* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download DEHYDRATION - ALKENE TEST EXERCISES
Woodward–Hoffmann rules wikipedia , lookup
Fischer–Tropsch process wikipedia , lookup
Physical organic chemistry wikipedia , lookup
Cracking (chemistry) wikipedia , lookup
George S. Hammond wikipedia , lookup
Asymmetric induction wikipedia , lookup
Hofmann–Löffler reaction wikipedia , lookup
Stille reaction wikipedia , lookup
Wolff rearrangement wikipedia , lookup
1,3-Dipolar cycloaddition wikipedia , lookup
Ene reaction wikipedia , lookup
Kinetic resolution wikipedia , lookup
Tiffeneau–Demjanov rearrangement wikipedia , lookup
Wolff–Kishner reduction wikipedia , lookup
Baylis–Hillman reaction wikipedia , lookup
Ring-closing metathesis wikipedia , lookup
Petasis reaction wikipedia , lookup
Strychnine total synthesis wikipedia , lookup
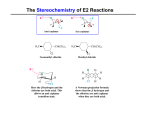
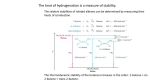
DEHYDRATION - ALKENE TEST EXERCISES 1. Give a detailed mechanism for the acid-catalyzed dehydration of cyclohexanol to cyclohexene. 2. What is the function of the acid catalyst in promoting the dehydration of alcohols? 3. Why would concentrated hydrochloric acid be an inappropriate catalyst for the dehydration of alcohols ? 4. Why is the formation of substitution products involving displacement of water by attack of bisulfate or of dihydrogen phosphate upon a protonated alcohol not a reaction of concern in these procedures ? 5. Write equations for the chemical reaction(s) that you will use to demonstrate the presence of alkene(s) in your distilled product. 6. Why is it important to dry the crude alkene(s) prior to the final distillation ? 7. What is the "limiting reagent" in the dehydration procedures ?