* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Antipsychotics in the Pipeline
Discovery and development of direct Xa inhibitors wikipedia , lookup
Discovery and development of integrase inhibitors wikipedia , lookup
Discovery and development of proton pump inhibitors wikipedia , lookup
Polysubstance dependence wikipedia , lookup
Orphan drug wikipedia , lookup
Compounding wikipedia , lookup
Neuropsychopharmacology wikipedia , lookup
Psychopharmacology wikipedia , lookup
Pharmaceutical industry wikipedia , lookup
Drug discovery wikipedia , lookup
Prescription costs wikipedia , lookup
Prescription drug prices in the United States wikipedia , lookup
Drug design wikipedia , lookup
Pharmacognosy wikipedia , lookup
Pharmacokinetics wikipedia , lookup
Neuropharmacology wikipedia , lookup
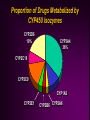
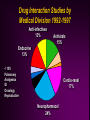
Drug Interactions Cara L. Alfaro, Pharm.D. Clinical Reviewer Division of Neuropharmacological Drug Products Food and Drug Administration This presentation was prepared by Dr. Alfaro in her private capacity. No official support or endorsement by the FDA is intended or should be inferred. Incidence of Drug-Drug Interactions True incidence difficult to determine Data for drug-related hospital admissions do not separate out drug interactions, focus on ADRs Lack of availability of comprehensive and easy to access databases Difficulty in assessing OTC and herbal drug therapy use Difficulty in determining contribution of drug interaction in morbidity of medically complicated patients Drug Interactions Pharmacodynamic Related to the drug’s effects in the body Receptor site occupancy Pharmacokinetic Related to the body’s effects on the drug Absorption, distribution, metabolism, elimination, Pharmacodynamic Interactions Pharmacodynamic Drug Interactions Additive, synergistic, or antagonistic effects from co-administration of two or more drugs Synergistic actions of antibiotics Overlapping toxicities - ethanol & benzodiazepines Antagonistic effects - anticholinergic medications (oxybutinin or amitriptyline w/ acetylcholinesterase inhibitors) Pharmacokinetic Drug Interactions Alteration in absorption Protein binding effects Alteration in elimination Changes in drug metabolism Pharmacokinetic Absorption Interactions Alterations in Absorption Administration with food For many drugs, decrease rate of absorption but not extent Indinavir - rapidly absorbed in fasted state, AUC and Cmax decreased by ~80% with high calorie/fat/protein meal Saquinavir - administration with high fat meal increases AUC by ~570% for this low F drug (4%) Patient issues?? Alterations in Absorption Chelation Irreversible binding of drugs in the GI tract Tetracyclines, quinolone antibiotics - ferrous sulfate (Fe+2), antacids (Al+3, Ca+2, Mg+2), dairy products (Ca+2) Usually separating administration of chelating drugs by 2+ hours decreases interaction effect Dose tetracycline 1 hour before or 2 hours after dairy products Thyroxine and Ferrous Sulfate Variability of interaction 100 Serum TSH (mU/L) TSH on stable thyroxine dose before and after 12 weeks co-ingestion with 300 mg FeSO4 TSH 1.6 ± 0.4 before TSH 5.4 ± 2.8 after 9/14 had clinical symptoms of hypothyroidism Thyroxine + FeSO4 invitro - complexation Mgmt?? 10 1 0.1 0 12 Study Week Campbell NRC et al. Ann Intern Med 1992;/117:1010-1013 Alterations in Absorption Alteration in GI motility Increased motility - cisapride (R.I.P.), metoclopramide Decreased motility - narcotics Altering GI tract pH Increase in GI pH (antacids, omeprazole, cimetidine) may decrease absorption of drugs which require acidic pH for