1411-Test2 - HCC Learning Web
... 1.Define the following types of reactions with an example for each: A. acid-base neutralization, ...
... 1.Define the following types of reactions with an example for each: A. acid-base neutralization, ...
Unit 15 Electrochemistry
... reactions to occur by driving the reaction in the opposite direction (basis of rechargeable batteries Take place in electrolytic cells Cathode is connected to – terminal to accept e-, anode is attached to + terminal to donate e(opposite of voltage) ...
... reactions to occur by driving the reaction in the opposite direction (basis of rechargeable batteries Take place in electrolytic cells Cathode is connected to – terminal to accept e-, anode is attached to + terminal to donate e(opposite of voltage) ...
powerpoint
... SURROUNDED BY WATER MOLECULES, THEY DISSOLVE AND ARE CALLED SOLUTES. – HYDROPHYLIC = “LIKE” WATER – HYDROPHOBIC = “FEAR” WATER Figure 0307 A CRYSTAL OF TABLE SALT DISSOLVING IN WATER Figure 0308 A WATER SOLUBLE PROTEIN ...
... SURROUNDED BY WATER MOLECULES, THEY DISSOLVE AND ARE CALLED SOLUTES. – HYDROPHYLIC = “LIKE” WATER – HYDROPHOBIC = “FEAR” WATER Figure 0307 A CRYSTAL OF TABLE SALT DISSOLVING IN WATER Figure 0308 A WATER SOLUBLE PROTEIN ...
What do you know about light?
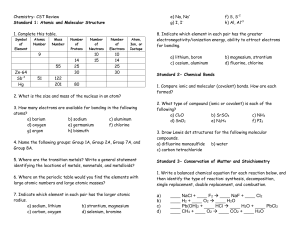
... • On its own the atomic number does not tell us the number of neutrons in an element. In order to determine the number of neutrons we need the mass number. ...
... • On its own the atomic number does not tell us the number of neutrons in an element. In order to determine the number of neutrons we need the mass number. ...
Honors Chemistry
... Uses - The main use of nitrogen lies in the fact that it is so unreactive. Many foods are packaged in nitrogen ( not air as it contains very reactive oxygen). Also used in aerosol cans ( replacing CFC’s ) Preparation Heating nitrogen containing compounds ammonium nitrite when heated yields nitrogen ...
... Uses - The main use of nitrogen lies in the fact that it is so unreactive. Many foods are packaged in nitrogen ( not air as it contains very reactive oxygen). Also used in aerosol cans ( replacing CFC’s ) Preparation Heating nitrogen containing compounds ammonium nitrite when heated yields nitrogen ...
9F Reactivity - Parrs Wood High School
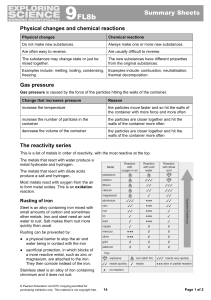
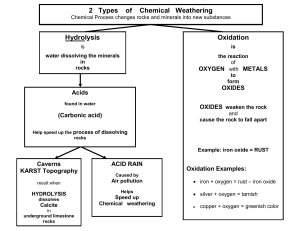
... Steel is an alloy containing iron mixed with small amounts of carbon and sometimes other metals. Iron and steel need air and water to rust. Salt makes them rust more quickly than usual. Rusting can be prevented by: ● a physical barrier to stop the air and water being in contact with the iron ● sacri ...
... Steel is an alloy containing iron mixed with small amounts of carbon and sometimes other metals. Iron and steel need air and water to rust. Salt makes them rust more quickly than usual. Rusting can be prevented by: ● a physical barrier to stop the air and water being in contact with the iron ● sacri ...
Chemistry- CST Review
... a) How many moles of O2, oxygen, are needed to produce 4.50 moles of CaO, calcium oxide? b) How many grams of Ca, calcium, would be needed to make 14.5 g of CaO, calcium oxide? ...
... a) How many moles of O2, oxygen, are needed to produce 4.50 moles of CaO, calcium oxide? b) How many grams of Ca, calcium, would be needed to make 14.5 g of CaO, calcium oxide? ...
chemistry basics note - bramalea2010-msmanning
... Draw and label Fig 7 below to show hydrogen bonding in water molecules. ...
... Draw and label Fig 7 below to show hydrogen bonding in water molecules. ...
Chemistry IGCSE Revision PDF File
... If a metal is more reactive than hydrogen its ions stay in solution and hydrogen bubbles off ...
... If a metal is more reactive than hydrogen its ions stay in solution and hydrogen bubbles off ...
AP Chemistry Summer Assignment 2016
... b. How many grams of calcium oxide will be produced after 12.25 grams of calcium carbonate are completely decomposed? c. What volume of carbon dioxide gas is produced from 12.25 grams of calcium carbonate at STP? d. What is the volume of carbon dioxide in L if the pressure is 785 mmHg and the temper ...
