* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Drug Interactions Every Health Care Provider Should Know
Survey
Document related concepts
Neuropsychopharmacology wikipedia , lookup
Orphan drug wikipedia , lookup
Polysubstance dependence wikipedia , lookup
Discovery and development of proton pump inhibitors wikipedia , lookup
Psychopharmacology wikipedia , lookup
Compounding wikipedia , lookup
List of off-label promotion pharmaceutical settlements wikipedia , lookup
Drug design wikipedia , lookup
Neuropharmacology wikipedia , lookup
Drug discovery wikipedia , lookup
Pharmacognosy wikipedia , lookup
Pharmaceutical industry wikipedia , lookup
Prescription costs wikipedia , lookup
Pharmacokinetics wikipedia , lookup
Transcript
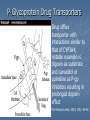
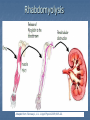
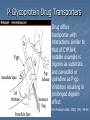
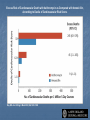
Drug drug interactions highlighting P450 Cytochromes Louis Wu, PA-C Presented at VAPAA Annual Meeting March 2, 2017 Financial Disclosures and Conflicts I have no financial disclosures nor conflicts concerning this talk. You are asking why I am here? Learning Objectives Develop a basic understanding of pharmacodynamics and pharmcokinetics as it applies to drug interactions Conceptualize the cytochrome P450 enzyme system; enzyme inhibition and enzyme induction Review potential hazards of drug interactions in older populations Be able to access drug interaction resources on the VA CPRS system as well as online such as FDA website and publications such as Medical Letter Why doesn’t your patient respond to the medication? Adherence Age of the patient Disease State Drug-drug and food-drug interactions Gender Genetics Others Langman and Dasgupta, in Pharmacogenomics in Clinical Therapeutics, 2012, pp 1-4 People react differently to drugs “One size does not fit all …” Toxic responders Non-responders Responders Patients with drug toxicity Phenotypes Patients with non-response to drug therapy Patient population with differing genotypes Patients with normal response to drug therapy What is the problem with drug-drug interactions? Precipitant drug Object (or substrate) drug Drug interaction between the two that increases or decreases the concentration of the object drug If significant enough, then the patient develops a drug toxicity or an inadequate therapeutic response from the object drug Hansten, PD.in Levy et al, Metabolic Drug Interactions, 2000, pg 715 Proportionality: Abaporu vs Still Life with Golden Bream Pharmacokinetics (PK) & pharmacodynamics (PD) PK - What the body does to the drug? Absorption; distribution, metabolism, excretion (ADME) PD - What the drug does to the body? Drug concentration at the site of action or in the plasma is related to a magnitude of effect Mechanism of drug interaction 1. Pharmacodynamic – one drug affects the ability of another drug to associate with its therapeutic target or receptor Examples: Methotrexate and Trimethoprim/sulfamethoxazole Epinephrine and non selective beta-blockers Grattagliano, J Fam Prac, 2010 (59) 6: 322-329 Mechanism of drug interaction 2. Pharmacokinetic: from drug absorption to elimination D - distribution A - absorption M – metabolism (P450 Cytochrome system) E - elimination Grattagliano, J Fam Prac, 2010 (59) 6: 322-329 Really fundamental concepts in drug biotransformation Lipid soluble drugs are poorly excreted in the urine. They tend to store in fat and/or circulate until they are converted (phase I biotransformation) to more water soluble metabolites or metabolites that conjugate (phase II biotransformation) with water soluble substances. Water soluble drugs are more readily excreted in the urine. They may be metabolized, but generally not by the