Lecture Notes 01 (continued): Transport of EM power down a long wire carrying a steady/DC current
... So how is the electrical/EM power transported {by the copper wires} from the power source (battery) to the resistor??? The “short” answer is that the EM power is transported from the battery to the resistor by the electromagnetic field(s) associated with the steady current I flowing through the circ ...
... So how is the electrical/EM power transported {by the copper wires} from the power source (battery) to the resistor??? The “short” answer is that the EM power is transported from the battery to the resistor by the electromagnetic field(s) associated with the steady current I flowing through the circ ...
Spin-orbit coupling effects in two
... [13],[14]. In coupled dots, the two-qubit quantum gates are realized by manipulating the exchange coupling between the electrons, which originates in the Coulomb interaction and the Pauli principle [8],[15]. How is the exchange coupling modified by the presence of the spin-orbit coupling? In general, ...
... [13],[14]. In coupled dots, the two-qubit quantum gates are realized by manipulating the exchange coupling between the electrons, which originates in the Coulomb interaction and the Pauli principle [8],[15]. How is the exchange coupling modified by the presence of the spin-orbit coupling? In general, ...
Chapter 5 ppt
... Theoretical yield: the maximum amount of product that would be formed from a particular reaction in an ideal world Actual yield: the amount of product formed from a particular reaction in the real world (usually less than the theoretical yield) ...
... Theoretical yield: the maximum amount of product that would be formed from a particular reaction in an ideal world Actual yield: the amount of product formed from a particular reaction in the real world (usually less than the theoretical yield) ...
Introduction to Chemistry
... count their teeth. Had he done so, he would have found that men and women have exactly the same number of teeth. In terms of physical science, Aristotle thought about dropping two balls of exactly the same size and shape but of different masses to see which one would strike the ground first. In his ...
... count their teeth. Had he done so, he would have found that men and women have exactly the same number of teeth. In terms of physical science, Aristotle thought about dropping two balls of exactly the same size and shape but of different masses to see which one would strike the ground first. In his ...
Chemical Bonding
... You will recall, from Chapter 1, that atomic theory describes electrons moving about the nucleus of the atom in energy levels, and that the electrons in the outermost energy level are called the valence electrons. It is the valence electrons of an atom that form chemical bonds. According to atomic t ...
... You will recall, from Chapter 1, that atomic theory describes electrons moving about the nucleus of the atom in energy levels, and that the electrons in the outermost energy level are called the valence electrons. It is the valence electrons of an atom that form chemical bonds. According to atomic t ...
Hubbard and Kondo lattice models in two dimensions: A QMC study
... that (i) the local screening of impurity spins determines the low-energy behavior of the spectral function and (ii) one cannot deform continuously the spectral function of the half-filled Hubbard model at J = 0 to that of the Kondo insulator at J > Jc . Our results are based on both T = 0 Quantum Mo ...
... that (i) the local screening of impurity spins determines the low-energy behavior of the spectral function and (ii) one cannot deform continuously the spectral function of the half-filled Hubbard model at J = 0 to that of the Kondo insulator at J > Jc . Our results are based on both T = 0 Quantum Mo ...
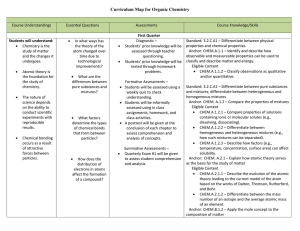
Organic Chemistry Curriculum Map - Belle Vernon Area School District
... Standard: 3.2.C.A4 – Predict how combinations of substances can result in physical and/or chemical changes. Anchor: CHEM.B.1.1 – Explain how the mole is a fundamental unit of chemistry. Eligible Content CHEM.B.1.1.1 – Apply the mole concept to representative particles (e.g., counting, determining ...
... Standard: 3.2.C.A4 – Predict how combinations of substances can result in physical and/or chemical changes. Anchor: CHEM.B.1.1 – Explain how the mole is a fundamental unit of chemistry. Eligible Content CHEM.B.1.1.1 – Apply the mole concept to representative particles (e.g., counting, determining ...
