INTRODUCTION TO ELEMENTARY PARTICLE PHYSICS
... The world of everyday life, of course, is governed by classical mechanics. But for objects that travel very fast (at speeds comparable to c), the classical rules are modified by special relativity, and for objects that are very small (comparable to the size of atoms, roughly speaking), classical mec ...
... The world of everyday life, of course, is governed by classical mechanics. But for objects that travel very fast (at speeds comparable to c), the classical rules are modified by special relativity, and for objects that are very small (comparable to the size of atoms, roughly speaking), classical mec ...
Word Format
... Many lines were not seen. This indicated that there were selection rules that determined what lines were present. ...
... Many lines were not seen. This indicated that there were selection rules that determined what lines were present. ...
Modern Physics
... matter exhibits the same “duality” that light exhibits Perhaps all matter has both characteristics as well ...
... matter exhibits the same “duality” that light exhibits Perhaps all matter has both characteristics as well ...
Book Reviews
... ª Outcome Independenceº (Shimony) on the other, has played a central role in philosophical discussions of quantum-mechanical nonlocality. In presenting his gloss on Bell’ s theorem in the way that he does, Norris implicitly assumes the distinction to be a spurious distinction, and leaves himself no ...
... ª Outcome Independenceº (Shimony) on the other, has played a central role in philosophical discussions of quantum-mechanical nonlocality. In presenting his gloss on Bell’ s theorem in the way that he does, Norris implicitly assumes the distinction to be a spurious distinction, and leaves himself no ...
Isra University Faculty of Arts and science Course Calendar 2016
... (dual and quad-polar), Hamiltonian corn, the elements of the matrix and the principle of symmetry, the equations of motion for transitions binaries electrodes and magnetic, vibration operations, the behavior of the case for the transition of dual-electrode, properties, transit behavior for the trans ...
... (dual and quad-polar), Hamiltonian corn, the elements of the matrix and the principle of symmetry, the equations of motion for transitions binaries electrodes and magnetic, vibration operations, the behavior of the case for the transition of dual-electrode, properties, transit behavior for the trans ...
Incompatible results of quantum measurements
... an o p e r a t o r A d e p e n d s only on A and on the state o f the q u a n t u m system being measured (here, the word " s t a t e " includes not only the wave function ~u, but also any h i d d e n variables that theorists m a y invent). In particular, that result does not d e p e n d on the choi ...
... an o p e r a t o r A d e p e n d s only on A and on the state o f the q u a n t u m system being measured (here, the word " s t a t e " includes not only the wave function ~u, but also any h i d d e n variables that theorists m a y invent). In particular, that result does not d e p e n d on the choi ...
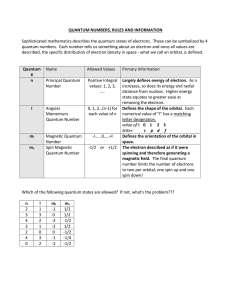
Quantum Number Table
... increases, so does its energy and radial distance from nucleus. Higher energy state equates to greater ease at removing the electron. Defines the shape of the orbital. Each numerical value of "l" has a matching letter designation. value of l: 0 1 2 3 letter: s p d f Defines the orientation of the or ...
... increases, so does its energy and radial distance from nucleus. Higher energy state equates to greater ease at removing the electron. Defines the shape of the orbital. Each numerical value of "l" has a matching letter designation. value of l: 0 1 2 3 letter: s p d f Defines the orientation of the or ...
CHEM 532 Physical Chemistry II (Quantum Chemistry) Fall 2013
... the variation method, time independent perturbation theory, degenerate perturbation theory, the anharmonic oscillator VIII. The Helium atom electron spin, ground state of He, excited electronic states of He, spin eigenfunctions of He IX. Many-electron wavefunctions indistinguishable particles, the P ...
... the variation method, time independent perturbation theory, degenerate perturbation theory, the anharmonic oscillator VIII. The Helium atom electron spin, ground state of He, excited electronic states of He, spin eigenfunctions of He IX. Many-electron wavefunctions indistinguishable particles, the P ...
Syllabus
... will review those aspects of quantum mechanics that play the most important role in this understanding. This includes the structure of simple two-particle species, the properties of bound and continuum states, the quantum theory of many-electron species, the quantum theory of scattering and transiti ...
