quantum, relativistic and classical physics
... momentum S which occurs when an atom is placed in a magnetic field aligned along the z direction. In this connection explain the role of the quantum numbers m and ms and define how L and S are related to the corresponding quantum numbers and s. [7 marks] (ii) Briefly note the experimental observa ...
... momentum S which occurs when an atom is placed in a magnetic field aligned along the z direction. In this connection explain the role of the quantum numbers m and ms and define how L and S are related to the corresponding quantum numbers and s. [7 marks] (ii) Briefly note the experimental observa ...
Early Quantum Theory and Models of the Atom
... The minimum energy level required to remove an electron from the ground state is called the ionization energy For hydrogen is it 13.6eV and precisely corresponds to the energy to go from E1 to E=0 Often shown in an Energy Level Diagram Vertical arrows show transitions Energy released or absorvedcan ...
... The minimum energy level required to remove an electron from the ground state is called the ionization energy For hydrogen is it 13.6eV and precisely corresponds to the energy to go from E1 to E=0 Often shown in an Energy Level Diagram Vertical arrows show transitions Energy released or absorvedcan ...
Lecture 3 Teaching notes
... get it just the same way we did in atoms: we put the first electron into the lowest state, the next one into the next-lowest state, and so on until we have run out of electrons. This will be the actual configuration of the system at zero temperature, since then any system is in its ground configurat ...
... get it just the same way we did in atoms: we put the first electron into the lowest state, the next one into the next-lowest state, and so on until we have run out of electrons. This will be the actual configuration of the system at zero temperature, since then any system is in its ground configurat ...
Balmer Series
... which are sequences of lines corresponding to atomic transitions, each ending or beginning with the same atomic state in hydrogen. Thus, for example, the Balmer Series involves transitions starting (for absorption) or ending (for emission) with the first excited state (n=2) of hydrogen, while the Ly ...
... which are sequences of lines corresponding to atomic transitions, each ending or beginning with the same atomic state in hydrogen. Thus, for example, the Balmer Series involves transitions starting (for absorption) or ending (for emission) with the first excited state (n=2) of hydrogen, while the Ly ...
Atoms: Some Basics
... provided the major impetus for developing quantum mechanics. Balmer’s empirical formula of 1885 had reproduced Angstrom’s observations of spectral lines in hydrogen to 0.1 Åaccuracy, but it was not until 1913 that Bohr gave an explanation for this based on a quantized mechanical model of the atom. ...
... provided the major impetus for developing quantum mechanics. Balmer’s empirical formula of 1885 had reproduced Angstrom’s observations of spectral lines in hydrogen to 0.1 Åaccuracy, but it was not until 1913 that Bohr gave an explanation for this based on a quantized mechanical model of the atom. ...
AP Review – Life and Chemistry Name: Date: ___B_ 1. The atomic
... Calcium’s electrons in orbitals are shown to the left. Notice how the two electrons in the valence shell (outermost shell) are paired? This is done sometimes when only two electrons are in the valence shell – it helps to make sure you don’t “lose them” in the diagram by separating them. To draw ...
... Calcium’s electrons in orbitals are shown to the left. Notice how the two electrons in the valence shell (outermost shell) are paired? This is done sometimes when only two electrons are in the valence shell – it helps to make sure you don’t “lose them” in the diagram by separating them. To draw ...
Interaction of Radiation with Matter
... atoms because they have an excess of energy or mass or both. Unstable atoms are said to be radioactive. In order to reach stability, these atoms give off, or emit, the excess energy or mass. These emissions are called radiation. ...
... atoms because they have an excess of energy or mass or both. Unstable atoms are said to be radioactive. In order to reach stability, these atoms give off, or emit, the excess energy or mass. These emissions are called radiation. ...
Dr.Eman Zakaria Hegazy Quantum Mechanics and Statistical
... The value of Z that minimize E can be interpreted as an effective charge. That fact that Z comes out to be less than 2 reflects the fact that each electron partially screens the nucleus from the other , so that net effective nuclear charge is reduced from 2 to 27/16 (1.68). ...
... The value of Z that minimize E can be interpreted as an effective charge. That fact that Z comes out to be less than 2 reflects the fact that each electron partially screens the nucleus from the other , so that net effective nuclear charge is reduced from 2 to 27/16 (1.68). ...
Population Analysis
... The number of electrons associated with basis function µ on center k is Pµ k ; µ k ( ∆ µ k ; µ k = 1 ) and the number of electrons shared between basis function m on center k and basis function n on center l is 2 Pµ k ;ν l ∆ µ k ;ν l where the factor of two obtains because P and D are symmetric. Exa ...
... The number of electrons associated with basis function µ on center k is Pµ k ; µ k ( ∆ µ k ; µ k = 1 ) and the number of electrons shared between basis function m on center k and basis function n on center l is 2 Pµ k ;ν l ∆ µ k ;ν l where the factor of two obtains because P and D are symmetric. Exa ...
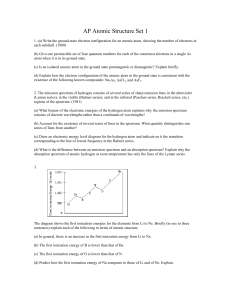
AP Atomic Structure Set 1
... 2. The emission spectrum of hydrogen consists of several series of sharp emission lines in the ultraviolet (Lyman series), in the visible (Balmer series), and in the infrared (Paschen series, Brackett series, etc,) regions of the spectrum. (1981) (a) What feature of the electronic energies of the hy ...
