ch28_lecture
... His model includes both classical and non-classical ideas His model included an attempt to explain why the atom was stable ...
... His model includes both classical and non-classical ideas His model included an attempt to explain why the atom was stable ...
Chapter 28
... His model includes both classical and non-classical ideas His model included an attempt to explain why the atom was stable ...
... His model includes both classical and non-classical ideas His model included an attempt to explain why the atom was stable ...
Ground State
... Pieter Zeeman, Lorentz “spectra line splitting” in magnetic filed 1902 Nobel Prize ...
... Pieter Zeeman, Lorentz “spectra line splitting” in magnetic filed 1902 Nobel Prize ...
Unit 2 Review Questions Fill in the blank In a(n) change, a new
... The mass number is the sum of electrons and protons in the atom. l. A Bohr diagram shows electrons in orbits about the nucleus. m. A row of the periodic table is called a period. n. The size of atoms increase down a column of the periodic table. o. Alkali metals include fluorine, chlorine, and iodin ...
... The mass number is the sum of electrons and protons in the atom. l. A Bohr diagram shows electrons in orbits about the nucleus. m. A row of the periodic table is called a period. n. The size of atoms increase down a column of the periodic table. o. Alkali metals include fluorine, chlorine, and iodin ...
The Chemical Earth (8.2.3)
... difference between solids, liquids and gases. 2. In terms of size and bonding, what is the difference between argon, oxygen, and helium. 3. Draw Lewis electron dot diagrams for: a. ...
... difference between solids, liquids and gases. 2. In terms of size and bonding, what is the difference between argon, oxygen, and helium. 3. Draw Lewis electron dot diagrams for: a. ...
Vocabulary:
... Nuclear Atom Model – An atom is mostly empty space with a dense, positively charged nucleus in the center and electrons moving around it. Neils Bohr – ...
... Nuclear Atom Model – An atom is mostly empty space with a dense, positively charged nucleus in the center and electrons moving around it. Neils Bohr – ...
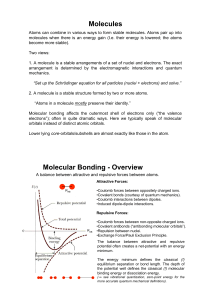
Molecules Molecular Bonding
... The energy minimum defines the classical (!) equilibrium separation or bond length. The depth of the potential well defines the classical (!) molecular binding energy or dissociation energy. (! see vibrational quantization, zero-point energy for the more accurate quantum mechanical definitions). ...
... The energy minimum defines the classical (!) equilibrium separation or bond length. The depth of the potential well defines the classical (!) molecular binding energy or dissociation energy. (! see vibrational quantization, zero-point energy for the more accurate quantum mechanical definitions). ...
Lecture 11
... which in turn yields a wave function unl that is not normalizable. Therefore we must have cq = 0 for some finite value of q, that we will denote q = nr > 0. According to Eq. (11.28) this can only happen if: ...
... which in turn yields a wave function unl that is not normalizable. Therefore we must have cq = 0 for some finite value of q, that we will denote q = nr > 0. According to Eq. (11.28) this can only happen if: ...
The Bohr atom and the Uncertainty Principle
... The probability of detecting a photon at a particular point is directly proportional to the square of lightwave amplitude function at that point P(x) is called probability density (measured in m-1) P(x) |A(x)|2 A(x)=amplitude function of EM wave Similarly for an electron we can describe it with a wa ...
... The probability of detecting a photon at a particular point is directly proportional to the square of lightwave amplitude function at that point P(x) is called probability density (measured in m-1) P(x) |A(x)|2 A(x)=amplitude function of EM wave Similarly for an electron we can describe it with a wa ...
LOYOLA COLLEGE (AUTONOMOUS), CHENNAI-600034 M.Sc. Part-A NOVEMBER 2015
... Derive time independent Schrodinger wave equation from time dependent equation. The force constant for H79Br is 392 Nm-1. Calculate the fundamental vibrational frequency and zero point energy of H79Br. Use the method of separation of variables to break up Schrodinger equation for a rigid rotor into ...
