* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Amino acids
Survey
Document related concepts
Signal transduction wikipedia , lookup
Interactome wikipedia , lookup
Point mutation wikipedia , lookup
Nucleic acid analogue wikipedia , lookup
Fatty acid synthesis wikipedia , lookup
Peptide synthesis wikipedia , lookup
Western blot wikipedia , lookup
Metalloprotein wikipedia , lookup
Two-hybrid screening wikipedia , lookup
Basal metabolic rate wikipedia , lookup
Protein–protein interaction wikipedia , lookup
Nuclear magnetic resonance spectroscopy of proteins wikipedia , lookup
Genetic code wikipedia , lookup
Amino acid synthesis wikipedia , lookup
Fatty acid metabolism wikipedia , lookup
Biosynthesis wikipedia , lookup
Transcript
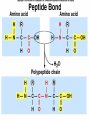
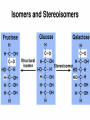
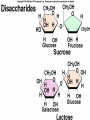
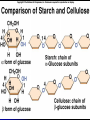
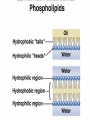
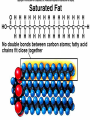
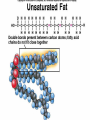
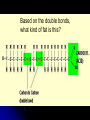
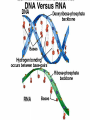
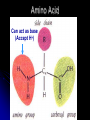
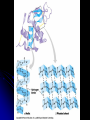
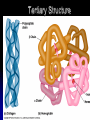
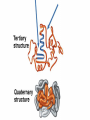
Organic Compounds Carbon containing compounds Form covalent bonds with (usually) other carbon or hydrogen atoms By virtue of its 4 valence electrons: Can form single, double, or triple covalent bonds with itself (and other atoms) Can form a variety of shapes (chains, rings, branches sheets, etc.) Carbon Molecules Aromatics Buckyball • Hydrocarbons – Organic molecules consisting only of carbon and hydrogen; energyrich • Functional Groups - Groups of molecules that have definite chemical properties they retain no matter where they occur. Macromolecules “large” molecules synthesized by living things Many are “organic” (contain a carbon core) Most have “functional groups” attached to carbon core (determine function of molecule) Biological Macromolecules Carbohydrates Lipids Proteins Nucleic Acids ATP Building Macromolecules Most organic macromolecules are polymers made by using enzymes to help link together smaller monomers. Done in a process called dehydration synthesis (or condensation). This process removes + one H from one molecule and one OH from another molecule (H20) and cause the two to link together Diversity in polymers is Achieved by arranging The same monomers in Different ways These different arrangements Are called ISOMERS Breaking macromolecules uses the reverse process called hydrolysis; process in which water is added to break apart polymers into monomers. Carbohydrates Carbohydrates are a loosely defined group of molecules that contain carbon, hydrogen, and oxygen in a 1:2:1 ratio (CH2O) Serve as energy storing molecules and structural elements Energy is stored in the C-H bonds; when the bond is broken, energy is released. Named according to size and structure (triose, pentose, hexose) Monosaccharides – “simple” sugars; glucose, fructose, galactose 3-6 carbon sugar C6H12O6 is the common fuel source for cells Many monosaccharides have the same empirical formula, but different arrangement of double bonds &/or OH- groups. This causes their properties to be different. Sugar Isomers Alternative forms of the same chemical formula. Glucose Fructose Galactose Glyceraldehyde (building blocks) (ENERGY) Ribose (RNA) (DNA) (Plants) – created when 2 monosaccharides are joined by dehydration synthesis. (C12H22O11) Purpose: so sugars can be transported w/i an organism without being metabolized. Disaccharides Ex: glucose + glucose = maltose glucose + fructose = sucrose glucose + galactose = lactose – formed from assembled disaccharides into an insoluble form and then stored. Storage: starch (plant polysaccharide), glycogen (animal starch stored in liver and muscles) Structural: pectin and cellulose (plant polysaccharides used to make plant cell walls), chitin (animal structural sugar) Polysaccharides – chains of glucose produced by plants, coil in water, are rendered insoluble. Enzymes can cleave them randomly into smaller, more soluble, fragments and then cut into smaller disaccharides of maltose and then smaller monosaccharides of glucose for cell metabolism. Starch – produced by animals; length and number of branches are larger than plant starches. Glycogen When the animal body needs energy, it looks to the liver to hydrolize glycogen and release the resulting monosaccharides to the cells. From there, the cells, through respiration release the energy stored in the bonds of the monosaccharide molecule. The energy is converted to another energy compound – ATP. Cellulose and Pectin Chain of glucose molecules that consists of all beta-glucose subunits. Cleavage of subunits requires an enzyme most organisms lack Why Fiber? We can’t break fiber down but we need it It makes us salivate Feel full Go to the bathroom Prevents colon cancer Cows, for example, have bacteria and protists in their gut that have the necessary enzymes for digesting the cellulose of grasses and grains. Therefore cows can get energy from this food – we cannot. This makes cellulose a good structural material. – branched polysaccharide produced by plants (citrus); sugars are cross-linked to form a mesh of glucose which can only be broken down by a specific enzyme. Different plants have different mesh sizes. (Used as gelling agents & diarrhea management) Pectin Chitin - Modified form of cellulose with nitrogen group added to the glucose units. Structural unit in many insects. carb trivia – Sugars have either aldehyde or ketone functional groups Sugars can be recognized by their “ose” endings Most sugars form rings in aqueous solutions More ARE ALL FATS BAD? Lipids Monomers are fatty acids and glycerol Compounds with > 2:1 H:O ratio and large numbers of C-H bonds C-H bonds are nonpolar and hydrophobic; fat molecules tend to cluster and are insoluble Lipids store energy well because of the C-H bonds Most lipids have more than 40 carbons 9 kcal of energy for fats compared to 4 kcal of energy for carbohydrates. People gain weight because their energy levels drop as they get older, but their food intake doesn’t Types of lipids: – used in cell membranes; one fatty acid is replaced with a polar phosphate head. Phospholipids Triglycerides (Fats, Oils) Fats – composed of a backbone glycerol molecule and 3 fatty acid chains (long hydrocarbon chains – triglyceride). Fatty acids can be of varying lengths; 14-20 carbons are average Saturated fats – all carbons on the internal structure have H+ bonded to them These fats (which come mostly from animals) tend to be solid at room temp; ex: Crisco; tropical oils (palm and coconut) Unsaturated + H fats – some are lacking on internal carbons and double bonds are in its place fats – more than one missing H+ and more than one double bond Tend to have low melting point because chains bend at double bonds. Usually liquid at room temperature. Polyunsaturated Based on the double bonds, what kind of fat is this? Saturated Fats Unsaturated Fats Solid at room temp. A. Usually liquid (except palm & B. One or more coconut oil) double bonds B. All single C-C=C-C=C-C-C bonds C. Found in Artic C-C-C-C-C-C-C-C animals C. Increases Cholesterol A. Hydrogenating (adding hydrogen) unsaturated fats to make them solid (peanut butter and margarine) are just as unhealthy as saturated fats Unhydrogenated Peanut Butter Animal fats are saturated, while most plant fats are unsaturated. Can convert oil into solid fat through hydrogenation (adding hydrogen). – 4 carbon ring lipid (ex: cholesterol), used to make hormones like testosterone and estrogen Terpenes – long lipid chains that contain pigments (ex: chlorophyll, rhodopsin, rubber) Steroids – 5 carbon ring head and 2 nonpolar tails; act as chemical messengers; used in muscle contractions, blood vessel dilation, ovulation, uterine contraction, blood clotting, inflammatory responses; aspirin inhibits prostaglandin production Prostaglandins Men need 4-7% body fat Women need 9-16% body fat Going below these percentages is harmful since your body can’t perform all its normal functions Low Carb Diets? Carbs are needed to synthesize glycogen which are needed for effective workouts Without glycogen, you cannot burn as much fat Body uses fuel in this order (Glycogen, Fat, Muscle) Low calorie diets make your body burn muscle which causes a loss in total calorie burning potential – muscle cells have more mitochondria) Weight comes off because of lost muscle but comes back as fat (NOT GOOD) Good Things About Fat More energy than carbs or proteins Helps absorb Vitamins A, C, etc. Healthy skin Gives us fatty acids for growth The Good – Omega-3 Fats Lower blood pressure Decrease risk of heart attack Protect against irregular heartbeats How much should you eat? YUM! You should consume 20 to 30% of your total calories from fat No more than 10% should be saturated fats Too much or too little can be a health risk! Nucleic Acids (Information Molecules) Nucleic acids are information storage devices, and serve as templates to produce precise copies of themselves. Nucleic acids are polymers made of monomers called “nucleotides” Nucleotide consists of Sugar (deoxyribose or ribose) Phosphate Nitrogen containing “base” (adenine, guanine, cytosine, thymine) ATP Adenosine Triphosphate Nucleotide derivative (adenine) Cell battery Manufactured in the mitochondria of cell as a result of the breakdown of glucose Energy of the molecule is stored in the bonds that hold the phosphates together Pop Quiz 1) What does hydrolysis do? 2) What are the building blocks of lipids? 