* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download MAP Kinase Pathways
Survey
Document related concepts
Endomembrane system wikipedia , lookup
Organ-on-a-chip wikipedia , lookup
Extracellular matrix wikipedia , lookup
Cell culture wikipedia , lookup
Protein phosphorylation wikipedia , lookup
Cell growth wikipedia , lookup
Cytokinesis wikipedia , lookup
G protein–coupled receptor wikipedia , lookup
Cellular differentiation wikipedia , lookup
Tyrosine kinase wikipedia , lookup
Biochemical cascade wikipedia , lookup
List of types of proteins wikipedia , lookup
Transcript
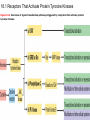
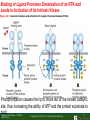
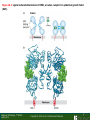
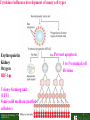
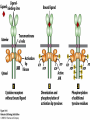
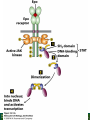
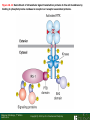
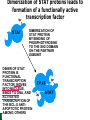
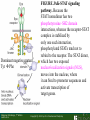
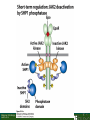
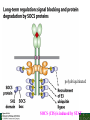
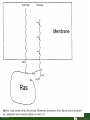
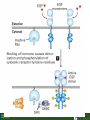
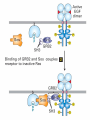
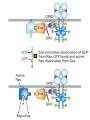
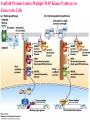
Lodish Berk Kaiser Krieger scott Bretscher Ploegh Matsudaira MOLECULAR CELL BIOLOGY SEVENTH EDITION CHAPTER 16 Signaling Pathways That Control Gene Expression 2015-10-13 Copyright © 2013 by W. H. Freeman and Company Chapter Opener A molecular valentine-dimerized extracellular domain of the epidermal growth factor receptor (red, yellow, and green) bound to two molecules of epidermal growth factor (magenta). Molecular Cell Biology, 7th Edition Lodish et al. Copyright © 2013 by W. H. Freeman and Company Extracellular signals can have both short- and long-term effects on cells. Short-term effects are usually triggered by modification of existing proteins or enzymes, as we saw in Chapter 15. Many extracellular signals also affect gene expression and thus induce long-term changes in cell function. Long-term changes include alterations in cell division and differentiation, such as occur during development and cell fate determination. Molecular Cell Biology, 7th Edition Lodish et al. Copyright © 2013 by W. H. Freeman and Company No signaling pathway acts in isolation. Many cells respond to multiple types of hormones and other signaling molecules; some mammalian cells express ~100 different types of cell-surface receptors, each of which binds a different ligand. Since many genes are regulated by multiple transcription factors that in turn are activated or repressed by different intracellular signaling pathways, expression of anyone gene can be regulated by multiple extracellular signals. Especially during early development, such "cross talk" between signaling pathways and the resultant sequential alterations in the pattern of gene expression eventually can become so extensive that the cell assumes a different developmental fate. Molecular Cell Biology, 7th Edition Lodish et al. Copyright © 2013 by W. H. Freeman and Company Figure 16.1 Several common cell-surface receptors and signal transduction pathways. Molecular Cell Biology, 7th Edition Lodish et al. Copyright © 2013 by W. H. Freeman and Company 16.1 Receptors That Activate Protein Tyrosine Kinases Figure 16.2 Overview of signal transduction pathways triggered by receptors that activate protein tyrosine kinases. Molecular Cell Biology, 7th Edition Lodish et al. Copyright © 2013 by W. H. Freeman and Company Numerous Factors Regulating Cell Division and Metabolism Are Ligands for Receptor Tyrosine Kinases (RTK) These RTK ligands include many, such as nerve growth factor (NGF), platelet-derived growth factor (PDGF), fibroblast growth factor (FGF), and Epidermal growth factor (EGF), that stimulate proliferation and differentiation of specific cell types. Others, such as insulin, regulate expression