chapter 5 gases
... (c) is a weak electrolyte. A small amount of the compound dissociates into ions in water. When NaCl dissolves in water it dissociates into Na and Cl ions. When the ions are hydrated, the water molecules will be oriented so that the negative end of the water dipole interacts with the positive sodiu ...
... (c) is a weak electrolyte. A small amount of the compound dissociates into ions in water. When NaCl dissolves in water it dissociates into Na and Cl ions. When the ions are hydrated, the water molecules will be oriented so that the negative end of the water dipole interacts with the positive sodiu ...
Chemistry Midterm Exam Review
... ____ 46. A change in the force of gravity on an object will affect its a. mass. c. weight. b. density. d. kinetic energy. ____ 47. Which of these is a measure of the amount of material? a. density c. volume b. weight d. mass ____ 48. Which of these statements about mass is true? a. Mass is expressed ...
... ____ 46. A change in the force of gravity on an object will affect its a. mass. c. weight. b. density. d. kinetic energy. ____ 47. Which of these is a measure of the amount of material? a. density c. volume b. weight d. mass ____ 48. Which of these statements about mass is true? a. Mass is expressed ...
The role of aqueous-phase oxidation in the A
... Atmospheric particulate matter (or "aerosol") is known to have important implications for cli- ...
... Atmospheric particulate matter (or "aerosol") is known to have important implications for cli- ...
High Temperature Corrosion of Stainless Steels in Low Oxygen
... providing knowledge about the corrosion attack/mechanism. In order to better understand the complex corrosion reactions that occurred in the field exposures, laboratory exposures were performed in more simplified and well-controlled environments. Since H2 and H2O can be found in many energy systems, ...
... providing knowledge about the corrosion attack/mechanism. In order to better understand the complex corrosion reactions that occurred in the field exposures, laboratory exposures were performed in more simplified and well-controlled environments. Since H2 and H2O can be found in many energy systems, ...
Multiple Choice Exam Review June 2016
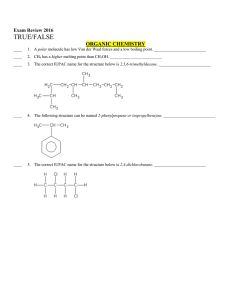
... ____ 14. All of the valence electrons in Fe2+ must have the same spin. _________________________ ____ 15. The shape of boron trifluoride, BF3, is tetrahedral. ______________________________ ____ 16. VSEPR theory predicts molecular shapes based on keeping protons as far apart as possible. ___________ ...
... ____ 14. All of the valence electrons in Fe2+ must have the same spin. _________________________ ____ 15. The shape of boron trifluoride, BF3, is tetrahedral. ______________________________ ____ 16. VSEPR theory predicts molecular shapes based on keeping protons as far apart as possible. ___________ ...
Problem Set 7
... an atom and where those particles are found. Define amu, too! The proton has a mass of 1 amu, the electron has a mass near 1/2000th an amu, the neutron has a mass of 1 amu. The protons and neutrons are typically found in the nucleus and they make up most of the mass of the atom. The electrons are ty ...
... an atom and where those particles are found. Define amu, too! The proton has a mass of 1 amu, the electron has a mass near 1/2000th an amu, the neutron has a mass of 1 amu. The protons and neutrons are typically found in the nucleus and they make up most of the mass of the atom. The electrons are ty ...
Chm 2
... d. 11.0 mol For the reaction represented by the equation Cl2 + 2KBr 2KCl + Br2, how many moles of potassium chloride are produced from 119 g of potassium bromide? a. 0.119 mol c. 0.581 mol b. 0.236 mol d. 1.00 mol For the reaction represented by the equation 3Fe + 4H2O Fe3O4 + 4H2, how many mole ...
... d. 11.0 mol For the reaction represented by the equation Cl2 + 2KBr 2KCl + Br2, how many moles of potassium chloride are produced from 119 g of potassium bromide? a. 0.119 mol c. 0.581 mol b. 0.236 mol d. 1.00 mol For the reaction represented by the equation 3Fe + 4H2O Fe3O4 + 4H2, how many mole ...
Preparation of Reducing Sugar Hydrolyzed from High
... crystalline orientation of cellulose, thus improving the yield of fermentable sugar in enzymatic hydrolysis [2-4]. Chemical methods have been employed to carry out pretreatment on grape stalks to regain both hemicellulose and cellulose [5]. However, the wastewater, which contained chemical substance ...
... crystalline orientation of cellulose, thus improving the yield of fermentable sugar in enzymatic hydrolysis [2-4]. Chemical methods have been employed to carry out pretreatment on grape stalks to regain both hemicellulose and cellulose [5]. However, the wastewater, which contained chemical substance ...
