Full-Text PDF
... mol % of tetrakis(acetonitrile)copper(I) triflate was able to efficiently catalyze the transformation of phenol 1 to the corresponding phenyl acetate 2 in 3 min and in quantitative yield. the model substrate and acetic anhydride (4 equivalents) as the acetylation agent, we found that 1 the model ...
... mol % of tetrakis(acetonitrile)copper(I) triflate was able to efficiently catalyze the transformation of phenol 1 to the corresponding phenyl acetate 2 in 3 min and in quantitative yield. the model substrate and acetic anhydride (4 equivalents) as the acetylation agent, we found that 1 the model ...
amines - Gneet`s
... Nitrogen atom of amino group is sp3 hybridised. Three of these orbitals get involved in sigma bond formation with other atoms where as the fourth orbital contains lone pair of electrons Due to presence of unshared pair of electron, the angle is less than 109.5o The angle around N atom in amine ...
... Nitrogen atom of amino group is sp3 hybridised. Three of these orbitals get involved in sigma bond formation with other atoms where as the fourth orbital contains lone pair of electrons Due to presence of unshared pair of electron, the angle is less than 109.5o The angle around N atom in amine ...
Full file at http://testbanksolution.eu/Test-Bank-Bank-for
... Full file at http://testbanksolution.eu/Test-Bank-Bank-for-Organic-Chemistry-7-E-by-Anslyn d. C2sp3 + H1s ANS: D 61. Which atomic orbitals overlap to form the carbon-hydrogen bonding molecular orbitals of ethene, H2C=CH2? a. C2p + H1s b. C2sp + H1s c. C2sp2 + H1s d. C2sp3 + H1s ANS: C 62. Which a ...
... Full file at http://testbanksolution.eu/Test-Bank-Bank-for-Organic-Chemistry-7-E-by-Anslyn d. C2sp3 + H1s ANS: D 61. Which atomic orbitals overlap to form the carbon-hydrogen bonding molecular orbitals of ethene, H2C=CH2? a. C2p + H1s b. C2sp + H1s c. C2sp2 + H1s d. C2sp3 + H1s ANS: C 62. Which a ...
Organic Chemistry II Introduction
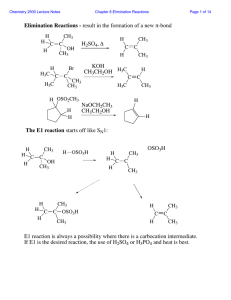
... • The one -step mechanisms is E2 reaction and two step reaction mechanism is called E1 reaction, respectively. Dr Seemal Jelani ...
... • The one -step mechanisms is E2 reaction and two step reaction mechanism is called E1 reaction, respectively. Dr Seemal Jelani ...
Glycosyl amines
... it leads to a delayed development of a tan. This melanogenesis-based tan takes more time to develop, but it is long lasting.[2] http://en.wikipedia.org/wiki/Melanin ...
... it leads to a delayed development of a tan. This melanogenesis-based tan takes more time to develop, but it is long lasting.[2] http://en.wikipedia.org/wiki/Melanin ...
Palladium(II)-Catalyzed Oxidative Cyclization Strategies Andreas K. Å. Persson
... palladium(II) readily forms π-complexes with a wide range of unsaturated hydrocarbons such as alkynes, alkenes and allenes. The coordination to palladium renders these fragments susceptible towards nucleophilic attack and/or migratory insertion. Many palladium(II)-catalyzed oxidative processes, suc ...
... palladium(II) readily forms π-complexes with a wide range of unsaturated hydrocarbons such as alkynes, alkenes and allenes. The coordination to palladium renders these fragments susceptible towards nucleophilic attack and/or migratory insertion. Many palladium(II)-catalyzed oxidative processes, suc ...
Boron and Metal Catalyzed CC and CH Bond Formation
... of these novel reactions have been developed. These include alternate routes to diphenylmethanes and 1,4-diynes from easily prepared dialkynylboron chlorides. In addition, E and Z alkenyl halides can now be prepared using boron trichloride without the use of butyllithium. The stereochemistry of the ...
... of these novel reactions have been developed. These include alternate routes to diphenylmethanes and 1,4-diynes from easily prepared dialkynylboron chlorides. In addition, E and Z alkenyl halides can now be prepared using boron trichloride without the use of butyllithium. The stereochemistry of the ...
lec-3- 211( Elim+ Re..
... catabolic metabolism of alcohol. It is commonly thought that the flush reaction is caused by an inability to metabolize alcohol. ...
... catabolic metabolism of alcohol. It is commonly thought that the flush reaction is caused by an inability to metabolize alcohol. ...
Enol esters: Versatile substrates in synthesis of fine and specialty
... As far as known, the only tandem process involving enol ester derivatives is the asymmetric hydroformylation of Z-enol acetates with rhodium(I) (S,S,S)-BDP catalysts yielding αacetoxcyaldehydes, which was first reported by both Roberto and Risi et al.9 They demonstrated that α-acetoxcyaldehydes can ...