... 2. The emission spectrum of hydrogen consists of several series of sharp emission lines in the ultraviolet (Lyman series), in the visible (Balmer series), and in the infrared (Paschen series, Brackett series, etc,) regions of the spectrum. (1981) (a) What feature of the electronic energies of the hy ...
Q: In which model of the atom do electrons orbit the nucleus? A
... of what block on the periodic table? A: p block ...
... of what block on the periodic table? A: p block ...
Quantum Number - Career Launcher
... If the nitrogen atom had electronic configuration 1s7, it would have energy lower than that of the normal ground state configuration 1s2 2s2 2p3, because the electrons would be closer to the nucleus. Yet 1s7 is not observed because it violates (a) Heisenberg’s uncertainty principle ...
... If the nitrogen atom had electronic configuration 1s7, it would have energy lower than that of the normal ground state configuration 1s2 2s2 2p3, because the electrons would be closer to the nucleus. Yet 1s7 is not observed because it violates (a) Heisenberg’s uncertainty principle ...
Document
... • 1926 – Erwin Schrödinger – formulated his non-relativistic Schrödinger equation, but it incorrectly predicted the magnetic moment of H to be zero in its ground state. • 1927 – T.E. Phipps & J.B. Taylor – reproduced the effect using H atoms in the ground state, thereby eliminating any doubts that m ...
... • 1926 – Erwin Schrödinger – formulated his non-relativistic Schrödinger equation, but it incorrectly predicted the magnetic moment of H to be zero in its ground state. • 1927 – T.E. Phipps & J.B. Taylor – reproduced the effect using H atoms in the ground state, thereby eliminating any doubts that m ...
The mathematical formulations of quantum mechanics are those
... These formulations of quantum mechanics continue to be used today. At the heart of the description are ideas of quantum state and quantum observable which are radically different from those used in previous models of physical reality. While the mathematics permits calculation of many quantities that ...
... These formulations of quantum mechanics continue to be used today. At the heart of the description are ideas of quantum state and quantum observable which are radically different from those used in previous models of physical reality. While the mathematics permits calculation of many quantities that ...
Chapter 9d Introduction to Quantum Mechanics
... space between two rigid reflecting walls but in three dimensional space. For hydrogen atom, a central proton holds the relatively light electron within a region of space whose dimension is of order of 0.1nm. ...
... space between two rigid reflecting walls but in three dimensional space. For hydrogen atom, a central proton holds the relatively light electron within a region of space whose dimension is of order of 0.1nm. ...
Ψ (x,t) = | Ψ (x,t) - University of Notre Dame
... Within the box, the energy is constant – hence ω and k are constant – and also the wavelength λ, and the momentum p Boundary conditions: The wave-function must go to zero outside the box, at very large positive and very large negative x-values Follow Bohr and De Broglie’s ideas and fix the number of ...
... Within the box, the energy is constant – hence ω and k are constant – and also the wavelength λ, and the momentum p Boundary conditions: The wave-function must go to zero outside the box, at very large positive and very large negative x-values Follow Bohr and De Broglie’s ideas and fix the number of ...
Lecture 1.6 PowerPoint
... • 1.6 – I can characterize an electron based on its 4 quantum numbers (n, l, ml, and ms). I can explain what each of these numbers indicate and discuss the importance of these numbers. • 1.7 – I can describe the shape, number, and energy level of the s, p, d, and f orbitals. Furthermore, I can draw ...
... • 1.6 – I can characterize an electron based on its 4 quantum numbers (n, l, ml, and ms). I can explain what each of these numbers indicate and discuss the importance of these numbers. • 1.7 – I can describe the shape, number, and energy level of the s, p, d, and f orbitals. Furthermore, I can draw ...
Bohr model
In atomic physics, the Rutherford–Bohr model or Bohr model, introduced by Niels Bohr in 1913, depicts the atom as a small, positively charged nucleus surrounded by electrons that travel in circular orbits around the nucleus—similar in structure to the solar system, but with attraction provided by electrostatic forces rather than gravity. After the cubic model (1902), the plum-pudding model (1904), the Saturnian model (1904), and the Rutherford model (1911) came the Rutherford–Bohr model or just Bohr model for short (1913). The improvement to the Rutherford model is mostly a quantum physical interpretation of it. The Bohr model has been superseded, but the quantum theory remains sound.The model's key success lay in explaining the Rydberg formula for the spectral emission lines of atomic hydrogen. While the Rydberg formula had been known experimentally, it did not gain a theoretical underpinning until the Bohr model was introduced. Not only did the Bohr model explain the reason for the structure of the Rydberg formula, it also provided a justification for its empirical results in terms of fundamental physical constants.The Bohr model is a relatively primitive model of the hydrogen atom, compared to the valence shell atom. As a theory, it can be derived as a first-order approximation of the hydrogen atom using the broader and much more accurate quantum mechanics and thus may be considered to be an obsolete scientific theory. However, because of its simplicity, and its correct results for selected systems (see below for application), the Bohr model is still commonly taught to introduce students to quantum mechanics or energy level diagrams before moving on to the more accurate, but more complex, valence shell atom. A related model was originally proposed by Arthur Erich Haas in 1910, but was rejected. The quantum theory of the period between Planck's discovery of the quantum (1900) and the advent of a full-blown quantum mechanics (1925) is often referred to as the old quantum theory.