PART 3-ICHO 11-15
... washed, dried and calcinated. The mass of the precipitate after the calcination to constant mass, was 0.3265 g. An aqueous ammonia solution was added in excess to the solution obtained after separation of the precipitate. A compound of metal B remained in the solution while all the other metals prec ...
... washed, dried and calcinated. The mass of the precipitate after the calcination to constant mass, was 0.3265 g. An aqueous ammonia solution was added in excess to the solution obtained after separation of the precipitate. A compound of metal B remained in the solution while all the other metals prec ...
Unit 7 Reaction Rates and Equilibrium Notes
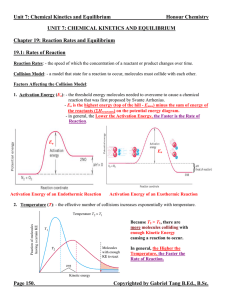
... Equilibrium Expression: - an expression relating the concentrations or pressures of the reactants and products when they are at the state of equilibrium. - it takes the form of the individual products raised to the power of their respective coefficients divided by the individual reactants raised to ...
... Equilibrium Expression: - an expression relating the concentrations or pressures of the reactants and products when they are at the state of equilibrium. - it takes the form of the individual products raised to the power of their respective coefficients divided by the individual reactants raised to ...
Reactions in Aqueous Solution
... the solvent3. An aqueous solution4 is a solution in which the solvent is water, whereas in a nonaqueous solution, the solvent is a substance other than water. Familiar examples of nonaqueous solvents are ethyl acetate, used in nail polish removers, and turpentine, used to clean paint brushes. In thi ...
... the solvent3. An aqueous solution4 is a solution in which the solvent is water, whereas in a nonaqueous solution, the solvent is a substance other than water. Familiar examples of nonaqueous solvents are ethyl acetate, used in nail polish removers, and turpentine, used to clean paint brushes. In thi ...
Chapter 4 "Reactions in Aqueous Solution"
... the solvent3. An aqueous solution4 is a solution in which the solvent is water, whereas in a nonaqueous solution, the solvent is a substance other than water. Familiar examples of nonaqueous solvents are ethyl acetate, used in nail polish removers, and turpentine, used to clean paint brushes. In thi ...
... the solvent3. An aqueous solution4 is a solution in which the solvent is water, whereas in a nonaqueous solution, the solvent is a substance other than water. Familiar examples of nonaqueous solvents are ethyl acetate, used in nail polish removers, and turpentine, used to clean paint brushes. In thi ...
Chemistry – A Molecular Sciences Appendices
... Notice that we did the operation on the numbers (six times twelve divided by one equals seventy-two) and on the units as well (feet times inches divided by feet equals inches; feet “cancel out”). Both the number and the unit changed, but the height did not (going from six feet to 72 inches, the pers ...
... Notice that we did the operation on the numbers (six times twelve divided by one equals seventy-two) and on the units as well (feet times inches divided by feet equals inches; feet “cancel out”). Both the number and the unit changed, but the height did not (going from six feet to 72 inches, the pers ...
Atmospheric Formation_TELTEK
... 2.2 (CH3)2NH Atkinson et al.19 studied the kinetics of the OH radical reaction with (CH3)2NH over the temperature range 299 – 426 K and reported a negative Arrhenius activation energy, kOH(T) = 2.89×10-11 × exp{(245 ± 150)K/T} and kOH = (6.54 ± 0.66) × 10-11 cm3 molecule-1 s-1 at 298 K. Carl and Cro ...
... 2.2 (CH3)2NH Atkinson et al.19 studied the kinetics of the OH radical reaction with (CH3)2NH over the temperature range 299 – 426 K and reported a negative Arrhenius activation energy, kOH(T) = 2.89×10-11 × exp{(245 ± 150)K/T} and kOH = (6.54 ± 0.66) × 10-11 cm3 molecule-1 s-1 at 298 K. Carl and Cro ...
Chem Agenda+ETDsHWK to End of Year 102714 Update
... Day 3: Atomic Theory Dalton-Thomson-Rutherford: BBs and Marbles Build your own super model Target: I can explain how to find the subatomic particle composition of any atom or isotope. A. Give the subatomic particle composition for Li, Mg, and F B. Why does atomic mass of C = 12.011? B. Reading the b ...
... Day 3: Atomic Theory Dalton-Thomson-Rutherford: BBs and Marbles Build your own super model Target: I can explain how to find the subatomic particle composition of any atom or isotope. A. Give the subatomic particle composition for Li, Mg, and F B. Why does atomic mass of C = 12.011? B. Reading the b ...
IIT-JEE (Advanced) - Brilliant Public School Sitamarhi
... If the amount of O2 evolved was 146.8 ml at S.T.P., calculate the % by weight of KClO4 in the residue. Q.13 A sample of calcium carbonate contains impurities which do not react with a mineral acid. When 2 grams of the sample were reacted with the mineral acid, 375 ml of carbon dioxide were obtained ...
