p15_11_6.pdf
... be obtained from the one-body density, but, instead of working directly with the density, TDDFT operates by propagating a system of non-interacting electrons, the Kohn–Sham system, that reproduces the exact interacting density. The potential in the equation for the Kohn–Sham orbitals is defined such ...
... be obtained from the one-body density, but, instead of working directly with the density, TDDFT operates by propagating a system of non-interacting electrons, the Kohn–Sham system, that reproduces the exact interacting density. The potential in the equation for the Kohn–Sham orbitals is defined such ...
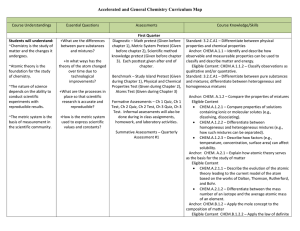
Chemistry Curriculum Map - Belle Vernon Area School District
... Standard: 3.1.C.A2 – Describe how changes in energy affect the rate of chemical reactions. Standard: 3.2.C.A1 – Explain the relationship of an elements position on the periodic table to its atomic number, ionization energy, electro-negativity, atomic size, and classification of elements. Anchor: CHE ...
... Standard: 3.1.C.A2 – Describe how changes in energy affect the rate of chemical reactions. Standard: 3.2.C.A1 – Explain the relationship of an elements position on the periodic table to its atomic number, ionization energy, electro-negativity, atomic size, and classification of elements. Anchor: CHE ...
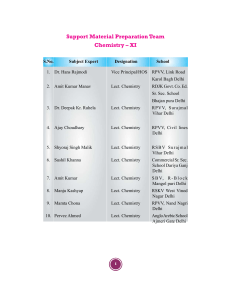
Support Material
... Percentage Composition, Empirical and Molecular Formula 32. Write the relationship between empirical formula and molecular formula. 33. Which is more informative ? Empirical formula or Molecular formula. 34. A subtance has molecular formula C6H12O6. What is its empirical formula. 35. Empirical formu ...
... Percentage Composition, Empirical and Molecular Formula 32. Write the relationship between empirical formula and molecular formula. 33. Which is more informative ? Empirical formula or Molecular formula. 34. A subtance has molecular formula C6H12O6. What is its empirical formula. 35. Empirical formu ...
Vibrationally enhanced tunneling as a mechanism for enzymatic hydrogen transfer
... or three initial states of motion along the C-H stretch that can contribute, this quantum approach seems appropriate. Even if excited states of the C-H bending modes of the transferred hydrogen are included, the number of terms in the average would be manageable. Unfortunately, calculating rate cons ...
... or three initial states of motion along the C-H stretch that can contribute, this quantum approach seems appropriate. Even if excited states of the C-H bending modes of the transferred hydrogen are included, the number of terms in the average would be manageable. Unfortunately, calculating rate cons ...
chemistry writing team
... Pauli’s Exclusion Principle : ‘‘No two electrons in an atom can have the same set of four quantum numbers.’’ Two electrons can have same values for n, l and ml provided their spins are opposite (ms is different). An orbital can have at the most two electrons if they have opporite spins. Hund’s Rule ...
... Pauli’s Exclusion Principle : ‘‘No two electrons in an atom can have the same set of four quantum numbers.’’ Two electrons can have same values for n, l and ml provided their spins are opposite (ms is different). An orbital can have at the most two electrons if they have opporite spins. Hund’s Rule ...
chemistry
... Many substances freely used these days are not available from natural sources, but this distinction is not at all useful for chemists, because it tells us little or nothing about the properties of the substance. Many natural substances can be man-made and samples from each source are absolutely iden ...
... Many substances freely used these days are not available from natural sources, but this distinction is not at all useful for chemists, because it tells us little or nothing about the properties of the substance. Many natural substances can be man-made and samples from each source are absolutely iden ...
The quantum states of muons in fluorides
... • Parameterizing the poten=al and solving for the muon wavefunc=on allows an accurate quantum correc=on of the hyperfine couplings. • However, it requires considerable effort, e.g. the tetrahedral wavefunc=on co ...
... • Parameterizing the poten=al and solving for the muon wavefunc=on allows an accurate quantum correc=on of the hyperfine couplings. • However, it requires considerable effort, e.g. the tetrahedral wavefunc=on co ...
Quantum Chaos and Quantum Computers
... operability. The time scales for development of quantum chaos and ergodicity are determined. In spite the fact that this phenomenon is rather dangerous for quantum computing it is shown that the quantum chaos border for inter-qubit coupling is exponentially larger than the energy level spacing betwe ...
... operability. The time scales for development of quantum chaos and ergodicity are determined. In spite the fact that this phenomenon is rather dangerous for quantum computing it is shown that the quantum chaos border for inter-qubit coupling is exponentially larger than the energy level spacing betwe ...
Simulations of prompt many-body ionization in a frozen Rydberg gas Robicheaux
... We did not perform a quantum calculation because it is beyond current capabilities, but it is important to have an idea of whether the classical results should be trusted. There is an important difference between classical and quantum calculations of ionization for a pair of Rydberg atoms. In the qu ...
... We did not perform a quantum calculation because it is beyond current capabilities, but it is important to have an idea of whether the classical results should be trusted. There is an important difference between classical and quantum calculations of ionization for a pair of Rydberg atoms. In the qu ...
