The Proteasomes
... concurrently. The distance between these two sites would be thus bridged by 8-9 amino acids of the substrate protein. However, once cut, it is still not clear how the individual short peptides are removed from the chamber, although some have speculated the proteasome may have side windows that serve ...
... concurrently. The distance between these two sites would be thus bridged by 8-9 amino acids of the substrate protein. However, once cut, it is still not clear how the individual short peptides are removed from the chamber, although some have speculated the proteasome may have side windows that serve ...
the ubiquitin system and a putative stimulatory role
... regulating the degradation of specific proteins. Such a regulatory role is very important. Ub functions in an ATP-dependent fashion. But why is this? We don't need energy to hydrolyze proteins. The reason ATP is required is because machinery is needed to specifically target the proteins that need to ...
... regulating the degradation of specific proteins. Such a regulatory role is very important. Ub functions in an ATP-dependent fashion. But why is this? We don't need energy to hydrolyze proteins. The reason ATP is required is because machinery is needed to specifically target the proteins that need to ...
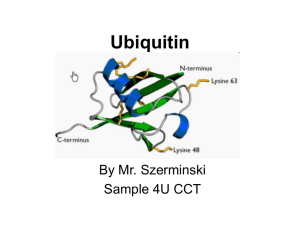
Ubiquitin
... Topics to be discussed • General info: - it is a regulatory protein that has been found in almost all tissues of eukaryotes - one of its functions: it directs protein recycling - can attach to proteins and label them for destruction. - discovery won the Nobel Prize for chemistry in 2004 ...
... Topics to be discussed • General info: - it is a regulatory protein that has been found in almost all tissues of eukaryotes - one of its functions: it directs protein recycling - can attach to proteins and label them for destruction. - discovery won the Nobel Prize for chemistry in 2004 ...
Leukaemia Section t(7;21)(p22;q22) Atlas of Genetics and Cytogenetics in Oncology and Haematology
... USP42 (ubiquitin specific protease 42), belongs to the ubiquitin specific protease family. Ubiquitins are highly conserved proteins. Ubiquitins target proteins for degradation in the proteasome. Some USPs, however, act in the opposite reaction. These ubiquitin specific proteases (cysteine proteases) ...
... USP42 (ubiquitin specific protease 42), belongs to the ubiquitin specific protease family. Ubiquitins are highly conserved proteins. Ubiquitins target proteins for degradation in the proteasome. Some USPs, however, act in the opposite reaction. These ubiquitin specific proteases (cysteine proteases) ...
3-in-1: A novel approach to study membrane protein pharmacology
... 3-in-1: A novel approach to study membrane protein pharmacology Membrane proteins make up about 25% of all proteins encoded by the human genome and are considered major drug targets. One type of membrane protein, the family of ligand-gated ion channels (LGICs), mediates crucial functions in the nerv ...
... 3-in-1: A novel approach to study membrane protein pharmacology Membrane proteins make up about 25% of all proteins encoded by the human genome and are considered major drug targets. One type of membrane protein, the family of ligand-gated ion channels (LGICs), mediates crucial functions in the nerv ...
Protein Degradation As discussed in last the last lecture, newly
... 2. E2- ubiquitin – an exchange reaction occurs, and the ubiquitin is transferred to a sulfhydryl group on E2, the ubiquitin-conjugating enzyme. The thioester bond is preserved and no ATP is required in this reaction. 3. E2-ubiquitin then interacts with E3, to which the substrate is specifically boun ...
... 2. E2- ubiquitin – an exchange reaction occurs, and the ubiquitin is transferred to a sulfhydryl group on E2, the ubiquitin-conjugating enzyme. The thioester bond is preserved and no ATP is required in this reaction. 3. E2-ubiquitin then interacts with E3, to which the substrate is specifically boun ...
Capturing denaturing proteins * Small Heat Shock Protein substrate
... network. Expression and/or mutation of sHSPs are linked to multiple diseases of protein misfolding, including neurodegenerative diseases, myopathies and cataract. The mechanism of sHSP chaperone action and interaction with substrates, therefore, has wide-ranging implications for understanding cellul ...
... network. Expression and/or mutation of sHSPs are linked to multiple diseases of protein misfolding, including neurodegenerative diseases, myopathies and cataract. The mechanism of sHSP chaperone action and interaction with substrates, therefore, has wide-ranging implications for understanding cellul ...
Introduction
... Turnover of cellular proteins was discovered in the 1930s in studies of Rudolf Schoenheimer, but it was in the 1960s that is became apparent that this was not just turnover, but a highly selective process. By the end of the 1970s two independent groups were working on two different topics: in the la ...