optimal absorption such as ketoconazole and itraconazole Ketoconazole Cp (mcg/ml) Ketoconazole Interactions pH-dependent absorption 8 7 6 5 4 3 2 1 0 Keto K + Sucral K + Ranit 0 0.5 1.5 2.5 4 6 Hours Piscitelli S et al. Antimicrob Agents Chemother 1991;35:1765-1771 12 Pharmacokinetic Protein Binding Displacement Interactions Protein Binding Interactions “…the overall clinical importance of plasma protein binding displacement interactions continues to be overstated…” “Despite the theoretical and experimental data to the contrary, the concept that plasma protein binding displacement is a common cause of clinically significant interactions may still be widely taught in some medical schools, often appears in textbooks and is accepted by many in the medical community and by drug regulators.” Sansom LN & Evans AM. Drug Safety 1995;12:227-233. Rolan PE. Br J Clin Pharmacol 1994;37:125-128. Protein Binding Interactions Competition between drugs for protein or tissue binding sites Increase in free (unbound) concentration may lead to enhanced pharmacological effect Many interactions previously thought to be PB interactions, were found to be primarily metabolism interactions Warfarin - sulfamethoxazole (partially metabolism interaction) PB interactions are not usually clinically significant Protein Binding Interactions Restrictively cleared drugs Small fraction of drug extracted during single passage through the eliminating organ CL is directly proportional to fu Increase in fu leads to proportional increase in CL and decrease in Css No change in Clu, Cssu will return to predisplacement value after transient increase phenytoin and valproic acid; decrease in phenytoin Css and Cu unchanged Principles of Clinical Pharmacology, pg 64 Protein Binding Interactions Nonrestrictively cleared drugs Eliminating organ removing most of the drug being presented to it, including the fraction bound to plasma proteins Increase in fu will not lead to a proportional increase in CL Protein Binding Interactions Drugs for which pure plasma protein binding displacement interactions will lead to sustained changes in Cssu Extensively bound to plasma proteins Nonrestrictively cleared Administered by non-oral route alfentanil, buprenorphine, lidocaine, verapamil Very few orally administered drugs exhibiting properties of extensive plasma protein binding, high hepatic first-pass extraction and narrow therapeutic index Pharmacokinetic Metabolism Interactions Drug Metabolism Interactions Drug metabolism inhibited or enhanced by coadministration of other drugs CYP 450 system has been the most extensively studied CYP3A4, CYP2D6, CYP1A2, CYP2B6, CYP2C9, CYP2C19 and others Phase 2 metabolic interactions (glucuronidation, etc.) occur, research in this area is increasing CYP 450 Substrates Metabolism by a single isozyme (predominantly) Few examples of clinically used drugs Desipramine/CYP2D6 Examples of drugs used primarily in research on drug interaction potential Debrisoquin, sparteine, dextromethorphan, mephenytoin Metabolism by multiple isozymes Most drugs metabolized by more than one isozyme Imipramine: CYP2D6, CYP1A2, CYP3A4, CYP2C19 If co-administered with CYP450 inhibitor, some isozymes may “pick up slack” for inhibited isozyme Proportion of Drugs Metabolized by CYP450 Isozymes CYP2D6 19% CYP3A4 36% CYP2C19 CYP2C9 CYP1A2 CYP2E1 CYP2B6 CYP2A6 CYP 450 Inhibitors Drugs can inhibit a specific CYP even though they are not metabolized by that isozyme Quinidine - most potent CYP2D6 inhibitor but metabolized primarily by CYP3A4 Drugs which are metabolized by a specific CYP may not potently inhibit that CYP Venlafaxine is metabolized