Charge Transport in Semiconductors Contents
... of any kind. Let us follow the motion of an electron in the valence band in the presence of a force (due to electric field), pointing to the right (+k) (see Figure 3). A force imparts momentum to the electron, moving it into the next nearest state on the right, the electron in the next nearest state ...
... of any kind. Let us follow the motion of an electron in the valence band in the presence of a force (due to electric field), pointing to the right (+k) (see Figure 3). A force imparts momentum to the electron, moving it into the next nearest state on the right, the electron in the next nearest state ...
Photocatalysis on TiOn Surfaces: Principles, Mechanisms, and
... exchange require orbital overlap between the interacting centers. When both processes are thermodynamically allowed, electron transfer predominates since the electron exchange process requires simultaneous overlap of two orbital pairs, whereas only one such overlap is necessary for electron t r a n ...
... exchange require orbital overlap between the interacting centers. When both processes are thermodynamically allowed, electron transfer predominates since the electron exchange process requires simultaneous overlap of two orbital pairs, whereas only one such overlap is necessary for electron t r a n ...
orange review book_2014_key
... compounds is that both (1) are heterogeneous (2) consist of two or more substances (3) are homogeneous (4) are heterogeneous 20. A dilute, aqueous potassium nitrate solution is best classified as a (1) homogeneous compound (2) homogeneous mixture (3) heterogeneous compound (4) heterogeneou ...
... compounds is that both (1) are heterogeneous (2) consist of two or more substances (3) are homogeneous (4) are heterogeneous 20. A dilute, aqueous potassium nitrate solution is best classified as a (1) homogeneous compound (2) homogeneous mixture (3) heterogeneous compound (4) heterogeneou ...
High Rydberg states of DABCO: Spectroscopy, ionization potential
... ~MATI! by Johnson and co-workers.2 Both techniques can yield high-resolution spectra3 of molecular ions by detecting precursor molecular Rydberg states through pulsed field ionization. The unexpected stability of these molecular Rydberg states as seen in ZEKE and MATI experiments4,5,6 has led to a n ...
... ~MATI! by Johnson and co-workers.2 Both techniques can yield high-resolution spectra3 of molecular ions by detecting precursor molecular Rydberg states through pulsed field ionization. The unexpected stability of these molecular Rydberg states as seen in ZEKE and MATI experiments4,5,6 has led to a n ...
Nuclear Spins in Quantum Dots
... (1.5) include contributions from different orbitals, so any inter-orbital scattering mechanism will lead to scattering between spin states. The magnitude of the spin scattering is determined by α and the orbital scattering mechanism. There are a number of possible admixture mechanisms in GaAs. The m ...
... (1.5) include contributions from different orbitals, so any inter-orbital scattering mechanism will lead to scattering between spin states. The magnitude of the spin scattering is determined by α and the orbital scattering mechanism. There are a number of possible admixture mechanisms in GaAs. The m ...
An experimental and theoretical guide to strongly interacting
... by the combination with ultracold atomic gases. The availability of commercial high power diode laser systems allow for efficient excitation into electronically highly excited states with excellent frequency resolution. Here the major motivation behind many experiments is to make use of the strong i ...
... by the combination with ultracold atomic gases. The availability of commercial high power diode laser systems allow for efficient excitation into electronically highly excited states with excellent frequency resolution. Here the major motivation behind many experiments is to make use of the strong i ...
3. Angular Momentum States.
... r, from the axis of rotation, and in the other case the center of mass is at a distance, r, from the axis of rotation. Quantum Electron Spin Angular Momentum The results of classical angular momentum provide a clear physical model for representation of orbital and spin angular momentum in terms of ...
... r, from the axis of rotation, and in the other case the center of mass is at a distance, r, from the axis of rotation. Quantum Electron Spin Angular Momentum The results of classical angular momentum provide a clear physical model for representation of orbital and spin angular momentum in terms of ...