... Derive time independent Schrodinger wave equation from time dependent equation. The force constant for H79Br is 392 Nm-1. Calculate the fundamental vibrational frequency and zero point energy of H79Br. Use the method of separation of variables to break up Schrodinger equation for a rigid rotor into ...
Chapter 12
... A quantum of energy is the amount of energy required to move an electron to the next highest energy level. The higher the energy level the farther the electron is from the nucleus (usually) Energy levels are more closely spaced further from the nucleus The higher the energy level the easier it is fo ...
... A quantum of energy is the amount of energy required to move an electron to the next highest energy level. The higher the energy level the farther the electron is from the nucleus (usually) Energy levels are more closely spaced further from the nucleus The higher the energy level the easier it is fo ...
Oops !Power Point File of Physics 2D lecture for Today should have
... General Principles for Atomic Structure for n-electron system: 1. n-electron system is stable when its total energy is minimum 2.Only one electron can exist in a particular quantum state in an atom...not 2 or more ! 3. Shells & SubShells In Atomic Structure : (a) ignore inter-electron repulsion (cru ...
... General Principles for Atomic Structure for n-electron system: 1. n-electron system is stable when its total energy is minimum 2.Only one electron can exist in a particular quantum state in an atom...not 2 or more ! 3. Shells & SubShells In Atomic Structure : (a) ignore inter-electron repulsion (cru ...
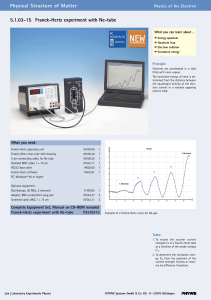
5.1.03-15 Franck-Hertz experiment with Ne
... 1913: An isolated atom consists of a positively charged nucleus about which electrons are distributed in successive orbits. He also postulated that only those orbits occur for which the angular momentum of the electron is an integral multiple of h/2p, i.e. n*h/2p, where n is an integer and h is Plan ...
... 1913: An isolated atom consists of a positively charged nucleus about which electrons are distributed in successive orbits. He also postulated that only those orbits occur for which the angular momentum of the electron is an integral multiple of h/2p, i.e. n*h/2p, where n is an integer and h is Plan ...
Chapter 3: The Structure of Matter
... different from the elements that make it •Example: Liquid water is made up of hydrogen gas and oxygen gas ...
... different from the elements that make it •Example: Liquid water is made up of hydrogen gas and oxygen gas ...
Bohr model
In atomic physics, the Rutherford–Bohr model or Bohr model, introduced by Niels Bohr in 1913, depicts the atom as a small, positively charged nucleus surrounded by electrons that travel in circular orbits around the nucleus—similar in structure to the solar system, but with attraction provided by electrostatic forces rather than gravity. After the cubic model (1902), the plum-pudding model (1904), the Saturnian model (1904), and the Rutherford model (1911) came the Rutherford–Bohr model or just Bohr model for short (1913). The improvement to the Rutherford model is mostly a quantum physical interpretation of it. The Bohr model has been superseded, but the quantum theory remains sound.The model's key success lay in explaining the Rydberg formula for the spectral emission lines of atomic hydrogen. While the Rydberg formula had been known experimentally, it did not gain a theoretical underpinning until the Bohr model was introduced. Not only did the Bohr model explain the reason for the structure of the Rydberg formula, it also provided a justification for its empirical results in terms of fundamental physical constants.The Bohr model is a relatively primitive model of the hydrogen atom, compared to the valence shell atom. As a theory, it can be derived as a first-order approximation of the hydrogen atom using the broader and much more accurate quantum mechanics and thus may be considered to be an obsolete scientific theory. However, because of its simplicity, and its correct results for selected systems (see below for application), the Bohr model is still commonly taught to introduce students to quantum mechanics or energy level diagrams before moving on to the more accurate, but more complex, valence shell atom. A related model was originally proposed by Arthur Erich Haas in 1910, but was rejected. The quantum theory of the period between Planck's discovery of the quantum (1900) and the advent of a full-blown quantum mechanics (1925) is often referred to as the old quantum theory.