CYP enzyme systems. https://courses.washington.edu/chat543/lectures/PowerPointSlides/Biotransformation_07ho.ppt What is P450 Cytochrome System? A composite of 30 plus isoenzymes referred to as CYP (cytochrome P450) Carries out first phase of biotransformation for a variety of drugs (the substrate drugs) Of significance: CYP 1A2 CYP 2C9 CYP 2C19 CYP 2D6 CYP 2E1 CYP 3A4 Grattagliano, J Fam Prac, 2010 (59) 6: 322-329 Cytochrome P450 Nomenclature CYP = cytochrome P450 2 = genetic family D = genetic sub-family 6 = specific gene NOTE: This nomenclature is genetically based; it does not imply chemical specificity Grattagliano, J Fam Prac, 2010 (59) 6: 322-329 ROLE OF CYP ENZYMES IN HEPATIC DRUG METABOLISM RELATIVE HEPATIC CONTENT OF CYP ENZYMES CYP2D6 2% % DRUGS METABOLIZED BY CYP ENZYMES CYP2E1 7% CYP 2C19 11% CYP 2C9 14% CYP2D6 23% CYP 2C 17% OTHER 36% CYP 1A2 12% CYP 3A4-5 26% CYP 1A2 14% CYP 3A4-5 33% Slide set, S.P. Markey, NIH, 2006 CYP2E 5% Drug Interactions (Liver) CYP Substrate CYP Inhibitor CYP Substrate CYP Inducer Substrate concentration Toxicity Substrate concentration Efficacy Inhibitors of P-450 Results in decreased metabolism and hepatic clearance of the substrate drug Example: Prolonged warfarin effect Warfarin as the substrate combined with Amiodarone as the inhibitor Lynch and Price, Am Fam Physician. 2007 Aug 1;76(3):391-396. Inducers of P-450 Results in increased metabolism and increased hepatic clearance of the substrate Example: Decreased Oral Contraceptive effect ethinyl estardiol as the substrate combined with Tegretol as the inducer Lynch and Price, Am Fam Physician. 2007 Aug 1;76(3):391-396. Drug Metabolism Enzyme Active drug (inactivated by enzyme) Drug Inactive Metabolite Prodrug (needs enzyme to produce active drug) Enzyme Prodrug Active Metabolite Then there are pro-drugs Pro-Drug Plus an inhibitor Pro-Drug does not break down to active metabolites Pro-drug stays pretty much unmetabolized Plus an inducer Pro-Drug breaks down to active metabolites Active metabolites are excessive P-Glycoprotein Drug Transporters Drug efflux transporter with interactions similar to that of CYP3A4; notable example is digoxin as substrate and carvedilol or quinidine as P-gp inhibitors resulting in prolonged digoxin effect The Medical Letter, 2003; (45): 46-48 Enzyme Potent Inhibitor** Potent Inducer^^ Substrate CYP1A2 Amiodarone (Cordarone), cimetidine (Tagamet), ciprofloxacin (Cipro), fluvoxamine (Luvox) Carbamazepine (Tegretol), phenobarbital, rifampin (Rifadin), tobacco Caffeine, clozapine (Clozaril), theophylline CYP2C9 Amiodarone, fluconazole (Diflucan), fluoxetine (Prozac), metronidazole (Flagyl), ritonavir (Norvir), trimethoprim/sulfameth oxazole (Bactrim, Septra) Carbamazepine, phenobarbital, phenytoin (Dilantin), rifampin Carvedilol (Coreg), celecoxib (Celebrex), glipizide (Glucotrol), ibuprofen (Motrin), irbesartan (Avapro), losartan (Cozaar) **—These will slow down substrate drug metabolism and increase drug effect. ^^—These will speed up substrate drug metabolism and decrease drug effect. Lynch and Price, Am Fam Physician. 2007 Aug 1;76(3):391-396. Case 1. Fluconazole and Warfarin EUROPEAN JOURNALOF EMERGENCYMEDICINE, 2002, 9,175-177 A 75-year-old man on chronic anticoagulation with warfarin for atrial fibrillation and transient ischaemic attacks Found to have Candidasis on EGD specimen and Fluconazole given for 4 weeks. Five weeks later presented to the emergency department with complaints of back pain of three days’ duration radiating to both legs and leg weakness. His INR was found to be 40 MRI of the thoracic spine demonstrated a posterior epidural haematoma extending from T3 to T10 and an emergent laminectomy was performed. CYP2C9 and Warfarin If possible, fluconazole