of multiple genes that control sugar and lipid metabolism in liver, muscle, and adipose (fat) cells. Molecular Cell Biology, 7th Edition Lodish et al. Copyright © 2013 by W. H. Freeman and Company Binding of Ligand Promotes Dimerization of an RTK and Leads to Activation of Its Intrinsic Kinase Figure 16.3 General structure and activation of receptor tyrosine kinases (RTKs). Phosphorylation causes the lip to move out of the kinase catalytic site, thus increasing the ability of ATP and the protein substrate to bind. Molecular Cell Biology, 7 Edition th Lodish et al. Copyright © 2013 by W. H. Freeman and Company Figure 16.4 Ligand-induced dimerization of HER1, a human receptor for epidermal growth factor (EGF). Molecular Cell Biology, 7th Edition Lodish et al. Copyright © 2013 by W. H. Freeman and Company Homo- and Hetero-oligomers of Epidermal Growth Factor Receptors Bind Members of the Epidermal Growth Factor Superfamily Figure 16.7 The HER family of receptors and their ligands. Molecular Cell Biology, 7th Edition Lodish et al. Copyright © 2013 by W. H. Freeman and Company Understanding the HERs has helped explain why a particular form of breast cancer is so dangerous and has led to an important drug therapy. Amplification of the HER2 gene occurs in approximately 25 percent of breast cancers, resulting in overexpression of HER2 protein in the tumor cells. Breast cancer patients with HER2 overexpression have a worse prognosis, including shortened survival, than do patients without this abnormality. Discovery of the role of HER2 overexpression in certain breast cancers led researchers to develop monoclonal antibodies specific for the HER2 protein. These have proved to be effective therapies for those breast cancer patients in which HER2 is overexpressed, reducing recurrence by about 50 percent in these patients. Molecular Cell Biology, 7th Edition Lodish et al. Copyright © 2013 by W. H. Freeman and Company Cytokines Influence Development of Many Cell Types • Erythropoietin (Epo) • Granulocyte Colony Stimulating Factor (G-CSF) • Thrombopoietin (Tpo) • Prolactin (Prl) • Growth Hormone (GH) interleukin 2 IL-4 interferon Molecular Cell Biology, 7th Edition Lodish et al. Copyright © 2013 by W. H. Freeman and Company Cytokine Receptors and the JAK-STAT Pathway Cytokine Receptors and Receptor Tyrosine Kinases Share Many Signaling Features • Hormone- induced receptor dimerization • Activation of JAK protein tyrosine kinase • Phosphorylation of tyrosine residues on the receptor • Receptor phosphotyrosine residues bind to SH2 domains on several signal transduction proteins • Activation of Stat transcription factors • Partnering of Stats with other transcription factors • Termination of signaling by activation of protein tyrosine phosphatases • Inhibition of signaling by CIS proteins containing only SH2 domains Molecular Cell Biology, 7 Edition Copyright © 2013 by W. H. Freeman and Company STAT: Lodish et al. signal transducer and activator of transcription th HEMATOPOIESIS Epo acts to stimulate the proliferation and differentiation of erythroid progenitor cells to mature red cells Myeloid G-CSF CFU-GM Granulocytes IL-3, GM-CSF, SCF IL-6 M -CSF CFU-MEG SCF Monocytes TPO IL-3, GM-CSF CFU-GEMM Platelets BFU-E Epo SCF CFU-E Epo GM -CSF IL-3 Erythrocytes Pluripotent Stem Cell CFU-Eo IL-3, GM-CSF Eosinophils Molecular Cell Biology, 7th Edition Lodish et al. Lymphoid Progenitor Copyright © 2013 by W. H. Freeman and Company Cytokines influence development of many cell types Prevent apoptosis Erythropoietin Kidney Oxygen HIF-1 3 to 5 terminal cell division Colony-forming unit (CFU) Semi-solid medium (methylcellulose) Molecular Cell Biology, 7th Edition Lodish et al. Copyright © 2013 by W. H. Freeman and Company Receptor-Associated JAK Kinases Activate STAT Transcription Factors Bound to a Cytokine Receptor The JAK2 kinase is tightly bound to the cytosolic domain of the erythropoietin