* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Infectivity of blood Adham
Survey
Document related concepts
Transmission (medicine) wikipedia , lookup
Social history of viruses wikipedia , lookup
Hospital-acquired infection wikipedia , lookup
Plant virus wikipedia , lookup
Viral phylodynamics wikipedia , lookup
Neonatal infection wikipedia , lookup
Henipavirus wikipedia , lookup
History of virology wikipedia , lookup
Virus quantification wikipedia , lookup
Microbicides for sexually transmitted diseases wikipedia , lookup
West Nile fever wikipedia , lookup
Human cytomegalovirus wikipedia , lookup
Infection control wikipedia , lookup
Diagnosis of HIV/AIDS wikipedia , lookup
Canine parvovirus wikipedia , lookup
Transcript
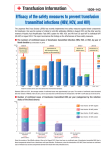
Infectivity of blood By Dr. Adham Abdulmonem Saleh M.B.B.Ch. – M.Sc in Anesthesia Assistant lecturer of Anesthesia & ICU Ain Shams University With the introduction of transfusion-induced AIDS, the infectivity by homologous blood transfusion has received renewed attention. In fact, for many years, blood banks use one or two tests (i.e., syphilis and hepatitis B surface antigen) to screen blood. In recent years, many more tests have been added. Overall, blood is probably safer than it has been for years. Infectious complications Viruses HCV HBV HIV HTLV Cytomegalovirus Epstein-Barr virus Parvovirus B19 West Nile v CJD Spirochetes Treponema pallidum Borrelia burgdorferi Parasites Plasmodia Trypanosoma cruzi Toxoplasma gondii Leishmania donovani Bacteria Staphylococcus Salmonella Yersinia enterocolitica Risk of transmitting infection to recipients has been dramatically reduced in the past decades, due to: Improved donor selection. Sensitive serologic screening assays. Application of viral inactivation procedures during manufacturing of plasma products. 1/1 00 1/1,0 00 1/10,0 00 1/100,0 00 1/1,000,0 00 Adapted from Transfusion 2000; 40:143-159 I- viral: Major sources of remaining risk are: Window period donation Viral variants not detect by current assays Immunosilent donor Laboratory testing error The greatest threat to the safety of blood supply is the donation by seronegative donors during the infectious window period Window period donation accounts for 90% or more of the residual risk (Report of the Interorganization Task Force on NAT Testing of Blood Donors, 2000) Period precedes the development of antibodies during the initial infection Eclipse phase of the window period is the very initial phase after exposure when virus replication is restricted to tissue sites and there is no detectable viraemia Infectious phase of window period is after eclipse and before seroconversion Events in early viral infection WP1 Exposure WP2 Viremia Eclipse Serological Detection Infectivity Estimates for WPs from Exposure to Seroconversion Following Discrete Parenteral Exposures Virus Source N Point Est HIV Needlestick 51 46 days 95% CI (range) 10 - 190 HCV Transfusion 46 71 days 33 - 128 HBV Transfusion 7/15 59 days (37 - 87) 51 days (36 - 72) HTLV Transfusion 24 Immunosilent carriers: chronic antibody negative carriers Persistent viremia in absence of detectable seroconversion Case reports for HIV, HCV, and HBV Recipient infection via transfusion has been documented for HCV Appears to be rare II. Bacterial Contamination unlikely in products stored for > 72 hours at 1-6 0 C gram –ve, gram +ve bacteria most frequent – Yersinia enterocolitica endotoxin Common in Platelets stored at room temperature for 5 days, with infection rate of 0.25% III. Protozoal Trypanosoma cruzi (Chaga’s disease) Malaria Toxoplasmosis Leishmaniasis Serological Testing for Infectious markers HIV – Ag Anti – HIV HBsAg Anti – HCV Test for syphilis Future Screening other virus for specific blood products for specific patient group, eg. screening Parvovirus B19 for Anti-D Ig Screening for new transfusion-transmitted viruses