Example 4: A one-electron atom is irradiated with visible light. The
... nucleus then a force of attraction between the two will develop. If we now want to remove the electron back to an infinite distance away from the nucleus we must supply energy to overcome the force of attraction between the nucleus and the electron. If we supply energy to a system, this, again by co ...
... nucleus then a force of attraction between the two will develop. If we now want to remove the electron back to an infinite distance away from the nucleus we must supply energy to overcome the force of attraction between the nucleus and the electron. If we supply energy to a system, this, again by co ...
In-Class Exam - Fayetteville State University
... 14. Isotopes are atoms that have the same number of ______ but differing number of ______. A) neutrons, protons B) protons, electrons C) neutrons, electrons D) electrons, protons ...
... 14. Isotopes are atoms that have the same number of ______ but differing number of ______. A) neutrons, protons B) protons, electrons C) neutrons, electrons D) electrons, protons ...
Introduction to Quantum Mechanic
... • What are the possible values of l and ml for an electron with the principle quantum number n=4? • If l=0, ml=0 • If l=1, ml= -1, 0, +1 • If l=2, ml= -2,-1,0,+1, +2 • If l=3, ml= -3, -2, -1, 0, +1, +2, +3 ...
... • What are the possible values of l and ml for an electron with the principle quantum number n=4? • If l=0, ml=0 • If l=1, ml= -1, 0, +1 • If l=2, ml= -2,-1,0,+1, +2 • If l=3, ml= -3, -2, -1, 0, +1, +2, +3 ...
lecture 11 (zipped power point)
... A photon having the cut-off frequency n0 has just enough energy to eject the photoelectron and none extra to appear as kinetic energy. Photon of energy less than hn0 has not sufficient energy to kick out any electron Approximately, electrons that are eject at the cutoff frequency will not leave the ...
... A photon having the cut-off frequency n0 has just enough energy to eject the photoelectron and none extra to appear as kinetic energy. Photon of energy less than hn0 has not sufficient energy to kick out any electron Approximately, electrons that are eject at the cutoff frequency will not leave the ...
Chemistry! - Duplin County Schools
... The letters are chemical symbols The small numbers are subscripts The large numbers are coefficients Knowing this, we can calculate how many atoms of each element are in a chemical formula • For example: ...
... The letters are chemical symbols The small numbers are subscripts The large numbers are coefficients Knowing this, we can calculate how many atoms of each element are in a chemical formula • For example: ...
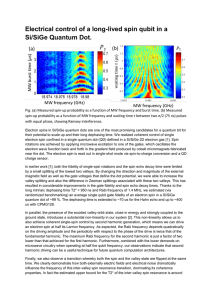
Electrical control of a long-lived spin qubit in a
... In earlier work [1], both the fidelity of single-spin rotations and the spin echo decay time were limited by a small splitting of the lowest two valleys. By changing the direction and magnitude of the external magnetic field as well as the gate voltages that define the dot potential, we were able to ...
... In earlier work [1], both the fidelity of single-spin rotations and the spin echo decay time were limited by a small splitting of the lowest two valleys. By changing the direction and magnitude of the external magnetic field as well as the gate voltages that define the dot potential, we were able to ...
Document
... Structural formulas are used to represent what the atoms look like when they are “brought together” in the chemical bond. To show this, the electron dot diagrams for each atom are drawn and puzzle-pieced together by filling their “empty spots” with each other’s valence electrons. Hint: you might wan ...
... Structural formulas are used to represent what the atoms look like when they are “brought together” in the chemical bond. To show this, the electron dot diagrams for each atom are drawn and puzzle-pieced together by filling their “empty spots” with each other’s valence electrons. Hint: you might wan ...
Document
... ACT: CheckPoint 3.2 Suppose the charge of the nucleus is doubled (+2e), but the e– charge remains the same ( the e remains the same (–e). e). How does r How does r for the for the ground state (n = 1) orbit compare to that in hydrogen? n2 2 For hydrogen: rn = ...
... ACT: CheckPoint 3.2 Suppose the charge of the nucleus is doubled (+2e), but the e– charge remains the same ( the e remains the same (–e). e). How does r How does r for the for the ground state (n = 1) orbit compare to that in hydrogen? n2 2 For hydrogen: rn = ...
chem 3374a quantum chemistry and spectroscopy
... 6. Principles of rotational spectroscopy. The Schrödinger equation for rotation in two and three dimensions. Angular momentum and its quantization. Spherical harmonics. The rigid rotor and rotational spectroscopy of diatomic molecules. 7. The structure and spectra of hydrogenic atoms. The Schrödin ...
