76 kJ/mole
... to get filled, half filled or empty outermost orbitals, and/or like to become isoelectronic with their closest noble gas neighbor ...
... to get filled, half filled or empty outermost orbitals, and/or like to become isoelectronic with their closest noble gas neighbor ...
Chemistry Note PowerPoint
... • An atom’s valance electrons are those that have the highest energy levels and are held most loosely. • The number of valance electrons determine many properties of that element, including the ways in which the atom combines with other atoms ...
... • An atom’s valance electrons are those that have the highest energy levels and are held most loosely. • The number of valance electrons determine many properties of that element, including the ways in which the atom combines with other atoms ...
Chapter 3 : Simple Bonding Theory Why do they make chemical
... • It is important to keep in mind that the models we are discussing are just that…..models. • We are operating under the assumption that when forming bonds, atoms “share” electrons using atomic ...
... • It is important to keep in mind that the models we are discussing are just that…..models. • We are operating under the assumption that when forming bonds, atoms “share” electrons using atomic ...
The (Integer) Quantum Hall Effect
... single-particle wavefunctions are still basically valid. In this situation, we are able to treat the system as a degenerate electron gas – the electrons are “free” and do not interact with each other, but we must take into account the Pauli exclusion principle, and cannot give two electrons the same ...
... single-particle wavefunctions are still basically valid. In this situation, we are able to treat the system as a degenerate electron gas – the electrons are “free” and do not interact with each other, but we must take into account the Pauli exclusion principle, and cannot give two electrons the same ...
Quantum Theory of the Atom
... energies emitted from photons acting like waves. • If it is not in the visible light range, it may be giving off other forms of electromagnetic radiation like radio, microwaves, infrared, ultra violet, x-rays, or gamma rays. • Used to determine which elements are present in a star (because stars are ...
... energies emitted from photons acting like waves. • If it is not in the visible light range, it may be giving off other forms of electromagnetic radiation like radio, microwaves, infrared, ultra violet, x-rays, or gamma rays. • Used to determine which elements are present in a star (because stars are ...
Budiansky Cover
... Panza’s little had forgotten and the good beast molecules?” donkey came from. reappeared. ...
... Panza’s little had forgotten and the good beast molecules?” donkey came from. reappeared. ...
Lecture 30: Molecular interactions
... H2O is a weak ligand. On the other hand, if we consider a strong ligand such as CN-, the splitting on the two sets of energy levels is greater. Now the minimum energy configuration of the system involves electron pairing as shown in the right figure. ...
... H2O is a weak ligand. On the other hand, if we consider a strong ligand such as CN-, the splitting on the two sets of energy levels is greater. Now the minimum energy configuration of the system involves electron pairing as shown in the right figure. ...
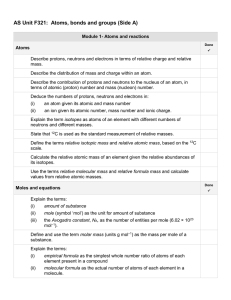
AS Unit F321 Unit 1 Side A check list
... State the formulae of the common acids: hydrochloric, 2ulphuric and nitric acids. State that common bases are metal oxides, metal hydroxides and ammonia. State that an alkali is a soluble base that releases OH– ions in aqueous solution. State the formulae of the common alkalis: sodium hydroxide, pot ...
... State the formulae of the common acids: hydrochloric, 2ulphuric and nitric acids. State that common bases are metal oxides, metal hydroxides and ammonia. State that an alkali is a soluble base that releases OH– ions in aqueous solution. State the formulae of the common alkalis: sodium hydroxide, pot ...
TAP507-0: Electron standing waves
... standing waves within an atom. It is therefore useful first to demonstrate standing waves on a stretched elastic cord. This is known as Melde’s experiment. ...
... standing waves within an atom. It is therefore useful first to demonstrate standing waves on a stretched elastic cord. This is known as Melde’s experiment. ...
Chapter 6 lecture 1
... Regardless of intensity, if the frequency of the radiation is not above a certain threshold, an electron will not be emitted by the metal This phenomenon cannot be explained using the ‘wave’ notion of radiation Einstein: assumes that radiation isn’t a continuous wave, but exists as particles, or pho ...
... Regardless of intensity, if the frequency of the radiation is not above a certain threshold, an electron will not be emitted by the metal This phenomenon cannot be explained using the ‘wave’ notion of radiation Einstein: assumes that radiation isn’t a continuous wave, but exists as particles, or pho ...
Module 4 Trivia Review
... Bohr used the prior knowledge of an atom and discovered that the electrons are actually orbiting around the nucleus that resembled the solar system. Bohr discovered and started Quantum Mechanics. Dmitri Mendeleev set out to organize the known elements according to their properties and called it the ...
... Bohr used the prior knowledge of an atom and discovered that the electrons are actually orbiting around the nucleus that resembled the solar system. Bohr discovered and started Quantum Mechanics. Dmitri Mendeleev set out to organize the known elements according to their properties and called it the ...
General Chemistry I Syllabus
... each model explains the behavior of substances. We see how metals and nonmetals combine via three types of bonding and learn how to depict atoms and ions with Lewis symbols. We look at how a bond forms and discuss the relations among bond order, energy, and length. We explore the relationship betwee ...
... each model explains the behavior of substances. We see how metals and nonmetals combine via three types of bonding and learn how to depict atoms and ions with Lewis symbols. We look at how a bond forms and discuss the relations among bond order, energy, and length. We explore the relationship betwee ...
Electron configuration
In atomic physics and quantum chemistry, the electron configuration is the distribution of electrons of an atom or molecule (or other physical structure) in atomic or molecular orbitals. For example, the electron configuration of the neon atom is 1s2 2s2 2p6.Electronic configurations describe electrons as each moving independently in an orbital, in an average field created by all other orbitals. Mathematically, configurations are described by Slater determinants or configuration state functions.According to the laws of quantum mechanics, for systems with only one electron, an energy is associated with each electron configuration and, upon certain conditions, electrons are able to move from one configuration to another by the emission or absorption of a quantum of energy, in the form of a photon.Knowledge of the electron configuration of different atoms is useful in understanding the structure of the periodic table of elements. The concept is also useful for describing the chemical bonds that hold atoms together. In bulk materials, this same idea helps explain the peculiar properties of lasers and semiconductors.