receptor (EpoR). Molecular Cell Biology, 7th Edition Lodish et al. Copyright © 2013 by W. H. Freeman and Company ERYTHROPOIETIN (EPO) THE PROTEIN THAT CONTROLS RED BLOOD CELL PRODUCTION 165 AMINO ACIDS ~ 40% CARBOHYDRATE • PRODUCED BY THE KIDNEY IN RESPONSE TO LOW O2PRESSURE IN THE BLOOD • BINDS TO EPO RECEPTORS ON THE SURFACE OF ERYTHROCYTE PROGENITOR CELLS IN THE BONE MARROW • STIMULATES THESE CELLS TO DIVIDE 5 TO 7 TIMES; EACH OF THE ~30 TO 100 DAUGHTERS THEN DIFFERENTIATES INTO A RED BLOOD CELL • USED CLINICALLY TO TREAT ANEMIA CAUSED BY KIDNEY FAILURE OR BY DISEASES SUCH AS AIDS Molecular Cell Biology, 7th Edition Lodish et al. Copyright © 2013 by W. H. Freeman and Company Epo synthesis induced in response EPO REGULATES REDis CELL MASS IN RESPONSE TO TISSUEto HYPOXIA hypoxia RED CELL MASS Tissue pO2 + Epo By means of the oxygen-sensitive transcription factor HIF-1, the kidney cells respond to low oxygen by synthesizing more erythropoietin and secreting it into the blood. Molecular Cell Biology, 7th Edition Lodish et al. Copyright © 2013 by W. H. Freeman and Company Molecular Cell Biology, 7th Edition Lodish et al. Copyright © 2013 by W. H. Freeman and Company Signal Transduction Proteins that Bind to the Erythropoietin Receptor JAK2 130 kDa CYTOSOLIC PROTEIN TYROSINE KINASE HOMOLOGOUS TO JAK1 AND TYK2 WIDELY EXPRESSED IN HEMATOPOIETIC CELLS AND FIBROBLASTS NO SH2 OR SH3 DOMAINS N-T ERMINAL CON SERVED Molecular Cell Biology, 7th Edition Lodish et al. PSEUDO-KINASE DOMAIN MODULATOR Y KIN ASE D OMAIN T YROSINE KINASE Copyright © 2013 by W. H. Freeman and Company Molecular Cell Biology, 7th Edition Lodish et al. Copyright © 2013 by W. H. Freeman and Company Figure 16.11 Surface model of an SH2 domain bound to a phosphotyrosine-containing peptide. Molecular Cell Biology, 7th Edition Lodish et al. Copyright © 2013 by W. H. Freeman and Company Figure 16.12 Recruitment of intracellular signal transduction proteins to the cell membrane by binding to phosphotyrosine residues in receptors or receptor-associated proteins. Molecular Cell Biology, 7th Edition Lodish et al. Copyright © 2013 by W. H. Freeman and Company P Dimerization of STAT proteins leads to formation of a functionally active transcription factor th Lodish et al. P DIMER OF STAT PROTEIN IS FUNCTIONAL TRANSCRIPTION FACTOR: MOVES INTO NUCLEUS, BINDS TO DNA, AND ACTIVATES TRANSCRIPTION OF THE BCL-X ANTIAPOPTOTIC PROTEIN AMONG OTHERS Molecular Cell Biology, 7 Edition P DIMERIZATION OF STAT PROTEIN BY BINDING OF PHOSPHOTYROSINE TO THE SH2 DOMAIN ON THE PARTNER SUBUNIT STAT STAT STAT Copyright © 2013 by W. H. Freeman and Company Dominant negative mutant Tyr Phe Molecular Cell Biology, 7th Edition Lodish et al. FIGURE JAK-STAT signaling pathway. Because the STAT homodimer has two phosphotyrosine–SH2 domain interactions, whereas the receptor-STAT complex is stabilized by only one such interaction, phosphorylated STATs tend not to rebind to the receptor. The STAT dimer, which has two exposed nuclear-localization signals (NLS), moves into the nucleus, where it can bind to promoter sequences and activate transcription of target genes. Copyright © 2013 by W. H. Freeman and Company Different STATs activate different genes in different cells. In erythroid progenitors:erythropoietin: STAT5Bcl-xL Indeed, mice lacking STAT5 are highly anemic because many of the erythroid progenitors undergo apoptosis even in the presence of high erythropoietin levels. Such mutant mice produce some erythrocytes and thus survive, because the erythropoietin receptor is linked to other anti-apoptotic pathways that do not involve STAT proteins (PI-3 kinase, PLC, MAP kinase…). Because different cell types have unique complements of transcription factors and unique epigenetic modifications on their chromatin, the genes that are available to the activated by any STAT are also different. Molecular Cell Biology, 7th Edition Lodish et