by CYP3A4 but is not a potent inhibitor of CYP3A4 Determining whether a drug is a substrate or an inhibitor (or inducer) of a specific CYP are different questions Examples of CYP 450 Substrates, Inhibitors, & Inducers Substrates* Inhibitors Inducers CYP3A4 Alprazolam Lovastatin Quetiapine Clarithromycin Ritonavir Ketoconazole Rifampin Carbamazepine CYP2D6 Risperidone Desipramine Donepezil Quinidine Fluoxetine Paroxetine None identified CYP1A2 Clozapine Theophylline Caffeine Fluvoxamine Cimetidine Smoking Omeprazole Cruciferous veg *Primary metabolic pathway CYP 450 Inducers The “usual suspects” Rifampin Rifabutin Carbamazepine Phenobarbital Phenytoin CYP 450 Enzyme Induction Gradual onset and offset Onset - accumulation of inducing agent and increase in enzyme production Offset - elimination of inducing agent and decay of enzymes Results in reduction of plasma concentration of substrate drugs CYP 450 Inhibitors The “usual suspects” Cimetidine Erythromycin Ketoconazole Ritonavir Fluoxetine, paroxetine (CYP2D6) Nefazodone (CYP3A4) CYP 450 Enzyme Inhibition Usually by competitive binding to enzyme site Onset and offset dependent on the half-life and time to steady-state of the inhibitor Fluoxetine & CYP2D6 Time to maximum interaction effect dependent on time required for substrate drug to reach new steady-state Fluoxetine, Sertraline & Desipramine Interaction 4000 Desipramine AUC 3500 3000 2500 2000 Sertraline Fluoxetine 1500 1000 500 0 DMI DMI + SSRI 3 wks DMI 3 week SSRI washout Preskorn SH et al. J Clin Psychopharmacol 1994;14:90-98 Review of NDAs # NMEs increased 2-fold # DDI studies increased 4.6 fold 600 500 400 # NMEs # DDIs 300 200 100 0 87 - 91 92 - 97 Marroum PJ et al. Clin Pharmacol Ther 2000;68:280-5 Drug Interaction Studies by Medical Division 1992-1997 Anti-infectives 13% Antivirals 15% Endocrine 13% < 10% Pulmonary Analgesics GI Oncology Reproductive Cardio-renal 17% Neuropharmacol 24% NDAs - Drug Interactions Most common single agent drug interactions Cimetidine Digoxin Antacids Warfarin Propranolol Theophylline Approaches to drug interaction studies > 1995 focused on mechanism based interactions Effects of drugs on specific CYP isozymes Predicting drug interactions Investigating Drug Interaction Potential of NMEs cDNA expressed isozymes Drug probes (in vivo) - drugs with fairly specific metabolic pathways Dextromethorphan, debrisoquin - CYP2D6 Midazolam - CYP3A4 Caffeine - CYP1A2 Bupropion - CYP2B6 Tolbutamide - CYP2C9 Dextromethorphan CYP2D6 dextrorphan Drug Labeling “An in vitro enzyme inhibition study utilizing human liver microsomes showed that ziprasidone had little inhibitory effect on CYP1A2, CYP2C9, CYP2C19, CYP2D6, and CYP3A4, and thus would not likely interfere with the metabolism of drugs primarily metabolized by these enzymes. In vivo studies have revealed no effect of ziprasidone on the pharmacokinetics of dextromethorphan…” Drug Withdrawals Drug Interactions & QT Prolongation Risk Benefit Analysis Labeling changes - impact?? Terfenadine Fexofenadine (Seldane®) (Allegra®) Cisapride (Propulsid®) Astemizole (Hismanal®) + Norcisapride? Norastemizole? Terfenadine & Ketoconazole Interaction 700 600 500 QTc (ms) Terf Cp at usual doses = undetectable QT prolongation correlated to terf Cp (R2 = 0.6, p = 0.001) ~45 ng/ml = 70 to 110 ms increase in QTc 400 300 200 100 0 Baseline Terf Terf + Keto Drug Interaction Liability Risk vs. Benefit Analysis (Competitive Marketplace Decision too) Therapeutic Area Terfenadine Astemizole Mibefradil (Posicor®) Ritonavir (Norvir®) Fluoxetine (Prozac®) Antihistamines Liability DDI - QT prolongation Antihypertensive CYP3A4 