should not be used for patients anticoagulated with warfarin. Otherwise close monitoring is required. dabigatran (Pradaxa) and rivaroxaban (Xarelto) Known as a direct thrombin inhibitor (dabigatran) or direct Factor Xa inhibitor (rivaroxaban) Both are approved to reduce the risk of stroke and blood clots (systemic embolism) in patients with non-valvular atrial fibrillation. XARELTO® is also indicated for the prophylaxis of deep vein thrombosis (DVT), which may lead to pulmonary embolism (PE) in patients undergoing knee or hip replacement surgery. Drug interactions with p-glycoproteins, CYP34A Prescriber's Letter 2011; 18(12):271220 Case 2. Simvastatin and Amiodarone Ann Pharmacother 2006;40:753-7. A 72-year-old white man presented with thigh weakness, achiness, and dark urine for 7 days. Coronary artery bypass had been performed earlier and Amiodarone 200 mg/day was started and later simvastatin 80 mg/day was added Labs included creatine kinase (CK) 19 620 U/L (reference range 60–224), blood urea nitrogen 50 mg/dL, creatinine 2.6 mg/dL, (AST) 912 U/L (30–60), (ALT) 748 U/L (30–60), urine myoglobin 71100 μg/L (<50), and serum myoglobin 13 877 μg/L (<110). Rhabdomyolysis Adapted from: Sinoway L, Li J. J Appl Physiol 2005;99:5–22. CYP3A4 and Statins Simvastatin is metabolized primarily by CYP3A4, and amiodarone is a recognized inhibitor of this enzyme Facts about statins A class of prescription drugs used together with diet and exercise to reduce blood levels of lowdensity lipoprotein (LDL) cholesterol Marketed as single-ingredient products, including Lipitor (atorvastatin), Lescol (fluvastatin), Mevacor (lovastatin), Altoprev (lovastatin extendedrelease), Livalo (pitavastatin), Pravachol (pravastatin), Crestor (rosuvastatin), and Zocor (simvastatin) Also marketed as combination products, including Advicor (lovastatin/niacin extended-release), Simcor (simvastatin/niacin extended-release), and Vytorin (simvastatin/ezetimibe) http://www.fda.gov/Drugs/DrugSafety/ucm293101.htm A word from the FDA Do not use simvastatin with these medications: Itraconazole Ketoconazole Gemfibrozil Cyclosporine Danazol Posaconazole Erythromycin Clarithromycin Telithromycin HIV protease inhibitors Nefazodone http://www.fda.gov/Drugs/DrugSafety/ucm256581.htm A word from the FDA Do not use more than 10mg of simvastatin with these medications: Verapamil , Diltiazem (Note: These drugs are contraindicated with Simcor as Simcor is only available with 20 mg or 40 mg of simvastatin.) Do not use more than 20mg of simvastatin with these medications: Amlodipine, Amiodarone , Ranolazine Avoid large quantities of grapefruit juice (>1 quart daily) Lovastatin Dose Limitations Contraindicated with lovastatin: Itraconazole Ketoconazole Posaconazole Erythromycin Clarithromycin Telithromycin HIV protease inhibitors Boceprevir Telaprevir Nefazodone http://www.fda.gov/Drugs/DrugSafety/ucm293101.htm#hcp Lovastatin Dose Limitations Avoid with lovastatin: Cyclosporine Gemfibrozil Do not exceed 20 mg lovastatin daily with: Danazol Diltiazem Verapamil Do not exceed 40 mg lovastatin daily with: Amiodarone Avoid large quantities of grapefruit juice (>1 quart daily) Case 3. Tamoxifen and SSRI Am J Psychiatry 165:10, October 2008 “Ms. B” is a 45-year-old woman who was diagnosed with major depressive disorder treated with fluoxetine One year ago, she was diagnosed with breast cancer and underwent surgery, followed by chemotherapy, and then radiation therapy. Tamoxifen (a selective estrogen receptor modulator) added to prevent breast cancer recurrence. What potential problems do you forsee? CYP2D6 and Fluoxetine CYP2D6 is necessary for conversion of the prodrug tamoxifen to its primary active metabolite endoxifen. Fluoxetine is a strong CYP2D6 