... b. How many grams of calcium oxide will be produced after 12.25 grams of calcium carbonate are completely decomposed? c. What volume of carbon dioxide gas is produced from 12.25 grams of calcium carbonate at STP? d. What is the volume of carbon dioxide in L if the pressure is 785 mmHg and the temper ...
Chemical Reactions
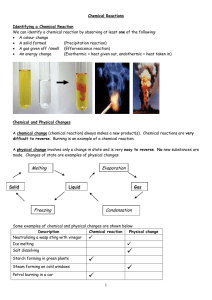
... The reactants are separated from each other by a plus sign and the products are separated from each other by a plus sign. There should be an arrow in the middle. Examples: When sodium is mixed with water, a purple alkaline solution of sodium hydroxide is produced and hydrogen gas is evolved. Sodium ...
... The reactants are separated from each other by a plus sign and the products are separated from each other by a plus sign. There should be an arrow in the middle. Examples: When sodium is mixed with water, a purple alkaline solution of sodium hydroxide is produced and hydrogen gas is evolved. Sodium ...
ELECTROANALYTICAL TECHNIQUES
... • Conc gradient created if ions do not rapidly migrate towards cathode • This gradient leads to resistance • This resistance due to changes in concentration of the electrolyte around the electrode is known as concentration polarisation. • occurs when the ions do not arrive at the cathode or the prod ...
... • Conc gradient created if ions do not rapidly migrate towards cathode • This gradient leads to resistance • This resistance due to changes in concentration of the electrolyte around the electrode is known as concentration polarisation. • occurs when the ions do not arrive at the cathode or the prod ...
NAME
... b) aluminum bromide solution and chlorine gas react to form aluminum chloride and bromine gas. ...
... b) aluminum bromide solution and chlorine gas react to form aluminum chloride and bromine gas. ...
Chemical Reactions
... only one in the products. To balance this we must insert a coefficient. H2 + ...
... only one in the products. To balance this we must insert a coefficient. H2 + ...
Unit 13 - Electrochemistry
... the relationship between electric forces and chemical reactions. Voltage: The potential difference or electromotive force, measured in volts; it represents the amount of work that moving an electric charge between two points would take. Electrode: A conductor used to establish electrical contact wit ...
... the relationship between electric forces and chemical reactions. Voltage: The potential difference or electromotive force, measured in volts; it represents the amount of work that moving an electric charge between two points would take. Electrode: A conductor used to establish electrical contact wit ...
Name__________________________ Period_______ Word
... Chemical formulas are used to represent the products and reactants in a reaction. Equations can also contain much more information. Conditions under which a reaction occurs are often found above the arrow. An example of a reaction condition is the heat symbol (∆ ), which indicates that the reactants ...
... Chemical formulas are used to represent the products and reactants in a reaction. Equations can also contain much more information. Conditions under which a reaction occurs are often found above the arrow. An example of a reaction condition is the heat symbol (∆ ), which indicates that the reactants ...
Intro to Soln Stoich
... ◦ O-H bonds are covalent, e- not shared equally ◦ Oxygen has a slight negative, hydrogen slight positive Polar molecule Oxygen has a strong attraction to cations, hydrogen to anions ...
... ◦ O-H bonds are covalent, e- not shared equally ◦ Oxygen has a slight negative, hydrogen slight positive Polar molecule Oxygen has a strong attraction to cations, hydrogen to anions ...
Chapter 9 Outline
... Heat of Fusion Latent heat – the energy required to cause a phase change. Heat of fusion – Energy needed to change 1.0g of a substance from solid to liquid phase. When the situation is reversed, and the liquid phase becomes a sold, this heat energy is released. ...
... Heat of Fusion Latent heat – the energy required to cause a phase change. Heat of fusion – Energy needed to change 1.0g of a substance from solid to liquid phase. When the situation is reversed, and the liquid phase becomes a sold, this heat energy is released. ...
IGCSE Revision document
... If a metal is more reactive than hydrogen its ions stay in solution and hydrogen bubbles off ...
... If a metal is more reactive than hydrogen its ions stay in solution and hydrogen bubbles off ...
X CHEMISTRY-1-2006 TIME-1½ MARKS 80 SECTION
... a. The important of arsenic oxide must be removed before passing the mixture of sulphnr Dioxide and air though the catalytic chamber b. For the production of concentrated sulphuric acid, sulphur trioxide is not direct by dissolve in water. c. Name the distance in which sulphur trionide is disowned. ...
... a. The important of arsenic oxide must be removed before passing the mixture of sulphnr Dioxide and air though the catalytic chamber b. For the production of concentrated sulphuric acid, sulphur trioxide is not direct by dissolve in water. c. Name the distance in which sulphur trionide is disowned. ...
Electrolysis of water
Electrolysis of water is the decomposition of water (H2O) into oxygen (O2) and hydrogen gas (H2) due to an electric current being passed through the water.This technique can be used to make hydrogen fuel (hydrogen gas) and breathable oxygen; though currently most industrial methods make hydrogen fuel from natural gas instead.