... Turnover of cellular proteins was discovered in the 1930s in studies of Rudolf Schoenheimer, but it was in the 1960s that is became apparent that this was not just turnover, but a highly selective process. By the end of the 1970s two independent groups were working on two different topics: in the la ...
Abstract I. DLC1 encodes a RhoA GTPase
... ligase (CRL4A) complex interaction with DDB1 and the FBXW5 substrate receptor. siRNA-mediated suppression of cullin 4A, DDB1, or FBXW5 expression restored DLC1 protein expression in NSCLC cell lines. FBXW5 suppression-induced DLC1 reexpression was associated with a reduction in the levels of activat ...
... ligase (CRL4A) complex interaction with DDB1 and the FBXW5 substrate receptor. siRNA-mediated suppression of cullin 4A, DDB1, or FBXW5 expression restored DLC1 protein expression in NSCLC cell lines. FBXW5 suppression-induced DLC1 reexpression was associated with a reduction in the levels of activat ...
Key concepts_Protein processing and modification
... complexes called translocons. A number of different mechanisms are employed in bacteria and eukaryotes. In particular, proteins can be translocated either directly from the ribosome, in cotranslational translocation, or from the cytoplasm, in post-translational translocation. The vast majority of pr ...
... complexes called translocons. A number of different mechanisms are employed in bacteria and eukaryotes. In particular, proteins can be translocated either directly from the ribosome, in cotranslational translocation, or from the cytoplasm, in post-translational translocation. The vast majority of pr ...
P024 Ubiquitin-independent in vitro degradation of nuclear hormone
... Nuclear Hormone Receptor (NRs), and its turnover, play a vital role in general gene regulation. Proteasome mediated degradation is the main protein turnover pathway in mammalian cells. Usually proteasome degradation requires previous poly-ubiquitylation of target proteins. We are interested in the d ...
... Nuclear Hormone Receptor (NRs), and its turnover, play a vital role in general gene regulation. Proteasome mediated degradation is the main protein turnover pathway in mammalian cells. Usually proteasome degradation requires previous poly-ubiquitylation of target proteins. We are interested in the d ...
intracellular protein synthesis, post
... Alfred L. G o l d b u . Dept. of Cell Biology, Harvard Medical School, Boston, MA 02115, U.S.A. ...
... Alfred L. G o l d b u . Dept. of Cell Biology, Harvard Medical School, Boston, MA 02115, U.S.A. ...
Abstract - BMB Reports
... signal in autophagy. We showed that many ER residents such as BiP contain evolutionally conserved arginylation permissive pro-N-degrons and that certain inducers like dsDNA or proteasome inhibitors cause their translocation into the cytoplasm where they bind midfolded proteins and undergo amino-term ...
... signal in autophagy. We showed that many ER residents such as BiP contain evolutionally conserved arginylation permissive pro-N-degrons and that certain inducers like dsDNA or proteasome inhibitors cause their translocation into the cytoplasm where they bind midfolded proteins and undergo amino-term ...
Lect21.RegulProtTurnover
... synthesis and degradation rates that determines the level of a protein in a cell, and changes in either rate can serve as means to regulate a protein’s concentration in the cell. ...
... synthesis and degradation rates that determines the level of a protein in a cell, and changes in either rate can serve as means to regulate a protein’s concentration in the cell. ...
Moonlighting and pleiotropy among regulators of the degradation
... features. Each of the two complexes is involved in a distinct cellular activity: the CSN promotes the function of a large family of ubiquitin E3 ligases known as CRLs, while the proteasome lid is required for ubiquitin hydrolysis from proteasome substrates before degradation. Both complexes determin ...
... features. Each of the two complexes is involved in a distinct cellular activity: the CSN promotes the function of a large family of ubiquitin E3 ligases known as CRLs, while the proteasome lid is required for ubiquitin hydrolysis from proteasome substrates before degradation. Both complexes determin ...
Student CSE paper
... intracellular quality control process by degrading mutated or abnormally folded proteins to prevent their accumulation as intracellular aggregates. Proteolysis by the ubiquitin/proteasome pathway involves two major steps: ubiquitination followed by degradation. A de-ubiquitination step also plays an ...
... intracellular quality control process by degrading mutated or abnormally folded proteins to prevent their accumulation as intracellular aggregates. Proteolysis by the ubiquitin/proteasome pathway involves two major steps: ubiquitination followed by degradation. A de-ubiquitination step also plays an ...
Proteasome

Proteasomes are protein complexes inside all eukaryotes and archaea, and in some bacteria. The main function of the proteasome is to degrade unneeded or damaged proteins by proteolysis, a chemical reaction that breaks peptide bonds.