al. Copyright © 2013 by W. H. Freeman and Company FIGURE 16-8 Overview of signal-transduction pathways triggered by ligand binding to the erythropoietin receptor (EpoR), a typical cytokine receptor. Molecular Cell Biology, 7 Edition th Lodish et al. Copyright © 2013 by W. H. Freeman and Company Multiple Mechanisms Down-Regulate Signaling from RTKs and Cytokine Receptors Receptor-Mediated Endocytosis Prolonged treatment of cells with ligand desensitization response clathrin-coated pits into endosomes HER1 receptors for this ligand are relatively long-lived, with an average half-life of 10 to 15 hours. HER1 mutants that lack kinase activity Molecular Cell Biology, 7th Edition Lodish et al. Copyright © 2013 by W. H. Freeman and Company Down-regulation of RTK signaling occurs by endocytosis and lysosomal degradation lysosomal degradation Ubiquitin E3 ubiquitin ligase; c-Cbl; "tag“; proteasome TGF-b: Ski Cytokines: SOCS, SPH-1 Receptor-mediated endocytosis Phospotyrosine phosphatases Molecular Cell Biology, 7th Edition Lodish et al. Copyright © 2013 by W. H. Freeman and Company Molecular Cell Biology, 7th Edition Lodish et al. Copyright © 2013 by W. H. Freeman and Company polyubiquitinated Molecular Cell Biology, 7th Edition Lodish et al. SOCS (CIS) is induced by STAT Copyright © 2013 by W. H. Freeman and Company 16.2 The Ras/MAP Kinase Pathway Mutant Ras: many types of human cancer bind but cannot hydrolyze GTP, are permanently in the “on” state and contribute to neoplastic transformation. Viral Ras: H(arvey)-ras, Ki-(rsten)-rasA, Ki-rasB and the N(euroblastoma)-ras gene Determination of the three-dimensional structure of the Ras-GAP complex explained the puzzling observation that most oncogenic, constitutively active Ras proteins (RasD) contain a mutation at position 12. Replacement of the normal glycine-12 with any other amino acid (except proline) blocks the functional binding of GAP, and in essence “locks” Ras in the active GTP-bound state. Molecular Cell Biology, 7th Edition Lodish et al. Copyright © 2013 by W. H. Freeman and Company Molecular Cell Biology, 7th Edition Lodish et al. Copyright © 2013 by W. H. Freeman and Company An Adapter Protein and Guanine Nucleotide– Exchange Factor Link Most Activated Receptor Tyrosine Kinases to Ras Fibroblast cells (3T3) remove serum (growth factors) arrest G0/G1 RasD Proliferation (S phase) arrest Molecular Cell Biology, 7th Edition Lodish et al. Copyright © 2013 by W. H. Freeman and Company + PDGF and EGF + anti-Ras + anti-Raf How does binding of a growth factor (e.g., EGF) to an RTK (e.g., the EGF receptor) lead to activation of Ras? Two cytosolic proteins— GRB2 and Sos —provide the key links (Figure 14-16). An SH2 domain in GRB2 binds to a specific phosphotyrosine residue in the activated receptor. GRB2 also contains two SH3 domains, which bind to and activate Sos. GRB2 thus functions as an adapter protein for the EGF receptor. Sos is a guanine nucleotide–exchange protein (GEF), which catalyzes conversion of inactive GDP-bound Ras to the active GTP-bound form. Molecular Cell Biology, 7th Edition Lodish et al. Copyright © 2013 by W. H. Freeman and Company Molecular Cell Biology, 7th Edition Lodish et al. Copyright © 2013 by W. H. Freeman and Company Molecular Cell Biology, 7th Edition Lodish et al. Copyright © 2013 by W. H. Freeman and Company Molecular Cell Biology, 7th Edition Lodish et al. Copyright © 2013 by W. H. Freeman and Company Molecular Cell Biology, 7th Edition Lodish et al. Copyright © 2013 by W. H. Freeman and Company By use of this screen, researchers identified the genes encoding three important proteins in the Sev pathway (see Figure 14-16): an SH2-containing adapter protein exhibiting 64 percent identity to human GRB2; a guanine nucleotide–exchange factor called Sos (Son of Sevenless) exhibiting 45 percent identity with its mouse counterpart; and a Ras protein exhibiting 80 percent identity with its