* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Amino Acids - Portal UniMAP
Survey
Document related concepts
Butyric acid wikipedia , lookup
Ribosomally synthesized and post-translationally modified peptides wikipedia , lookup
Catalytic triad wikipedia , lookup
Citric acid cycle wikipedia , lookup
Fatty acid metabolism wikipedia , lookup
Fatty acid synthesis wikipedia , lookup
Metalloprotein wikipedia , lookup
Point mutation wikipedia , lookup
Nucleic acid analogue wikipedia , lookup
Proteolysis wikipedia , lookup
Peptide synthesis wikipedia , lookup
Protein structure prediction wikipedia , lookup
Genetic code wikipedia , lookup
Amino acid synthesis wikipedia , lookup
Transcript
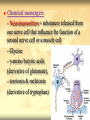
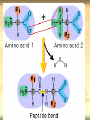
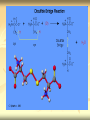
PTT103 BIOCHEMISTRY Amino Acids Pn Khadijah Hanim Abdul Rahman Introduction Proteins - the most structural, sophisticated molecules known - vary extensively in structure - are polymer constructed from the same set of 20 amino acids Polymer of amino acids = polypetides Proteins consists of 1 or more polypeptides folded and coiled into specific conformations Amino Acids structure Amino Acids contain a central carbon atom (α-carbon) to which an amino group, a carboxylate group, a hydrogen atom and an R (side chain) group are attached Amino acids shown in their prevailing ionic forms at pH7, approx pH within a cell Name and three-letter abbreviation of amino acid The exception : proline differ from other standard amino acid Its amino group is secondary Formed by ring closure between R group and the amino nitrogen Standard amino acid (primary amino group) Proline (secondary amino group) Amino acid structure The amino and carboxylic groups of amino acids readily ionized At physiological pH (7) - carboxyl group of an amino acid is unprotonated. conjugate base form (-COO-) - amino group of an amino acid is protonated. in its conjugate acid form (+NH3) Thus, each amino acid can behave as an acid or base referred as amphoteric (substance that can act as acid or base) Neutral molecules which bear an equal number of positive and negative charges simultaneously are called zwitterions The R group give each amino acid its unique properties Structure of amino acid at pH7 Amino acid classes Sequence of amino acids determines the threedimensional configuration of each protein Amino acids classified based on their capacity to interact with water 4 classes of amino acids : a) neutral nonpolar (Gly, Ala, Val, Leu, Ile, Phe, Trp, Met, Cys, Pro) b) neutral polar (ser, thr, tyr, asn, gln) c) acidic (Asp, glu) d) basic (lys, arg, his) a) Neutral nonpolar amino acids Term‘Neutral’ – contains hydrocarbon R groups- do not bear +ve or –ve charge So they interact poorly with water and play important role in maintaining the threedimensional structure of proteins 2 types of hydrocarbon R groups: a) aromatic (contain cyclic structure that constitute a class of unsaturated hydrocarbons) eg. Phenylalanine, tryptophan b) aliphatic (nonaromatic hydrocarbon) eg. Glycine, alanine, valine, leucine, isoleucine, proline, methionine, cysteine Methionine & cysteine contain sulfur atom in the aliphatic side chain In met, the nonbonding electrons of the sulfur atom can form bonds with electrophiles such as metal ions. The nonpolar sulfhydryl group (-SH) of cys can form a weak H bond with O or N. Sulfhydryl groups are highly reactiveimportant component in many enzymes. b) Neutral polar amino acids Polar amino acids = hydrophilic (water loving) Have functional group capable of forming hydrogen bonding, so easily interact with water Serine, threonine, tyrosine, asparagine, glutamine Serine, threonine, tyrosine - contain polar hydroxyl group (-OH) - Thus enable them to from hydrogen bonding (important factor in protein structure) Asparagine & glutamine - are amide derivatives of aspartic acid and glutamic acid (acidic amino acids) - amide funtional group are highly polar, so can form hydrogen bonding (effet on protein stability) c) Acidic amino acids Contain carboxylate R group The side chains of aspartic acid & glutamic acid are polar and negatively charged at physiological pH, so they often referred as aspartate and glutamate d) Basic amino acids Lysine, Arginine, Histidine Are polar and positively charge at physiological pH. Therefore can form ionic bonds with acidic amino acids. Lys contain amine R group which accepts a proton from water to form conjugate acid (-NH3+) His is a weak base because it partially ionized at pH 7. His act as buffer. Important role in catalytic activity of enzymes. Biologically active amino acids 1) 2) 3) The 20 standard amino acids undergo a bewildering number of chemical transformations. Many amino acids are synthesizes not to be residues of polypeptides but to function independently. Besides being components of protein, amino acids have several biological roles : Chemical messengers Precursors Metabolite intermediates Chemical messengers - Neurotransmitters = substances released from one nerve cell that influence the function of a second nerve cell or a muscle cell - Glycine - γ-amino butyric acids (derivative of glutamate), - serotonin & melatonin (derivative of tryptophan) - Hormones = chemical signal molecules produced in one cell that regulate the function of other cells - Thyroxine (tyrosine derivative), thyroid hormon secreted by thyroid gland - Indole acetic acid (tryptophan derivative), is an auxin plant hormones. Stimulate growth of the root Precursors - a compound that participate in the chemical reaction to produces another compound - amino acids are precursors of variety of complex nitrogen-containing molecules - Nitrogenous base components of nucleotides and nucleic acids, chlorophyll Metabolic intermediates - several amino acids act as metabolic intermediates - eg. Arginine, citrulline, ornithine (components of urea cycle) - The synthesis of urea, a molecule formed in the livers- principle mechanism for disposal of nitrogenous waste Amino acid stereoisomers α-carbon of 19 amino acids attached to 4 diff. groups, referred as asymmetric/chiral carbons Molecule with chiral carbon, can exist as stereoisomers Stereoisomers - isomeric molecule that have the same molecular formula - but differ only in the threedimensional orientations of their atoms in space. Molecules with chiral carbon are not superimposable on their mirror image in the same way that a left hand is not superimposable on its mirror image, a right hand They are known as enantiomers of one another L-Alanine and D-Alanine are mirror image to one another Glyceraldehyde is the reference compound for optical isomers (to differ between L and D) Most asymmetric molecules found in living organisms only occur in only 1 stereoisomeric form, either D or L. Chirality has had a profound effect on the structural and functional properties of biomolecules For eg: the right-handed helices result from the presence of L-amino acids. In addition, because enzymes are chiral molecules, they only bind substrate molecules in one enantiomeric form. Proteases, enzyme that degrade proteins by hydrolyzing peptide bonds, cannot degrade polypeptides composed of D-amino acids. Question Certain bacterial species have outer layers composed of polymers made of D-amino acids. Immune system cells, whose task is to attack and destroy foreign cells, cannot destroy those bacteria. Suggest a reason for this phenomenon. Titration of amino acids Amino acids contain ionizable group the predominant ionic form of amino acids in solution depends on pH Titration of amino acid : - illustrate the effect of pH on amino acids structure - a useful tool in determining the reactivity of amino acid side chains when amino acid is dissolved in water, it exist predominantly in the isoelectric form Upon titration with base, it act as an acid (donate proton) Upon titration with acid, it act as a base (accept proton) +H N-CH -COO3 2 + HCl +H3N-CH2-COOH + Cl(base) (acid) (1) +H N-CH -COO3 2 + NaOH H2N-CH2-COO- + Na+ +H2O (acid) (base) (2) Titration of alanine Consider alanine, a simple amino acid contain 2 titrable groups During titration with strong base, NaOH, alanine loses 2 protons. In a strongly acidic sol (pH 0), ala present mainly in the form of in which the carboxyl group is uncharged. At this point, glycine net charge = +1, because ammonium group is protonated. Lowering of the H+ conc results in the COOH group losing a proton to become COO-. At this point, ala has no net charge and electrically neutral. The pH at which this occur is called the isoelectric point (pI) Because no net charge at pI, amino acids are least soluble at this pH. The pI for ala may be calculated as: pI = pK1 + pK2 2 pK1 and pK2 of glycine are 2.34 and 9.7. The pI value for glycine : pI = 2.34 + 9.7 = 6 2 As the titration continues, the ammonium group will lose its proton, leaving an uncharged amino group This molecule has a net –ve charge because of the carboxylate group. Titration of glutamic acid with NaOH Amino acids with ionizable side chains (acidic & basic a.a) have more complex titration curve. For eg. Glutamic acid has a carboxyl side chain group (R group) At low pH, glutamic acid has net charge +1. As base added, the a-carboxyl group loses a proton to become carboxylate group. Glutame has no net charge. As more base added, the 2nd carboxyl group (side chain) loses a proton The molecule now has a net charge of -1 Adding more base, results in the ammonium ion loses its proton At this point, glutamate has a net charge of -2 The pI value for glutamate is the pH halfway between the pKa values for the two carboxyl group pI = 2.19 + 4.25 = 3.22 2 Note: pI is the pH at which amino acid has a net charge zero. For acidic amino acids pI = pK1 + pK2 2 For basic amino acids : pI = pK2 + pK3 2 Amino acid reactions The functional groups of organic molecules determine which reactions they may undergo. Amino acids with their COOH groups, amino groups and various R groups can undergo numerous chemical reactions. i.e peptide bond & disulfide bridge formation (effect protein structure) Peptide bond formation Polypeptides are linear polymers composed of amino acids linked together by peptide bonds Peptide bonds are amide linkage (CO-NH) formed when the carboxyl group of one amino acid react with amino group of another amino acid This reaction is a dehydration (mol. water is removed) So, the linked amino acids are referred to as amino acid residues. When two amino acid molecules are linked, the product is called a dipeptide. eg. Serine and glycine can form dipeptides glycylserine or serylglycine As amino acids are added and the chain lengthens, the prefix reflect the number of residues eg. Tripeptides contain three amino acid residues H O H O H O + + + H3N C C O H3N C C O H3N C C O R1 -H2O R1 -H2O R1 H O H O H O + H3N C C N C C N C C O H H R1 R1 R1 Peptide bonds Amino acid residue with the free amino group is called the N-terminal residue and is written to the left Amino acid residue with free carboxyl group is called C-terminal residue and is written to the right Peptides are named by using their amino acid sequence, start from their N-terminal residue eg. Alanylglycylphenilalanine Cysteine oxidation The sulfhydryl group of cysteine is highly reactive Common reaction = reversible oxidation that form disulfide Two molecules of cysteine oxidized to form a cystine (molecule that contain disulfide bond) When 2 cys residues form such bond, it is referred to as a disulfide bridge. Disulfide bridges help to stabilize many polypeptides and proteins.