Test Review Answers File
... Hydrogen, lithium, sodium, potassium, rubidium, cesium, francium 8. Why are the Noble Gases considered “inert” gases? List all the elements in this group. -undergo fewest chemical reactions, low chemical reactivity -helium, neon, argon, krypton, xenon, radon 9. List which elements are chemically sim ...
... Hydrogen, lithium, sodium, potassium, rubidium, cesium, francium 8. Why are the Noble Gases considered “inert” gases? List all the elements in this group. -undergo fewest chemical reactions, low chemical reactivity -helium, neon, argon, krypton, xenon, radon 9. List which elements are chemically sim ...
How to write up a practical: General review
... Objectives TO KNOW the terms protons, electrons and neutrons and some of their properties TO BE ABLE draw the basic structure of the atom. TO UNDERSTAND how these particles are physically arranged in relation to each other. ...
... Objectives TO KNOW the terms protons, electrons and neutrons and some of their properties TO BE ABLE draw the basic structure of the atom. TO UNDERSTAND how these particles are physically arranged in relation to each other. ...
Chapter 1: The Mole
... Mass Number (A) Atomic Number (Z) Z = number of protons. A = Z + number of neutrons. ...
... Mass Number (A) Atomic Number (Z) Z = number of protons. A = Z + number of neutrons. ...
Atoms, Molecules and Ions In This Chapter
... particles, which we now know of as the electron. • Thomson determined the charge to mass ratio for the electron and proposed a model for the atom called the plum pudding atom. • Thomson won the Nobel Prize for Physics in ...
... particles, which we now know of as the electron. • Thomson determined the charge to mass ratio for the electron and proposed a model for the atom called the plum pudding atom. • Thomson won the Nobel Prize for Physics in ...
Chapter 3: The Structure of Matter
... •There are more than 118 known elements; 92 are natural elements •A natural element is one that is found in nature ...
... •There are more than 118 known elements; 92 are natural elements •A natural element is one that is found in nature ...
Science - Atomic Structure
... • Review (from grade 5): Structure of atoms: protons, neutron, electrons Molecules Compounds are formed by combining two or more elements and have properties different from the constituent elements. ...
... • Review (from grade 5): Structure of atoms: protons, neutron, electrons Molecules Compounds are formed by combining two or more elements and have properties different from the constituent elements. ...
Comprehensive Science 3 Module 4 Practice Test
... Atoms of different elements combine to make different compounds 5. The elements listed at the far right side of the periodic table are _______. Metalloids Nonmetals Metals Transitional Metals ...
... Atoms of different elements combine to make different compounds 5. The elements listed at the far right side of the periodic table are _______. Metalloids Nonmetals Metals Transitional Metals ...
Element: a pure, simple substance that can`t be broken down into
... What is the smallest unit of matter that we can find everywhere, even in tuna fish? What charge do electrons have? What are elements? Who organized the atomic elements? What do we call a horizontal row on the periodic table? What do we call the vertical columns on the periodic table? The number of p ...
... What is the smallest unit of matter that we can find everywhere, even in tuna fish? What charge do electrons have? What are elements? Who organized the atomic elements? What do we call a horizontal row on the periodic table? What do we call the vertical columns on the periodic table? The number of p ...
Atomic Structure ppt
... • Protons and neutrons have essentially the same mass. • The mass of an electron is so small we ignore it. ...
... • Protons and neutrons have essentially the same mass. • The mass of an electron is so small we ignore it. ...
Lecture 2
... protons in an atom (as in a nuclear reaction) changes the element. • While atoms of the same element must have the same atomic number, they may have different mass numbers. If so, they are referred to as isotopes. Most elements have more than one naturally occurring isotope: ...
... protons in an atom (as in a nuclear reaction) changes the element. • While atoms of the same element must have the same atomic number, they may have different mass numbers. If so, they are referred to as isotopes. Most elements have more than one naturally occurring isotope: ...