... As far as known, the only tandem process involving enol ester derivatives is the asymmetric hydroformylation of Z-enol acetates with rhodium(I) (S,S,S)-BDP catalysts yielding αacetoxcyaldehydes, which was first reported by both Roberto and Risi et al.9 They demonstrated that α-acetoxcyaldehydes can ...
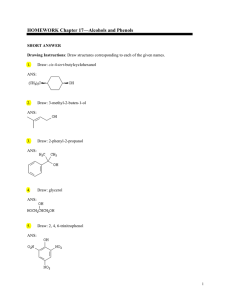
HOMEWORK Chapter 17—Alcohols and Phenols
... In E2 elimination, dehydration proceeds most readily when the two groups to be eliminated have a trans-diaxial relationship. In this compound, the only hydrogen with the proper geometric relationship to the −OH group is at C6 so the major product of this reaction is 3methylcyclohexene. ...
... In E2 elimination, dehydration proceeds most readily when the two groups to be eliminated have a trans-diaxial relationship. In this compound, the only hydrogen with the proper geometric relationship to the −OH group is at C6 so the major product of this reaction is 3methylcyclohexene. ...
Recent advances in homogeneous nickel catalysis
... systems capable of activating the C–O bonds of functional groups other than triflates—such as ethers, esters, carbamates and carbonates (Fig. 3c)—are highly desired. Reactions based on such catalyst systems would represent an ideal combination of ready access to coupling partners that can be mildly ...
... systems capable of activating the C–O bonds of functional groups other than triflates—such as ethers, esters, carbamates and carbonates (Fig. 3c)—are highly desired. Reactions based on such catalyst systems would represent an ideal combination of ready access to coupling partners that can be mildly ...
Anionic rearrangement of 2-benzyloxypyridine derivatives and a synthetic approach to aldingenin B
... TABLE OF CONTENTS List of Tables .......................................................................................................... vii List of Figures ........................................................................................................ viii Abstract .................... ...
... TABLE OF CONTENTS List of Tables .......................................................................................................... vii List of Figures ........................................................................................................ viii Abstract .................... ...
Advanced Practical Organic Chemistry
... potassium, manganese, sodium and chlorine, respectively. Molecules with Carbon Most atoms are only capable of forming small molecules. However, one or two can form larger molecules. By far and away the best atom for making large molecules with, is carbon. Carbon can make molecules that have tens, hu ...
... potassium, manganese, sodium and chlorine, respectively. Molecules with Carbon Most atoms are only capable of forming small molecules. However, one or two can form larger molecules. By far and away the best atom for making large molecules with, is carbon. Carbon can make molecules that have tens, hu ...
ch01
... a place where is negative. a place where is positive. a place where = 0. a place where 2 is large. a place where 2 is negative. Ans: C ...
... a place where is negative. a place where is positive. a place where = 0. a place where 2 is large. a place where 2 is negative. Ans: C ...
OChem1 Course Pack
... 9. (10 pts) Draw two chair conformations for cis-1-t-butyl-3-methylcyclohexane that are related through a ring-flip. Then circle which conformation you expect to be more stable and explain why. Then do the same for the trans isomer and, finally, indicate whether the cis or trans isomer should be mo ...
... 9. (10 pts) Draw two chair conformations for cis-1-t-butyl-3-methylcyclohexane that are related through a ring-flip. Then circle which conformation you expect to be more stable and explain why. Then do the same for the trans isomer and, finally, indicate whether the cis or trans isomer should be mo ...
Woodward–Hoffmann rules

The Woodward–Hoffmann rules, devised by Robert Burns Woodward and Roald Hoffmann, are a set of rules in organic chemistry predicting the barrier heights of pericyclic reactions based upon conservation of orbital symmetry. The Woodward–Hoffmann rules can be applied to understand electrocyclic reactions, cycloadditions (including cheletropic reactions), sigmatropic reactions, and group transfer reactions. Reactions are classified as allowed if the electronic barrier is low, and forbidden if the barrier is high. Forbidden reactions can still take place but require significantly more energy.The Woodward–Hoffmann rules were first formulated to explain the striking stereospecificity of electrocyclic reactions under thermal and photochemical control. Thermolysis of the substituted cyclobutene trans-1,2,3,4-tetramethylcyclobutene (1) gave only one diastereomer, the (E,E)-3,4-dimethyl-2,4-hexadiene (2) as shown below; the (Z,Z) and the (E,Z) diastereomers were not detected in the reaction. Similarly, thermolysis of cis-1,2,3,4-tetramethylcyclobutene (3) gave only the (E,Z) diastereomer (4).Due to their elegance and simplicity, the Woodward–Hoffmann rules are credited with first exemplifying the power of molecular orbital theory to experimental chemists. Hoffmann was awarded the 1981 Nobel Prize in Chemistry for this work, shared with Kenichi Fukui who developed a similar model using frontier molecular orbital (FMO) theory; because Woodward had died two years before, he was not eligible to win what would have been his second Nobel Prize for Chemistry.


![Chem Soc Rev - [ RSC ] Publishing](http://s1.studyres.com/store/data/022750285_1-eab192c7d8bd21532ac3979b6ccdf310-300x300.png)