mammalian counterparts. Molecular Cell Biology, 7th Edition Lodish et al. Copyright © 2013 by W. H. Freeman and Company Binding of Sos Protein to Inactive Ras Causes a Conformational Change That Activates Ras The adapter protein GRB2 contains two SH3 domains, which bind to Sos, a guanine nucleotide–exchange factor, in addition to an SH2 domain, which binds to phosphotyrosine residues in RTKs. Formation of this complex depends on the ability of GRB2 to bind simultaneously to the receptor and to Sos. Thus receptor activation leads to relocalization of Sos from the cytosol to the membrane, bringing Sos near to its substrate, namely, membrane-bound RasGDP. Molecular Cell Biology, 7th Edition Lodish et al. Copyright © 2013 by W. H. Freeman and Company MAP Kinase Pathways Activated Ras promotes formation at the membrane of signaling complexes containing three sequentially acting protein kinases that are associated with a scaffold protein. This kinase cascade culminates in activation of MAP (mitogenactivating protein) kinase, a serine/threonine kinase also known as ERK (extracellular regulated kinase). After translocating into the nucleus, MAP kinase can phosphorylate many different proteins, including transcription factors that regulate expression of important cell-cycle and differentiation-specific proteins. Activation of MAP kinase in two different cells can lead to similar or different cellular responses, as can its activation in the same cell following stimulation by different hormones. Molecular Cell Biology, 7th Edition Lodish et al. Copyright © 2013 by W. H. Freeman and Company An Adapter Protein and Guanine Nucleotide– Exchange Factor Link Most Activated Receptor Tyrosine Kinases to Ras Fibroblast cells (3T3) remove serum (growth factors) arrest G0/G1 RasD Proliferation (S phase) arrest GF …….. Ribosomal S6* ribosomal S6 kinase* (RSK) (mitogen-activating protein) MAP kinase* (P-Y and P-S MAPK) MEK* (MAPKK) Raf (MAPKKK) Molecular Cell Biology, 7th Edition Lodish et al. Copyright © 2013 by W. H. Freeman and Company + PDGF and EGF + anti-Ras + anti-Raf 14.4 MAP kinase pathways Growth factors Molecular Cell Biology, 7th Edition Lodish et al. Copyright © 2013 by W. H. Freeman and Company Signals Pass from Activated Ras to a Cascade of Protein Kinases A remarkable convergence of biochemical and genetic studies in yeast, C. elegans, Drosophila, and mammals has revealed a highly conserved cascade of protein kinases that operate in sequential fashion downstream from activated Ras as follows: 1. Activated Ras binds to the N-terminal domain of Raf, a serine/threonine kinase. 2. Raf binds to and phosphorylates MEK, a dual-specificity protein kinase that phosphorylates both tyrosine and serine residues. 3. MEK phosphorylates and activates MAP kinase, another serine/threonine kinase. 4. MAP kinase phosphorylates many different proteins, including nuclear transcription factors, that mediate cellular responses. Molecular Cell Biology, 7th Edition Lodish et al. Copyright © 2013 by W. H. Freeman and Company Activation of Raf Kinase This inactive conformation is stabilized by a dimer of the 143-3 protein, which binds phosphoserine residues in a number of important signaling proteins. Each 14-3-3 monomer binds to a phosphoserine residue in Raf, one to phosphoserine-259 in the N-terminal domain and the other to phosphoserine-621 (see Figure 14-21). These interactions are thought to be essential for Raf to achieve a conformational state such that it can bind to activated Ras. The binding of RasGTP, which is anchored to the membrane, to the N-terminal domain of Raf relieves the inhibition of Raf’s kinase activity and also induces a conformational change in Raf that disrupts its association with 14-3-3. Raf phosphoserine-259 then is dephosphorylated (by an unknown phosphatase) and other serine or threonine residues on Raf become phosphorylated by yet other kinases. Molecular Cell