Chemical Bonds
... made up of tiny particles called atoms can exist in the form of elements and compounds Copper, iron, and lead are elements that can exist by themselves. ...
... made up of tiny particles called atoms can exist in the form of elements and compounds Copper, iron, and lead are elements that can exist by themselves. ...
Elementary my dear Watson review
... The rows are called periods and they are arranged by atomic number. We use numbers to identify them. The columns are called families. Each column contains elements that have similar properties. The elements are metals, non-metals or metalloids (having both properties of metals and non-metals). ...
... The rows are called periods and they are arranged by atomic number. We use numbers to identify them. The columns are called families. Each column contains elements that have similar properties. The elements are metals, non-metals or metalloids (having both properties of metals and non-metals). ...
Review Notes - Biochemistry
... The goal of all atoms is to have a _STABLE_ outer energy level. The goal leads to bonding of atoms. 2 types of bonding: 1. Ionic Bonding: When _1_ or more electrons are _TRANSFERRED_ from one atom to another. Ion: an atom with a_CHARGE_. When an electron is gained it will be _NEGATIVE_charged ...
... The goal of all atoms is to have a _STABLE_ outer energy level. The goal leads to bonding of atoms. 2 types of bonding: 1. Ionic Bonding: When _1_ or more electrons are _TRANSFERRED_ from one atom to another. Ion: an atom with a_CHARGE_. When an electron is gained it will be _NEGATIVE_charged ...
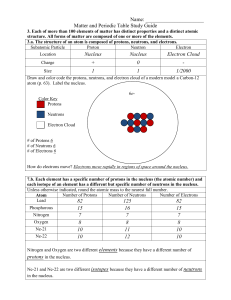
Matter and the Periodic Table Study Guide Answer Key
... Semimetals/Metalloids have properties of both metals and non-metals. 3.b. Compounds are formed by combining two or more different elements and compounds have properties that are different from their constituent elements. 3.f. Use the periodic table to identify elements in simple compounds. Compound ...
... Semimetals/Metalloids have properties of both metals and non-metals. 3.b. Compounds are formed by combining two or more different elements and compounds have properties that are different from their constituent elements. 3.f. Use the periodic table to identify elements in simple compounds. Compound ...
Chapter 5 - Effingham County Schools
... nucleus, called the ________ ________. For example, a hydrogen atom has 1 proton so its atomic number is 1. The total number of _______ and _________ in an atom’s nucleus is called its atomic mass number. _______ are atoms of the same element that have a different number of neutrons. Ions are formed ...
... nucleus, called the ________ ________. For example, a hydrogen atom has 1 proton so its atomic number is 1. The total number of _______ and _________ in an atom’s nucleus is called its atomic mass number. _______ are atoms of the same element that have a different number of neutrons. Ions are formed ...
04 Atoms_ molecules _ ions
... • Review PPs 1 – 4 • Work the attached assignment & turn it in tomorrow. ...
... • Review PPs 1 – 4 • Work the attached assignment & turn it in tomorrow. ...
Chemistry: The Nature of Matter
... Electron configuration – where electrons are within the atom Example: Oxygen – atomic number of 8, with 6 valance electrons ...
... Electron configuration – where electrons are within the atom Example: Oxygen – atomic number of 8, with 6 valance electrons ...
File
... The properties of a compound are unique and differ from the elements that make up the compound. A chemical equation is a written representation of the process that occurs in a chemical reaction. A chemical equation is written with the reactants on the left side of an arrow (yield symbol) and the pro ...
... The properties of a compound are unique and differ from the elements that make up the compound. A chemical equation is a written representation of the process that occurs in a chemical reaction. A chemical equation is written with the reactants on the left side of an arrow (yield symbol) and the pro ...
1 - cloudfront.net
... How is the number of neutrons in the nucleus of an atom calculated? All atoms are neutral, with the number of protons equaling the ___. Isotopes of the same element have different _____. Using the periodic table, determine the number of neutrons in 16O. What does the number 84 represent in the name ...