Smooth Scaling of Valence Electronic Properties in Fullerenes: From
... with those of the other nonicosahedral fullerenes. The linear fits that define the icosahedral and nonicosahedral capacitance scaling lines both are very strong, as seen from the large values of R2 for each displayed in Fig. 1. Additionally, it is seen there that the icosahedral and nonicosahedral s ...
... with those of the other nonicosahedral fullerenes. The linear fits that define the icosahedral and nonicosahedral capacitance scaling lines both are very strong, as seen from the large values of R2 for each displayed in Fig. 1. Additionally, it is seen there that the icosahedral and nonicosahedral s ...
Characterizing Molecular Interactions in Chemical Systems
... Molecular interactions govern the structure of chemical systems by establishing attractive and repulsive balances in-between atoms. These interactions vary in strength and type. Here, we provide a brief characterization of them, and highlight some of their properties. We refer the reader to [47] for ...
... Molecular interactions govern the structure of chemical systems by establishing attractive and repulsive balances in-between atoms. These interactions vary in strength and type. Here, we provide a brief characterization of them, and highlight some of their properties. We refer the reader to [47] for ...
International Journal of Quantum Chemistry 114:1041
... In such a situation it represents a conceptual interest how the classical picture of minimal basis core and valence orbitals can be recovered by an appropriate a posteriori analysis of wave functions – the present paper is devoted to this problem. For sake of simplicity, we shall concentrate on clo ...
... In such a situation it represents a conceptual interest how the classical picture of minimal basis core and valence orbitals can be recovered by an appropriate a posteriori analysis of wave functions – the present paper is devoted to this problem. For sake of simplicity, we shall concentrate on clo ...
Personal Tutor - Macmillan Learning
... Example: Convert 1456 g to kilograms. In Figure 2 the meaning of kilo- tells that 1 kilogram would be equal to 103 grams. Equivalance: 1 kilogram (kg) = 1 X 10 3 grams (g) The equivalence between kilograms and grams can be used to create a conversion factor. A conversion factor, often called a unit ...
... Example: Convert 1456 g to kilograms. In Figure 2 the meaning of kilo- tells that 1 kilogram would be equal to 103 grams. Equivalance: 1 kilogram (kg) = 1 X 10 3 grams (g) The equivalence between kilograms and grams can be used to create a conversion factor. A conversion factor, often called a unit ...
Bohr model
In atomic physics, the Rutherford–Bohr model or Bohr model, introduced by Niels Bohr in 1913, depicts the atom as a small, positively charged nucleus surrounded by electrons that travel in circular orbits around the nucleus—similar in structure to the solar system, but with attraction provided by electrostatic forces rather than gravity. After the cubic model (1902), the plum-pudding model (1904), the Saturnian model (1904), and the Rutherford model (1911) came the Rutherford–Bohr model or just Bohr model for short (1913). The improvement to the Rutherford model is mostly a quantum physical interpretation of it. The Bohr model has been superseded, but the quantum theory remains sound.The model's key success lay in explaining the Rydberg formula for the spectral emission lines of atomic hydrogen. While the Rydberg formula had been known experimentally, it did not gain a theoretical underpinning until the Bohr model was introduced. Not only did the Bohr model explain the reason for the structure of the Rydberg formula, it also provided a justification for its empirical results in terms of fundamental physical constants.The Bohr model is a relatively primitive model of the hydrogen atom, compared to the valence shell atom. As a theory, it can be derived as a first-order approximation of the hydrogen atom using the broader and much more accurate quantum mechanics and thus may be considered to be an obsolete scientific theory. However, because of its simplicity, and its correct results for selected systems (see below for application), the Bohr model is still commonly taught to introduce students to quantum mechanics or energy level diagrams before moving on to the more accurate, but more complex, valence shell atom. A related model was originally proposed by Arthur Erich Haas in 1910, but was rejected. The quantum theory of the period between Planck's discovery of the quantum (1900) and the advent of a full-blown quantum mechanics (1925) is often referred to as the old quantum theory.