemical bonds that hold atoms together. In bulk materials, this same idea helps explain the peculiar properties of lasers and semiconductors.