... 6. Principles of rotational spectroscopy. The Schrödinger equation for rotation in two and three dimensions. Angular momentum and its quantization. Spherical harmonics. The rigid rotor and rotational spectroscopy of diatomic molecules. 7. The structure and spectra of hydrogenic atoms. The Schrödin ...
Reduction and Oxidation
... bond. It can be considered as the charge that an atom would have if the more electronegative atom in a bond obtains the two electrons of the bond completely. When an element is ascribed a particular oxidation number, it is called the element is in a specific oxidation sta ...
... bond. It can be considered as the charge that an atom would have if the more electronegative atom in a bond obtains the two electrons of the bond completely. When an element is ascribed a particular oxidation number, it is called the element is in a specific oxidation sta ...
Chem 1 Worksheets WSHEET 1: Working with Numbers Practice
... 4. Who proposed the principle which states that one cannot simultaneously know the exact position and velocity of a particle? _________________ 5. Select the arrangement of electromagnetic radiation which starts with the lowest energy and increases to greatest energy. A. radio, visible, infrared, ul ...
... 4. Who proposed the principle which states that one cannot simultaneously know the exact position and velocity of a particle? _________________ 5. Select the arrangement of electromagnetic radiation which starts with the lowest energy and increases to greatest energy. A. radio, visible, infrared, ul ...
Flame Test Lab
... The difference in energies between the two levels is emitted in the form of a photon (aka “quantum”) of electromagnetic radiation. The energy of each photon is described by the equation E = hv, where h is Planck’s constant (6.63 x 10 -34 Js) and v is the frequency of the radiation. If the wavelength ...
... The difference in energies between the two levels is emitted in the form of a photon (aka “quantum”) of electromagnetic radiation. The energy of each photon is described by the equation E = hv, where h is Planck’s constant (6.63 x 10 -34 Js) and v is the frequency of the radiation. If the wavelength ...
final study guide answers - Ponce
... _________ A particle that has no charge _________ The part of the atom where all of the mass of the atom is contained _________ Positively charge particles found in the atom _________ Atoms that have the same number of protons but a different number of neutrons. _________The sum of the protons and n ...
... _________ A particle that has no charge _________ The part of the atom where all of the mass of the atom is contained _________ Positively charge particles found in the atom _________ Atoms that have the same number of protons but a different number of neutrons. _________The sum of the protons and n ...
Evidence of Correlation in Spin Excitations of Few
... (QDs) have novel ground and excited states that manifest Coulomb interactions at the nanoscale [1]. States of very few electrons are prime candidates for spintronic applications and for the implementation of quantum bits in nanoscale devices [2]. Great attention is, therefore, devoted to the study o ...
... (QDs) have novel ground and excited states that manifest Coulomb interactions at the nanoscale [1]. States of very few electrons are prime candidates for spintronic applications and for the implementation of quantum bits in nanoscale devices [2]. Great attention is, therefore, devoted to the study o ...
Atoms and Molecules - New Age International
... ∴ a sin kl + b cos kl = 0 It can only be possible when, nπ l where x is called quantum number and is equal to 1, 2, 3 ... ∞. ...
... ∴ a sin kl + b cos kl = 0 It can only be possible when, nπ l where x is called quantum number and is equal to 1, 2, 3 ... ∞. ...
Electron configuration
In atomic physics and quantum chemistry, the electron configuration is the distribution of electrons of an atom or molecule (or other physical structure) in atomic or molecular orbitals. For example, the electron configuration of the neon atom is 1s2 2s2 2p6.Electronic configurations describe electrons as each moving independently in an orbital, in an average field created by all other orbitals. Mathematically, configurations are described by Slater determinants or configuration state functions.According to the laws of quantum mechanics, for systems with only one electron, an energy is associated with each electron configuration and, upon certain conditions, electrons are able to move from one configuration to another by the emission or absorption of a quantum of energy, in the form of a photon.Knowledge of the electron configuration of different atoms is useful in understanding the structure of the periodic table of elements. The concept is also useful for describing the chemical bonds that hold atoms together. In bulk materials, this same idea helps explain the peculiar properties of lasers and semiconductors.