Inhibition - DDIs (Calcium Channel Blocker) Competitive in class? HIV Protease Inhibitor Antidepressant CYP3A4, CYP2D6 Inhibition - DDIs CYP2D6 Inhibition - DDIs Attempted R-fluoxetine Pharmaceutical Industry Competitive in Market CYP1A2 Inhibition Fluvoxamine & Clozapine Drug Interaction N = 16, clozapine dose 202 ± 36 mg/day 600 * Cp (ng/ml) 500 400 § 300 Clozapine DM-clozapine 200 100 0 Baseline *p<0.0001 vs. baseline §p<0.05 vs. baseline + Fluvoxamine 50mg x 14 days Wetzel H et al. J Clin Psychopharmacol 1998;18:2-9 CYP2D6 Inhibition Correlation to Paroxetine Cp log Post Paroxetine DM/DP 1 0 EM -1 PM Outlier, 2D6*2 gene duplication -2 2 R = 0.718 p = 0.008 -3 0.6 0.8 1.0 1.2 1.4 1.6 1.8 log Paroxetine Cp (ng/ml) Alfaro CL et al. J Clin Pharmacol 2000;40:58-66 Herb - Drug Interactions Herb-Drug Interactions Limitations Since not regulated by FDA, safety & efficacy not required Little information available regarding drug interactions Extrapolation of data to available products difficult Independent lab tests many products (http://www.consumerlabs.com/) 6/13 SAMe preparations did not pass testing no detectable SAMe noted in one product 8/17 valerian preparations did not pass testing 4 - no detectable levels of valerenic acid 4 - 1/2 the amount claimed on the label St. John’s wort: CYP3A4 Induction Effects Indinavir Indinavir + SJW 18 Indinavir Cp (µg/ml) 16 14 12 10 8 6 4 2 0 0 0.5 1 2 3 4 5 Time Piscitelli SC et al. Lancet 2000;355:547-8 8 normal volunteers Indinavir AUC determined before and after 14 days SJW 300 mg TID Indinavir AUC decreased by 57 ± 19% in presence of SJW Garlic - Saquinavir Interaction 3500 3000 AUC (h*ng/mL) N = 10 healthy subjects Saquinavir 1200 mg TID x 3d - AUC Garlic caplets BID x ~3 weeks Repeat saquinavir AUC Discontinue garlic x 10 days Repeat saquinavir AUC 2500 2000 1500 1000 500 0 Saq Piscitelli S et al. Clin Infect Dis 2002;34:234-238 Saq + Garlic Saq Grapefruit Juice Interactions Flavinoids in grapefruit juice can inhibit gastrointestinal CYP3A4 and first pass metabolism Can increase concentrations of various CYP3A4 substrates - esp. those with low F Saquinavir AUC increases 50 - 200% Benzodiazepines Calcium channel blockers Wide variability - amount of GF juice, timing of intake and drug dosing, interpatient variability in CYP3A4 gut activity Felodipine AUC (nmol*h/L) Grapefruit Juice & Felodipine 180 160 140 120 100 80 60 40 20 0 * * * * Control 0 1 4 10 24 Hours After GF Before F Admin *Sign. Diff from Control Lundahl J et al. Eur J Clin Pharmacol 1995;49:61-67 Beneficial Drug Interactions Saquinavir & ritonavir Saquinavir poorly absorbed, TID dosing, high pill burden Combination with ritonavir results in 20-fold increase in Css Allows for BID dosing and decreased dose from 1800 mg TID to 400 mg BID Cyclosporin and ketoconazole Clozapine and fluvoxamine?? Recognizing Drug Interactions High index of suspicion Patient’s demonstrating exaggerated toxicity or drug effects Patient could also be poor metabolizer of dependent isozyme Genotyping may aid in future, but would not pick up “phenocopy” effects Patient’s demonstrating treatment failure or loss of drug effect Induction vs. absorption interactions Evaluation of Drug Interactions What is the time-course of the interaction Immediately or over a period of time Clozapine and rifampin Is it a drug class effect Cimetidine vs. ranitidine; ketoconazole vs. fluconazole Is the interaction clinically significant Therapeutic index of drugs, toxicity?, loss of efficacy? How should the interaction be managed? Drug Interaction Resources Correction to Dr. Flockhart’s website: http://medicine.iupui.edu/flockhart