Shell Model Approach to Nuclear Reactions
... • Based on the Rigged Hilbert Space formulation of quantum mechanics • Contour is identified and discretized for each partial wave • Many-body Slater determinants are built out of resonant and scattering states • GSM Hamiltonian matrix is computed (using external complex scaling) and Lanczos-diagona ...
... • Based on the Rigged Hilbert Space formulation of quantum mechanics • Contour is identified and discretized for each partial wave • Many-body Slater determinants are built out of resonant and scattering states • GSM Hamiltonian matrix is computed (using external complex scaling) and Lanczos-diagona ...
Full-Text PDF
... also needed. All of these problems can be solved by computational techniques based on the principles of quantum mechanics. The aim of this review is to introduce molecular modeling methods briefly and emphasize the use of these methods in TiO2 photocatalysis. The methods used for obtaining informati ...
... also needed. All of these problems can be solved by computational techniques based on the principles of quantum mechanics. The aim of this review is to introduce molecular modeling methods briefly and emphasize the use of these methods in TiO2 photocatalysis. The methods used for obtaining informati ...
Hyperradiance-a new paradigm in 60 years of superradiance
... these schemes have very low success probability. Deterministic entanglement has been produced with about a dozen qubits in the form of W -states [12, 13], but we are still far away from the realization of W -states for an arbitrary number of qubits. Calculations show that these states, which can be ...
... these schemes have very low success probability. Deterministic entanglement has been produced with about a dozen qubits in the form of W -states [12, 13], but we are still far away from the realization of W -states for an arbitrary number of qubits. Calculations show that these states, which can be ...
Manifestation and Origin of the Isotope Effect
... and a single orbital electron. Later, in 1920, the radii of a few heavy nuclei were measured by Chadwick and were found to be of the order of 10−14 m., much smaller than the order of 10−10 m for atomic radii (for details, see e.g. [7-12]). The building blocks of nuclei are neutrons and protons, two ...
... and a single orbital electron. Later, in 1920, the radii of a few heavy nuclei were measured by Chadwick and were found to be of the order of 10−14 m., much smaller than the order of 10−10 m for atomic radii (for details, see e.g. [7-12]). The building blocks of nuclei are neutrons and protons, two ...
Tailoring Rydberg interactions via F\" orster resonances: state
... Ensembles of ultracold Rydberg atoms have proven to be a powerful tool for producing novel interacting quantum systems with wide-ranging applications from quantum information processing to quantum simulation [1]. The underlying reason turning Rydberg gases into such a rich system is the strong dipol ...
... Ensembles of ultracold Rydberg atoms have proven to be a powerful tool for producing novel interacting quantum systems with wide-ranging applications from quantum information processing to quantum simulation [1]. The underlying reason turning Rydberg gases into such a rich system is the strong dipol ...
elements of chemistry unit
... Once the number and types of shared electrons has been determined, assign each shared electron to the more electronegative element. ELECTRONEGATIVITY An element’s ability to attract electrons is its electronegativity. In general, the halogens and group 16 atoms have the highest electronegativity val ...
... Once the number and types of shared electrons has been determined, assign each shared electron to the more electronegative element. ELECTRONEGATIVITY An element’s ability to attract electrons is its electronegativity. In general, the halogens and group 16 atoms have the highest electronegativity val ...
- Kendriya Vidyalaya No. 2 Raipur
... should contain equal number of molecules. Dalton's Atomic Theory All substances are made up of tiny, indivisible particles called atoms. Atoms of the same element are identical in shape, size, mass and otherproperties. Atoms of different elements are different in all respects. Atom is the smallest u ...
... should contain equal number of molecules. Dalton's Atomic Theory All substances are made up of tiny, indivisible particles called atoms. Atoms of the same element are identical in shape, size, mass and otherproperties. Atoms of different elements are different in all respects. Atom is the smallest u ...
Bohr model
In atomic physics, the Rutherford–Bohr model or Bohr model, introduced by Niels Bohr in 1913, depicts the atom as a small, positively charged nucleus surrounded by electrons that travel in circular orbits around the nucleus—similar in structure to the solar system, but with attraction provided by electrostatic forces rather than gravity. After the cubic model (1902), the plum-pudding model (1904), the Saturnian model (1904), and the Rutherford model (1911) came the Rutherford–Bohr model or just Bohr model for short (1913). The improvement to the Rutherford model is mostly a quantum physical interpretation of it. The Bohr model has been superseded, but the quantum theory remains sound.The model's key success lay in explaining the Rydberg formula for the spectral emission lines of atomic hydrogen. While the Rydberg formula had been known experimentally, it did not gain a theoretical underpinning until the Bohr model was introduced. Not only did the Bohr model explain the reason for the structure of the Rydberg formula, it also provided a justification for its empirical results in terms of fundamental physical constants.The Bohr model is a relatively primitive model of the hydrogen atom, compared to the valence shell atom. As a theory, it can be derived as a first-order approximation of the hydrogen atom using the broader and much more accurate quantum mechanics and thus may be considered to be an obsolete scientific theory. However, because of its simplicity, and its correct results for selected systems (see below for application), the Bohr model is still commonly taught to introduce students to quantum mechanics or energy level diagrams before moving on to the more accurate, but more complex, valence shell atom. A related model was originally proposed by Arthur Erich Haas in 1910, but was rejected. The quantum theory of the period between Planck's discovery of the quantum (1900) and the advent of a full-blown quantum mechanics (1925) is often referred to as the old quantum theory.