inhibitor, and therefore inhibits the conversion tamoxifen to endoxifen and compromise the efficacy of tamoxifen. Citalopram (Celexa®), sertraline (Zoloft®), and venlafaxine (Effexor®) do not significantly inhibit CYP2D6 http://www.azcert.org/medical-pros/education/index.cfm Case 4. Plavix and PPIs Cardiology in Review • Volume 17, Number 4, July/August 2009 A 52-year-old man found to have 80% stenosis in the right coronary artery and a drug-eluting stent placed. On discharge, patient was on metoprolol 25 mg twice daily, lisinopril 5 mg once daily, pravastatin 40 mg once daily, aspirin 325 mg once daily, clopidogrel 75 mg once daily, and pantoprazole 40 mg once daily (for stress ulcer prophylaxis during coronary care unit stay). Should a PPI be continued for gastrointestinal (GI) ulcer prophylaxis? If so, is there a preferred PPI to use in this case? CYP2C19 and Plavix Clopidogrel (Plavix®) is a prodrug. Omeprazole (Prilosec®) is thought to inhibit the CYP2C19 metabolism of clopidogrel to its active metabolite. Concomitant use of omeprazole and clopidogrel to be avoided. Unknown to what extent other PPIs may interact with clopidogrel. However, esomeprazole (Nexium®) also inhibits CYP2C19 and should also be avoided. http://www.azcert.org/medical-pros/education/index.cfm Do you like controversies? FDA Advisory: “Concomitant use of clopidogrel and omeprazole should be avoided … may not get full protective anticlotting effect” The Medical Letter: “Patients at risk for upper GI bleeding who take clopidogrel should also take a PPI, but not Omeprazole… Pantoprazole would be reasonable choice” The Medical Letter, Nov 29, 2010 (52): 93 Do you like controversies? ACCF / ACG / AHA 2010 Expert Consensus: “Pharmacokinetic and pharmacodynamic studies… suggest that concomitant use of clopidogrel and a PPI reduces the antiplatelet effects of clopidogrel … It is not established that changes in these surrogate endpoints translate into clinically meaningful differences” J. Am. Coll. Cardiology. 2010; (56): 2051-2066 Case 5. Digoxin and Itraconazole Annals of Int Med, 1992; 116 (6): 525 68 y/o man with chronic atrial fib treated with Digoxin for rate control Found to have elbow swelling with positive fungal culture on aspirate Treated with ketoconazole with no effect and switched to itraconazole Two weeks later, developed nausea and vomiting which resolved after itraconazole was stopped P-Glycoprotein Drug Transporters Drug efflux transporter with interactions similar to that of CYP3A4; notable example is digoxin as substrate and carvedilol or quinidine as P-gp inhibitors resulting in prolonged digoxin effect The Medical Letter, 2003; (45): 46-48 Elderly and Polypharmcy By some estimates patients 65 and older average 8 different drugs daily Frequently used: Nonnarcotic analgesic Calcium channel blocker Diuretics Antidepressants ACE inhibitors Rathore et al, Fam Med (1998); 30: 733-739 Why the elderly are more susceptible to adverse drug reactions Aging state: renal clearance diminished, hepatic drug metabolizing enzyme activity lessened, and drug sensitivity heightened Commonly seen: prolonged opioid, benzodiazepine and CNS depressant effects and bleeding from anticoagulation; paradoxically less responsive to beta adrenergic blockers and agonists Cooney and Pascuzzi, Clin Geriatr Med, 2009 (25): 221-233 The Beers Criteria (age over 65) Medications or medication classes that generally should be avoided due to ineffectiveness or unnecessarily high risk when safer alternatives are available Medications should not be used due to concurrent medical conditions Fick et al, Arch Int Med 2003; (163): 2716-24 Beers: Drugs to avoid (with severity rating) Propoxyphene (Darvon) Low Anticholinergics and antihistamines HIgh