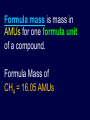* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Moles
Survey
Document related concepts
Depletion force wikipedia , lookup
Chemistry: A Volatile History wikipedia , lookup
Electrolysis of water wikipedia , lookup
Particle-size distribution wikipedia , lookup
IUPAC nomenclature of inorganic chemistry 2005 wikipedia , lookup
Elementary particle wikipedia , lookup
Size-exclusion chromatography wikipedia , lookup
History of chemistry wikipedia , lookup
Hydrogen atom wikipedia , lookup
Molecular dynamics wikipedia , lookup
Vapor–liquid equilibrium wikipedia , lookup
History of molecular theory wikipedia , lookup
Stoichiometry wikipedia , lookup
Transcript
The Mole How people see chemistry experimentally is different than how they see it theoretically. Theoretically we use atoms and molecules Experimentally we use mass and volume How did we solve this problem? The MOLE A little history related to the development of the mole While trying to find Chemical Formulas… Chemists Discovered a way of determining Atomic Masses & the Mole Chemists discovered that as long as temperature and pressure is kept constant gas rxns followed whole number ratios 2L Hydrogen 1L +1L Nitrogen Oxygen +3L 1 L Steam Hydrogen Ammonia 1L This helped determine chemical formulas as follows 2L Hydrogen +1L Oxygen 1 L Water Then Water is 1 H2O1 1L Nitrogen Ammonia +3L Hydrogen 1L Then Ammonia is 1 N1H3 To find formulas this way, Avogadro realized that you have to assume that the number of gas particles determines volume not how massive the particles are H2 O2 Cl2 In other words, 1 L of any gas has the same number of gas particles no matter what the composition of the gas. That allowed chemists to find relative atomic masses Chemists took equal volumes of gases and weighed them to get the relative atomic masses in AMUs. Relative atomic masses were developed: E.g. If Nitrogen weighs 14 times as much as Hydrogen. 1 atom Nitrogen = 14 amu 1 atom Hydrogen = 1 amu This is where the MOLE is created Chemists realized that they could keep the already established relative atomic masses, but change the unit to grams if they had enough volume. All they had to do is find that volume. What they found is that 22.4 L of a gas will allow you to keep the numerical value of the atomic weight, but the unit is now in grams. 22.4 L He = 4 grams He The Mole 22.4 L which was termed a MOLE is the amount of gas to make the AMUs convert directly to grams 22.4 L N2 = 1 mole N2 = 28 g N2 22.4 L only works at S.T.P. Standard Temp and Pressure STP is 25 °C and 1 atmosphere 22.4 L H2 @ STP = 1 mole H2 = 2.01 g H2 22.4 L N2 @ STP = 1 mole N2 = 28.0 g N2 22.4 L O2 @ STP = 1 mole O2 = 32.0 g O2 22.4 L CO2 @ STP = 1 mole CO2 = 44.0 g CO2 22.4 L Cl2 @ STP = 1 mole Cl2 = 71.0 g Cl2 NOTE: Mole is standing for a certain number of particles (atoms, molecules, etc). Chemists just did not know how many particles yet. At this point chemists Could find the formulas of compounds made from gases Had a way to measure atomic masses of anything that was a gas Had a way to relate Masses and Volumes to a Relative Number of Particles DO WS – Formula & Molar Mass Lab – Molar Mass Methane The Mole is essential to chemical manufacturing because the mole links the atomic scale to the human scale atoms & molecules mass & volume REVIEW OF MASS RELATED TERMINOLOGY Atomic mass mass of 1 atom in AMUs read from P.T. Atomic Mass C = 12.011 AMU H = 1.0079 AMU Formula mass is mass in AMUs for one formula unit of a compound. Formula Mass of CH4 = 16.05 AMUs Formula units represent the number of particles whether they are atoms, molecules or whole ionic compounds. Coefficients in a chemical reaction represent formula units E.g. 2 H2 + 1 C 1 CH4 A molar mass is the mass in grams of Avogadro’s # of the substance or 22.4 L of gas at S.T.P.. A mole is a more practical amount for use in experiments and industry Atomic mass of C = 12.011 amu Molar mass of C = 12.011 g Formula mass of CO2 = 44 amu Molar mass of CO2 = 44 g How many particles are in a mole (also known as 22.4 L of a gas at STP or the molar mass of a substance)? Through Pure Mathematics and later confirmed by expensive equipment we found…………. 1 Mole ____ = 6.022 x 1023 ____ 1 Mole C = 6.022 x 1023 atoms C 1 Mole NH3 = 6.022 x 1023 formula units NH3 1 Mole KCl = 6.022 x 1023 formula units KCl A mole is 6.022 x 10 something. 23 of It is a name for a number and the number of gas particles in 22.4 L of a gas!!!! 6.022 x is called 23 10 (NA) Avogadro’s Number So Again a Mole is ……. 1 mol __ = 6.02 x 1023__ = __ g__ and if it’s a gas the 1 mol ___ = 22.4 L of _____ @ STP Helpful reference chart Molecules F.U.s Atoms 1 mole 6.02 x 1023 6.02 x 1023 1 mole 1 mole ? grams Moles 22.4 L 1 mole Volume (Liters) 1 mole 22.4 L Mass (grams) ? grams 1 mole Practice Worksheets You own a New Neon sign store and are making a sign that will take 0.5 L of Ne. You call a gas supplying store and Ne costs $100 for 100 g. You charged $200 for the sign. The glass and electrical equipment cost $30. Did you make or lose money on the sign? Review Problems: Using Mole, Volume, Mass and Formula Units END