... If the amount of O2 evolved was 146.8 ml at S.T.P., calculate the % by weight of KClO4 in the residue. Q.13 A sample of calcium carbonate contains impurities which do not react with a mineral acid. When 2 grams of the sample were reacted with the mineral acid, 375 ml of carbon dioxide were obtained ...
Chapter 4
... Strategy: Hydrogen displacement: Any metal above hydrogen in the activity series will displace it from water or from an acid. Metals below hydrogen will not react with either water or an acid. Solution: Only (b) Li and (d) Ca are above hydrogen in the activity series, so they are the only metals in ...
... Strategy: Hydrogen displacement: Any metal above hydrogen in the activity series will displace it from water or from an acid. Metals below hydrogen will not react with either water or an acid. Solution: Only (b) Li and (d) Ca are above hydrogen in the activity series, so they are the only metals in ...
2 - Montville.net
... world of atoms and molecules to the real world of grams . Stoichiometry problems are classified between the information given in the problem and the information you are expected to find, the unknown. The given and the unknown may be expressed in grams or ...
... world of atoms and molecules to the real world of grams . Stoichiometry problems are classified between the information given in the problem and the information you are expected to find, the unknown. The given and the unknown may be expressed in grams or ...
SCH3U: Final Exam Review
... ____ iii. When the volume of a gas is decreased by a factor of 5, the temperature is decreased by a factor of 5. ____ iv. When the pressure of a gas is halved, the temperature is halved. ____ v. When the volume of a gas is increased by a factor of 5,the temperature is decreased by a factor of 5. B) ...
... ____ iii. When the volume of a gas is decreased by a factor of 5, the temperature is decreased by a factor of 5. ____ iv. When the pressure of a gas is halved, the temperature is halved. ____ v. When the volume of a gas is increased by a factor of 5,the temperature is decreased by a factor of 5. B) ...
Chapter 12
... If we had the specified amount of all ingredients listed, could we make 4 dozen cookies? What if we had 6 eggs and twice as much of everything else, could we make 9 dozen cookies? What if we only had one egg, could we make 3 dozen cookies? ...
... If we had the specified amount of all ingredients listed, could we make 4 dozen cookies? What if we had 6 eggs and twice as much of everything else, could we make 9 dozen cookies? What if we only had one egg, could we make 3 dozen cookies? ...
Document
... pizzas, we burn a pizza, drop one on the floor, or other uncontrollable events happen so that we only make two pizzas. The actual amount of product made in a chemical reaction is called the actual yield. We can determine the efficiency of making pizzas by calculating the percentage of the maximum nu ...
... pizzas, we burn a pizza, drop one on the floor, or other uncontrollable events happen so that we only make two pizzas. The actual amount of product made in a chemical reaction is called the actual yield. We can determine the efficiency of making pizzas by calculating the percentage of the maximum nu ...
Chapter 4 Chemical Quantities and Aqueous Reactions
... it also determines the amounts of the other ingredients we will use! ...
... it also determines the amounts of the other ingredients we will use! ...
Chapter 4 Chemical Quantities and Aqueous Reactions
... pizzas, we burn a pizza, drop one on the floor, or other uncontrollable events happen so that we only make two pizzas. The actual amount of product made in a chemical reaction is called the actual yield. We can determine the efficiency of making pizzas by calculating the percentage of the maximum nu ...
... pizzas, we burn a pizza, drop one on the floor, or other uncontrollable events happen so that we only make two pizzas. The actual amount of product made in a chemical reaction is called the actual yield. We can determine the efficiency of making pizzas by calculating the percentage of the maximum nu ...
Solutions - ChemConnections
... Mass balance indicates that we have the same number and type of atoms on both sides of the equation (so that mass is conserved). Similarly, net charge must also be conserved. We cannot have a buildup of charge on one side of the reaction or the other. In redox reactions, electrons are used to balanc ...
... Mass balance indicates that we have the same number and type of atoms on both sides of the equation (so that mass is conserved). Similarly, net charge must also be conserved. We cannot have a buildup of charge on one side of the reaction or the other. In redox reactions, electrons are used to balanc ...
Instructor`s Resource Manual
... emphasis on descriptive chemistry is necessary to provide the motivation for learning chemical concepts. We have also added Concept Checks and Conceptual Problems to aid the student in learning the concepts. In these, we ask students questions that require them to think and to solve problems by firs ...
... emphasis on descriptive chemistry is necessary to provide the motivation for learning chemical concepts. We have also added Concept Checks and Conceptual Problems to aid the student in learning the concepts. In these, we ask students questions that require them to think and to solve problems by firs ...