Indomethacin (Indocin) High Diphenhydramine (Benadryl) High Pentazocine (Talwin) High Meperidine (Demerol) High Trimethobenzamide (Tigan) High Non COX selective NSAIDs – long term use (Feldene, Daypro) High Muscle relaxants High Daily Fluoxetine (Prozac) High Flurazepam (Dalmane) High Amiodarone (Cordarone) High Amitriptyline (Elavil) High Nitrofurantoin (Macrodantin) High Doxepin (Sinequan) High Mineral Oil High Long acting benozodiazepines High Nifedipine – short acting (Procardia, Adalat) High Disopyramide (Norpace) High Doxazosin (Cardura) Low Beers: Avoid with disease states Disease state Drug Severity rating Heart failure Disopyramide (Norpace) High Hypertension Pseudoephedrine High Seizure disorder Chlorpromazine (Thorazine), Clozapine (Clozaril) High Bladder outlet obstruction Anticholinergics and antihistamines High Stress incontinence Alpha blockers (Prazosin), long High acting benzodiazepines Arrhythmias Tricyclic antidepressants (amitriptyline, doxepin) High Insomina Decongestants, theophylline High Parkinson disease Metoclopramide (Reglan); tacrine (Cognex) High Syncope or falls Benzodiazepines and tricyclics High Estimated Rates of Emergency Hospitalizations for Adverse Drug Events in Older U.S. Adults, 2007–2009. Budnitz DS et al. N Engl J Med 2011;365:2002-2012 Elderly and Adverse Drug Effects Four medications or medication classes were implicated alone or in combination in 67.0% of emergency hospitalizations Warfarin (33.3%) Insulins (13.9%), Oral antiplatelet agents (13.3%), and Oral hypoglycemic agents (10.7%) High-risk medications were implicated in only 1.2% hospitalizations. Common presentation on admission Acute hemorrhages Hypoglycemia Neurologic symptoms (loss of consciousness, seizures, changes in mental status). Electrolyte or fluid-volume disturbances or nonspecific weakness NEJM, 2011; 365: 2002-2012 3 drug interactions in elderly leading to hospitalizations Population-based case control study of Canadian residents over age 66 Hospitalization with glyburide, digoxin, or ACE-I when combined with other drugs vs similar matched controls Glyburide + co-trimoxazole Digoxin + clarithyromycin ACE-I + potassium sparing diuretic Juurlink et al., JAMA (2003), 289: 1652-1657 Hypertensive Drug Effects (especially in the elderly) Increased effects Decreased effects Beta blockers Cimetidine, Quinidine, Fluoxetine NSAIDs, Rifamycins, Phenobarbital, Antacids (Al, Ca) Calcium Channel Blockers Cimetidine, Macrolides, Antifungal Azoles Grapefruit juice St. John’s Wort Rifamycins Phenytoin ACE-I and ARB Diuretics, Chlorpromazine NSAIDs Antacids Diuretics Alpha blockers ----------Phosphodiesterase inhibitors NSAIDs, steroids, cholestyramine, colestipol NSAIDs Cooney and Pascuzzi, Clin Geriatr Med, 2009 (25): 221-233 Other Drug Interactions BP drug Added drug Effect Beta blockers Antihyperglycemics Non-dihydropyridine calcium channel blocker Masks hypoglycemia Decreased AV conduction leading to bradycardia Calcium channel blockers Digoxin, carbamazepine, midazolam, risperidone Risk of toxicity with nondihydropyridines ACE-I and ARB Potassium supplements, potassium sparing diuretics, trimethoprim; Loop diuretics Hyperkalemia Lithium Digoxin Risk of lithium toxicity Risk of digoxin toxicity from diuretic induced hypokalemia Diuretics Increased risk hypovolemia Cooney and Pascuzzi, Clin Geriatr Med, 2009 (25): 221-233 Minimizing Drug Interaction Risks L – List each drug’s name and dosage I – Indication and clearly defined therapeutic goal; discontinue if not S – Simple regimen as possible with once or twice a day dosing T – Treat multiple symptoms with single drug rather than multiple drugs E – Educate patients about adverse events and address all non prescriptions and supplements N – Narrow therapeutic window drugs, avoid Werder, J Fam Prac 2005 (4) 5 To Reduce Risk of Adverse Drug Interactions Use of alternative noninteracting medications Adjust dose of object drug: when possible based on changes in response rather than prophylactically ( to avoid sub or supra dosing) Adjust dosing times (object drug two hours before or six hours after the binding drug) Monitor for altered response (lab monitoring) Computerized system to avoid adverse drug interactions (still patients may have multiple providers and multiple pharmacies) Alternative Noninteracting Medications Drug Class Interaction Susceptible Alternatives Benzodiazepine CYP3A4 Inhibition Alprazolam, Diazepam, Midazolam, Triazolam Lorazepam, Oxazepam, Temazepan Calcium Channel Blockers CYP3A4 Inhibition Diltiazem, Verapamil Dihydropyridine CCB Coumarin anticoagulants CYP2C9 Inhibition Acenocoumarol, Warfarin Phenprocoumon dabigatran and rivaroxaban HMG-CoA reductase inhibitor CYP3A4 Inhibition Lovastatin Simvastatin Fluvastatin, Pravastatin Hansten, P.D. in Levy et al, Metabolic Drug Interactions, 2000, pg 725 Alternative Noninteracting Medications Drug Class Interaction Susceptible Alternatives H2-receptor antagonists Various CYP 450 isoenzymes Inhibition Cimetidine Famotidine, Nizatidine, Ranitidine Macrolide antibiotics CYP3A4 Inhibition Clarithromycin, Erthyromycin Azithromycin, Dirithromycin Quinolone antibiotics CYP1A2 Inhibition Ciprofloxacin, Enoxacin, Grepafloxacin Levofloxacin, Ofloxacin, Lovafloxacin, Sparfloxacin, Trovafloxacin Selective serotonin reuptake inhibitors CYP2D6 Inhibition Fluoxetine, Paroxetine Citalopram, Fluvoxamine, Sertraline Azithromycin and CV deaths 1. FDA Safety Announcement Mar 12,2013 2.Health care professionals should consider the risk of fatal heart rhythms with azithromycin when considering treatment options for patients who are already at risk for cardiovascular events 3.Alternative drugs in the macrolide class, or non-macrolides such as the fluroquinolones also have the potential for QT prolongation or other significant side effects http://www.fda.gov/Drugs/DrugSafety/ucm341822.htm Excess Risk of Cardiovascular Death with Azithromycin as Compared with Amoxicillin, According to Decile of Cardiovascular Risk Score. Ray WA et al. N Engl J Med 2012;366:1881-1890 Avoiding Drug Interactions for Consumers http://www.fda.gov/ForConsumers/ConsumerUpdates/ucm096386.htm Reporting Adverse Drug Effects to Food and Drug Administration (FDA) http://www.fda.gov/Safety/MedWatch/HowToReport/ default.htm General References 1. Cozza, K. Drug Interactions. 2003, American Psychiatric Publishing Inc 2. www.hanstenandhorn.com (100 Top Drug Interactions) 3. www.drug-interactions.com (P450-mediated interactions) 4. http://medicine.iupui.edu/clinpharm/ddis/ 5. https://crediblemeds.org/healthcare-providers/drug-druginteraction/ 6. For Simvastatin product information http://www.accessdata.fda.gov/drugsatfda_docs/label/2012/019766s085lbl.pdf http://www.medscape.com/viewarticle/750421 7. www.themedicalletter.com (pay site) 8. www.prescriberletter.com (pay site) 9. www.lexi.com (pay site and apps) In Summary Health care providers must pay attention to patients taking multiple drugs Significant drug interactions are clinically important if they retard or accentuate the normal effects of drugs Consult tables such as the P450 cytochrome system or your electronic pharmacology software or call your pharmacist if there is a question about interactions Elderly patients require special attention Education of the patient and ongoing education of the clinician to drug interactions is essential to good care Questions Backers of the USS Texas, now a museum, hope to stop further corrosion to its hull by moving it to dry land.