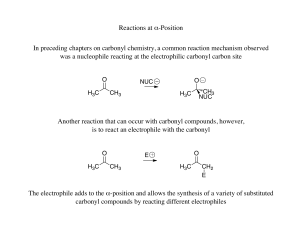
Organic synthesis and methodology related to the malaria drug artemisinin
... Figure 64: Initial Substrate Screening with MeCN ..................................... 121 Figure 65: Initial Substrate Screening with CH2Cl2 .................................... 122 Figure 66: Use of Electron-Rich Ethoxyacetylene ..................................... 123 Figure 67: Qualitative Scre ...
... Figure 64: Initial Substrate Screening with MeCN ..................................... 121 Figure 65: Initial Substrate Screening with CH2Cl2 .................................... 122 Figure 66: Use of Electron-Rich Ethoxyacetylene ..................................... 123 Figure 67: Qualitative Scre ...
Process for the preparation of ethene and/or propene
... specification No. 6 046 372. In the known process, the ...
... specification No. 6 046 372. In the known process, the ...
IOSR Journal of Applied Chemistry (IOSR-JAC)
... Items essentials for human survival such as, food, medicine etc. are produced by chemical reactions directly or indirectly in nature or in industry. Other items such as cosmetics, which are also deemed essentials in affluent societies, are also obtained by above means. It is the biggest challenge in ...
... Items essentials for human survival such as, food, medicine etc. are produced by chemical reactions directly or indirectly in nature or in industry. Other items such as cosmetics, which are also deemed essentials in affluent societies, are also obtained by above means. It is the biggest challenge in ...
Novel Transition Metal-Catalysed Syntheses of Carboxylic Acid
... The largest category of such reactions features the ruthenium metathesis catalysts developed by Grubbs and co-workers.[14] These alkylidene complexes are truly remarkable in terms of scope of their activity, so much so that an account of their nonmetathetic reactions has been recently published.[15] ...
... The largest category of such reactions features the ruthenium metathesis catalysts developed by Grubbs and co-workers.[14] These alkylidene complexes are truly remarkable in terms of scope of their activity, so much so that an account of their nonmetathetic reactions has been recently published.[15] ...
NAME Chem 204 Spring, 1990 Final Exam FINAL EXAM: Chaps. 1
... b) How many bonds exhibiting both restricted rotation and geometric isomerism are in (XII)? (XII) Thus, how many geometric isomers are possible because of restricted rotation? ...
... b) How many bonds exhibiting both restricted rotation and geometric isomerism are in (XII)? (XII) Thus, how many geometric isomers are possible because of restricted rotation? ...
Document
... • Always choose the longest chain that contains both atoms of the double bond. • In cycloalkenes, the double bond is located between C1 and C2, and the “1” is usually omitted in the name. • The ring is numbered clockwise or counterclockwise to give the first substituent the lower number. • Compounds ...
... • Always choose the longest chain that contains both atoms of the double bond. • In cycloalkenes, the double bond is located between C1 and C2, and the “1” is usually omitted in the name. • The ring is numbered clockwise or counterclockwise to give the first substituent the lower number. • Compounds ...
Alcohols
... Reaction is common in the laboratory and in living organisms • In the laboratory the reaction can be carried out in a single step if a strong acid is used as catalyst • The reactivity of the carboxylic acid is enhanced by first converting it into a carboxylic acid chloride, which then reacts with th ...
... Reaction is common in the laboratory and in living organisms • In the laboratory the reaction can be carried out in a single step if a strong acid is used as catalyst • The reactivity of the carboxylic acid is enhanced by first converting it into a carboxylic acid chloride, which then reacts with th ...
Ch. 6 - Department of Chemistry and Biochemistry
... Professor William Tam received his B.Sc. at the University of Hong Kong in 1990 and his Ph.D. at the University of Toronto (Canada) in 1995. He was an NSERC postdoctoral fellow at the Imperial College (UK) and at Harvard University (USA). He joined the Department of Chemistry at the University of Gu ...
... Professor William Tam received his B.Sc. at the University of Hong Kong in 1990 and his Ph.D. at the University of Toronto (Canada) in 1995. He was an NSERC postdoctoral fellow at the Imperial College (UK) and at Harvard University (USA). He joined the Department of Chemistry at the University of Gu ...
HIGHLY SELECTIVE RHODIUM–CATALYZED C–H BORYLATIONS IN
... First and foremost I am greatly indebted to my supervisor Dr. Cathleen Crudden. The time spent working with you helped me to better understand the trade while growing both as a scientist and a person. Leaving the lab, I feel my training and experiences have best prepared me for any future challenges ...
... First and foremost I am greatly indebted to my supervisor Dr. Cathleen Crudden. The time spent working with you helped me to better understand the trade while growing both as a scientist and a person. Leaving the lab, I feel my training and experiences have best prepared me for any future challenges ...
CHAPTER 1—COVALENT BONDING AND
... c. The carbon-carbon triple bond of an alkene is exactly three times as strong as a carbon-carbon single bond of an alkane. d. The sp3C H bond of an alkane is longer than the spC H bond of an alkyne. ...
... c. The carbon-carbon triple bond of an alkene is exactly three times as strong as a carbon-carbon single bond of an alkane. d. The sp3C H bond of an alkane is longer than the spC H bond of an alkyne. ...
synthesis and reactions of tris dialkyl dithiocarbamates of group 15
... phMe2, Et3, Me3 and But3; X = Cl, Br, or I); the bromide complexes in this series were found to be isostructural with their chloride analogues. However interpretation of the spectra of these complexes is much more problematic as the spectra are clearly not characteristic. This was obviously noted fo ...