... will review those aspects of quantum mechanics that play the most important role in this understanding. This includes the structure of simple two-particle species, the properties of bound and continuum states, the quantum theory of many-electron species, the quantum theory of scattering and transiti ...
Course Syllabus
... Note: I always recommend the Feynman Lectures on Physics, Vol. 3, as a most beautiful, illuminating source of Quantum Mechanics at an “elementary” level. Volume 3 of the Feynman Lectures represents a famous experiment at teaching Quantum Mechanics “correctly” at the sophomore level. In addition, the ...
... Note: I always recommend the Feynman Lectures on Physics, Vol. 3, as a most beautiful, illuminating source of Quantum Mechanics at an “elementary” level. Volume 3 of the Feynman Lectures represents a famous experiment at teaching Quantum Mechanics “correctly” at the sophomore level. In addition, the ...
II. Units of Measurement
... radiation first (IR – heat) and then glow red, and then yellow, and then white? ...
... radiation first (IR – heat) and then glow red, and then yellow, and then white? ...
Sec 4-1 Chapter 4 Notes
... In 1913, Niels Bohr came to work with Rutherford. Rutherford knew that there were errors in his model. Bohr then proposed the atom’s e- was linked to the photon emissions. He included that the ecan circle the nucleus in allowed paths or orbits. E1 is called ground state ...
... In 1913, Niels Bohr came to work with Rutherford. Rutherford knew that there were errors in his model. Bohr then proposed the atom’s e- was linked to the photon emissions. He included that the ecan circle the nucleus in allowed paths or orbits. E1 is called ground state ...
Pauli Exclusion Principle Quiz
... Pauli Exclusion Principle Quiz 1. The location of any electron in an atom can be described by ____ unique quantum numbers. ...
... Pauli Exclusion Principle Quiz 1. The location of any electron in an atom can be described by ____ unique quantum numbers. ...
Contemporary Quantum Optics
... A. Aspect, P. Grangier, and G. Roger, Phys. Rev. Lett. 49, 91 (1982). ...
... A. Aspect, P. Grangier, and G. Roger, Phys. Rev. Lett. 49, 91 (1982). ...
PHYS3111, 3d year Quantum Mechanics General Info
... For the first tutorial I recommend problems 1,2,4,9,11. The problem 10 is in assignment, so it is excluded from the tutorial. For the second tutorial I recommend problems 13,14,17,19. Problems 21,22 are in assignment, so they are excluded from the tutorial. For the third tutorial I recommend problem ...
... For the first tutorial I recommend problems 1,2,4,9,11. The problem 10 is in assignment, so it is excluded from the tutorial. For the second tutorial I recommend problems 13,14,17,19. Problems 21,22 are in assignment, so they are excluded from the tutorial. For the third tutorial I recommend problem ...
“SUPERPOSITION” “interference term”
... In simplest (“BCS”) theory, Cooper pairs, once formed, must automatically ...
... In simplest (“BCS”) theory, Cooper pairs, once formed, must automatically ...
From quantum to quantum computer
... (1) One of the founders of the quantum concept (2) A first, thought there must be something wrong with the quantum theory. (3) After much debate with Bohr, he finally was convinced that QM gives correct results, but it could not be the final theory. It is incomplete! ...
... (1) One of the founders of the quantum concept (2) A first, thought there must be something wrong with the quantum theory. (3) After much debate with Bohr, he finally was convinced that QM gives correct results, but it could not be the final theory. It is incomplete! ...
Presentation
... Generation of quasiclassical Bohr -like wave packets using half-cycle pulses J. J. Mestayer, B. Wyker, F. B. Dunning, C. O. Reinhold, S. Yoshida, and J. Burgdörfer We demonstrate the experimental realization of Bohr -like atoms by applying a pulsed unidirectional field, termed a half-cycle pulse (HC ...
... Generation of quasiclassical Bohr -like wave packets using half-cycle pulses J. J. Mestayer, B. Wyker, F. B. Dunning, C. O. Reinhold, S. Yoshida, and J. Burgdörfer We demonstrate the experimental realization of Bohr -like atoms by applying a pulsed unidirectional field, termed a half-cycle pulse (HC ...