3) Why is cellulose difficult to digest? 4) How do animals store glucose? 5) “Double helix” describes what? 6) Phospholipids always orient so their tails face: 7) How is sucrose (disaccharide) created from glucose and galactose (both monosaccharides) Proteins Proteins are polymers of amino acids. Covalent bond linking two amino acids = peptide bond. Proteins composed of one or more long chains = polypeptides Polypeptides composed of amino acids linked by peptide bonds. Amino Acid Can act as base (Accept H+) Amino Acids Build Proteins acids – molecules that contain an amino group (-NH2) or carboxyl group (-COOH), a H+, and a functional (R) group all bonded to a central carbon Amino acids determine the shape of the protein Amino 20 common amino acids in nature (Make tens of thousands of proteins in human) The functional (side) groups give the amino acid its unique chemical properties and thus the protein properties 20 Common Amino Acids grouped into five chemical classes, based on functional (R) groups: Nonpolar amino acids Polar uncharged amino acids Ionizable amino acids Aromatic amino acids Special-function amino acids – contain –CH2 or –CH3 (Hydrophobic) Nonpolar uncharged – Contain O or only –H (Hydrophilic) Polar – acid (R groups are negative) or base groups (R groups = +) Ionizable Aromatic – contain rings with alternating single and double bonds Special functions – cause chaining, linking, or bending of protein molecule 2 categories of proteins: Structural – fibrous; muscle hair, cell markers Functional – globular; enzymes, antibodies, venom, peptide messengers, globulins, hormones Protein Functions Enzyme Catalysis - catalase Defense - antibodies Transport – cell membranes Support - cytoskeleton Motion – cilia, flagella Regulation – receptor proteins Proteins consist of long amino acid chains folded into complex shapes. Primary Structure – 1o Specific amino acid sequence; chains; characteristic of structural proteins Structure - 2o Folding of amino acid chain by hydrogen bonding into coils (alpha) and pleats (beta); characteristic of structural proteins Motif – variations on secondary structure; ex: b barrel, Beta-alpha-beta) Secondary Motifs Water helps to position motifs and also helps to fold nonpolar side groups into the interior. Stability of a protein is influenced by how well its interior fits together. Spider silk Bullet-proof clothing Wear-resistant lightweight clothing Ropes, nets, seat belts, parachutes Rust-free panels on motor vehicles or boats Biodegradable bottles Bandages, surgical thread Artificial tendons or ligaments, supports for weak blood vessels. Tertiary o 3 Structure folded shape due to hydrophobic Final interactions with water hydrogen and ionic bonding between R groups disulfide bridges. bonding determined by primary structure (types of amino acids and side groups) Quaternary o 4 – Structure When 2 or more polypeptide chains join to form a functional protein. Ex: Hemoglobin is composed of 2 a chains and 2 b chains. Tertiary Structure Substitution of amino acids due to mutations, can interfere with protein stability Characteristic of functional proteins o 1 o 2 and are usually structural proteins o o 3 and 4 are usually functional (globular) proteins. How Proteins Fold Normal cells contain special proteins (chaperonins) that help new proteins fold correctly. Exact workings are controversial. Chaperonin deficiencies (and, therefore, improper protein folding) may play role in certain diseases. How Proteins Unfold Alteration of a protein’s environment may cause denaturation. Usually renders protein biologically inactive. pH extremes, temperature, ionic concentration (salt) of protein’s environment Some denaturation is reversible; most are not. Pop Quiz 1) Term for a “large molecule made of similar subunits”? 2) Building blocks of proteins? 3) Hair is made of protein. What category of protein? 4) Which protein structure is not affected by denaturing heat? 5) What are the monomers of protein? Helpful Websites Kinetics Tutorial MathemoleSite; Molecules, Mass Water and pH Arizona University Chemistry Review Covalent Bonds pH Tutorial Maricopa Atoms and Molecules Review Water Chemistry Chemistry of pH Periodic Table Animated Water Tutorial pH Scale pH/pOH Game Functional Groups Amino Acids Monosaccharide Resource Macromolecules Protein Explorer Carbon Facts