... How is the number of neutrons in the nucleus of an atom calculated? All atoms are neutral, with the number of protons equaling the ___. Isotopes of the same element have different _____. Using the periodic table, determine the number of neutrons in 16O. What does the number 84 represent in the name ...
The Periodic Table OL Page 1 of 2 G. Galvin Name: Periodic Table
... periodic table -define atomic number (Z) and mass number (A) -define relative atomic mass (Ar) using 12C scale -define isotope -describe the composition of isotopes using hydrogen and carbon as an example -describe the organisation of particles in atoms of elements numbers 1-20 -classify the ...
... periodic table -define atomic number (Z) and mass number (A) -define relative atomic mass (Ar) using 12C scale -define isotope -describe the composition of isotopes using hydrogen and carbon as an example -describe the organisation of particles in atoms of elements numbers 1-20 -classify the ...
Atom The smallest piece of matter that still has the properties of the
... A table of the chemical elements arranged in order of atomic number, usually in rows, so that elements with similar atomic structure and similar chemical properties appear in vertical columns. ...
... A table of the chemical elements arranged in order of atomic number, usually in rows, so that elements with similar atomic structure and similar chemical properties appear in vertical columns. ...
Power point on the Periodic Table
... Vertical columns in the periodic table: group Numbered from 1 to 18 Elements in the same family (group) share similar physical and chemical properties The group also tells you the amount of valence electrons ...
... Vertical columns in the periodic table: group Numbered from 1 to 18 Elements in the same family (group) share similar physical and chemical properties The group also tells you the amount of valence electrons ...
Chemical element
A chemical element (or element) is a chemical substance consisting of atoms having the same number of protons in their atomic nuclei (i.e. the same atomic number, Z). There are 118 elements that have been identified, of which the first 94 occur naturally on Earth with the remaining 24 being synthetic elements. There are 80 elements that have at least one stable isotope and 38 that have exclusively radioactive isotopes, which decay over time into other elements. Iron is the most abundant element (by mass) making up the Earth, while oxygen is the most common element in the crust of the earth.Chemical elements constitute approximately 15% of the matter in the universe: the remainder is dark matter, the composition of it is unknown, but it is not composed of chemical elements.The two lightest elements, hydrogen and helium were mostly formed in the Big Bang and are the most common elements in the universe. The next three elements (lithium, beryllium and boron) were formed mostly by cosmic ray spallation, and are thus more rare than those that follow. Formation of elements with from six to twenty six protons occurred and continues to occur in main sequence stars via stellar nucleosynthesis. The high abundance of oxygen, silicon, and iron on Earth reflects their common production in such stars. Elements with greater than twenty six protons are formed by supernova nucleosynthesis in supernovae, which, when they explode, blast these elements far into space as planetary nebulae, where they may become incorporated into planets when they are formed.When different elements are chemically combined, with the atoms held together by chemical bonds, they form chemical compounds. Only a minority of elements are found uncombined as relatively pure minerals. Among the more common of such ""native elements"" are copper, silver, gold, carbon (as coal, graphite, or diamonds), and sulfur. All but a few of the most inert elements, such as noble gases and noble metals, are usually found on Earth in chemically combined form, as chemical compounds. While about 32 of the chemical elements occur on Earth in native uncombined forms, most of these occur as mixtures. For example, atmospheric air is primarily a mixture of nitrogen, oxygen, and argon, and native solid elements occur in alloys, such as that of iron and nickel.The history of the discovery and use of the elements began with primitive human societies that found native elements like carbon, sulfur, copper and gold. Later civilizations extracted elemental copper, tin, lead and iron from their ores by smelting, using charcoal. Alchemists and chemists subsequently identified many more, with almost all of the naturally-occurring elements becoming known by 1900. The properties of the chemical elements are summarized on the periodic table, which organizes the elements by increasing atomic number into rows (""periods"") in which the columns (""groups"") share recurring (""periodic"") physical and chemical properties. Save for unstable radioactive elements with short half-lives, all of the elements are available industrially, most of them in high degrees of purity.