... phMe2, Et3, Me3 and But3; X = Cl, Br, or I); the bromide complexes in this series were found to be isostructural with their chloride analogues. However interpretation of the spectra of these complexes is much more problematic as the spectra are clearly not characteristic. This was obviously noted fo ...
Oxidation of Reduced Sulfur Species: Carbon
... active. The abstraction path leading to OCS + O was characterized using the W1U method43 as implemented in the Gaussian 09 program.44 We modified this method to base it on geometry optimization and vibrational frequencies at the QCISD/6-311G(2d,d,p) level of theory, followed by several component step ...
... active. The abstraction path leading to OCS + O was characterized using the W1U method43 as implemented in the Gaussian 09 program.44 We modified this method to base it on geometry optimization and vibrational frequencies at the QCISD/6-311G(2d,d,p) level of theory, followed by several component step ...
Graphene-Catalyzed Direct Friedel–Crafts Alkylation Reactions
... recovered GO (pH = 3.95 at 0.29 mg mL−1) indicated the slightly acidic nature of the GO carbocatalyst, and no changes in the acidity after the reaction.48 (5) EDXS analysis (energydispersive X-ray spectroscopy) of the GO material before the reaction showed C/O atomic ratio of 1.95, which increased t ...
... recovered GO (pH = 3.95 at 0.29 mg mL−1) indicated the slightly acidic nature of the GO carbocatalyst, and no changes in the acidity after the reaction.48 (5) EDXS analysis (energydispersive X-ray spectroscopy) of the GO material before the reaction showed C/O atomic ratio of 1.95, which increased t ...
Recent developments in the applications of palladium complexes
... great interest due to their application in cross-coupling reactions and hence, is under continuous development. Palladium is, along with ruthenium, the most widely used transition metal with NHC ligands. Besides cross-coupling reactions, Pd-NHC systems display remarkable performances in many other o ...
... great interest due to their application in cross-coupling reactions and hence, is under continuous development. Palladium is, along with ruthenium, the most widely used transition metal with NHC ligands. Besides cross-coupling reactions, Pd-NHC systems display remarkable performances in many other o ...
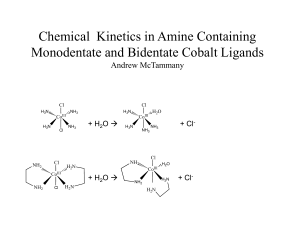
Chemical Kinetics in Monodentate and Bidentate Cobalt Compounds
... repeated. The trans-Co(NH3)4Cl2 should be synthesized using a lower temperature and lower concentration of acid. A mixture of HCl and H2SO4 could be used instead. From there the effect of different ligands can be evaluated. ...
... repeated. The trans-Co(NH3)4Cl2 should be synthesized using a lower temperature and lower concentration of acid. A mixture of HCl and H2SO4 could be used instead. From there the effect of different ligands can be evaluated. ...
Comparison of density functionals for energy and structural
... ? Fe(H2 O) 6 ? 2? , ?J. Chem. Phys. 120, 9473 ?2004?? to include a second compound—namely, the hexaminoferrous cation, ? Fe(NH3 ) 6 ? 2? —and several additional functionals. In particular, the present study includes the highly parametrized generalized gradient approximations ?GGAs? known as HCTH and ...
... ? Fe(H2 O) 6 ? 2? , ?J. Chem. Phys. 120, 9473 ?2004?? to include a second compound—namely, the hexaminoferrous cation, ? Fe(NH3 ) 6 ? 2? —and several additional functionals. In particular, the present study includes the highly parametrized generalized gradient approximations ?GGAs? known as HCTH and ...
Woodward–Hoffmann rules

The Woodward–Hoffmann rules, devised by Robert Burns Woodward and Roald Hoffmann, are a set of rules in organic chemistry predicting the barrier heights of pericyclic reactions based upon conservation of orbital symmetry. The Woodward–Hoffmann rules can be applied to understand electrocyclic reactions, cycloadditions (including cheletropic reactions), sigmatropic reactions, and group transfer reactions. Reactions are classified as allowed if the electronic barrier is low, and forbidden if the barrier is high. Forbidden reactions can still take place but require significantly more energy.The Woodward–Hoffmann rules were first formulated to explain the striking stereospecificity of electrocyclic reactions under thermal and photochemical control. Thermolysis of the substituted cyclobutene trans-1,2,3,4-tetramethylcyclobutene (1) gave only one diastereomer, the (E,E)-3,4-dimethyl-2,4-hexadiene (2) as shown below; the (Z,Z) and the (E,Z) diastereomers were not detected in the reaction. Similarly, thermolysis of cis-1,2,3,4-tetramethylcyclobutene (3) gave only the (E,Z) diastereomer (4).Due to their elegance and simplicity, the Woodward–Hoffmann rules are credited with first exemplifying the power of molecular orbital theory to experimental chemists. Hoffmann was awarded the 1981 Nobel Prize in Chemistry for this work, shared with Kenichi Fukui who developed a similar model using frontier molecular orbital (FMO) theory; because Woodward had died two years before, he was not eligible to win what would have been his second Nobel Prize for Chemistry.