Biology, 7th Edition Lodish et al. Copyright © 2013 by W. H. Freeman and Company MAP Kinase Regulates the Activity of Many Transcription Factors Controlling Early-Response Genes Addition of a growth factor (e.g., EGF or PDGF) to quiescent cultured mammalian cells in G0 causes a rapid increase in the expression of as many as 100 different genes. ????? These are called early-response genes because they are induced well before cells enter the S phase and replicate their DNA. One important early-response gene encodes the transcription factor c-Fos. Together with other transcription factors, such as c-Jun (AP-1) c-Fos induces expression of many genes encoding proteins necessary for cells to progress through the cell cycle. Most RTKs that bind growth factors utilize the MAP kinase pathway to activate genes encoding proteins like c-Fos that propel the cell through the cell cycle. Molecular Cell Biology, 7th Edition Lodish et al. Copyright © 2013 by W. H. Freeman and Company Figure 16.22 Induction of gene transcription by MAP kinase. The enhancer that regulates the c-fos gene contains a serum-response element (SRE), so named because it is activated by many growth factors in serum. This complex enhancer contains DNA sequences that bind multiple transcription factors. Molecular Cell Biology, 7th Edition Lodish et al. Copyright © 2013 by W. H. Freeman and Company As evidence for this model, abundant expression in cultured mammalian cells of a mutant dominant negative TCF that lacks the serine residues phosphorylated by MAP kinase blocks the ability of MAP kinase to activate gene expression driven by the SRE enhancer. Moreover, biochemical studies showed directly that phosphorylation of SRF by active p90RSK increases the rate and affinity of its binding to SRE sequences in DNA, accounting for the increase in the frequency of transcription initiation. Thus both transcription factors are required for maximal growth factor– induced stimulation of gene expression via the MAP kinase pathway, although only TCF is directly activated by MAP kinase. Molecular Cell Biology, 7th Edition Lodish et al. Copyright © 2013 by W. H. Freeman and Company Scaffold Proteins Isolate Multiple MAP Kinase Pathways in Eukaryotic Cells Molecular Cell Biology, 7th Edition Lodish et al. Copyright © 2013 by W. H. Freeman and Company d) You overexpress Stat that cannot be phosphorylated because its critical tyrosine has been mutated to a phenylanaline. Molecular Cell Biology, 7th Edition Lodish et al. Copyright © 2013 by W. H. Freeman and Company You are studying the response of a fibroblast cell line to treatment with fibroblast growth factor which you know binds to and acts through a receptor tyrosine kinase. The cell line grows and divides when treated with growth factor, but do not in absence of growth factor. A)You express a mutant form of the fibroblast growth factor receptor (FGFR) lacking its entire cytoplasmic domain to a level similar to the wild type receptor. You observe that the cells grow much more slowly than normal when treated with growth factor. Explain. B)You inject anti-Ras antibodies that prevent Ras from binding Raf into a few cells. What is the growth and division phenotype of these cells? Why? C) You express a constitutively active Ras that remains in its GTP bound form. You observe that the cells divide and grow even in the absence of growth factor. Explain. D) You treat the cells with tyrosine kinase inhibitor, and observe the effect on the cell growth and division. What do you see and why? E) FGFR contains a tyrosine that involves in the desensitization of FGFR. You overexpress FGFR that cannot be phosphorylated the tyrosine because the tyrosine has been mutated to a phenylanaline. You observe that the cells grow much more faster than normal when treated with growth factor. Explain. Molecular Cell Biology, 7th Edition Lodish et al. Copyright © 2013 by W. H. Freeman and Company