* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download South Pasadena · AP Chemistry
Analytical chemistry wikipedia , lookup
Chemical thermodynamics wikipedia , lookup
Determination of equilibrium constants wikipedia , lookup
History of chemistry wikipedia , lookup
Debye–Hückel equation wikipedia , lookup
Dimensional analysis wikipedia , lookup
IUPAC nomenclature of inorganic chemistry 2005 wikipedia , lookup
Drug discovery wikipedia , lookup
Physical organic chemistry wikipedia , lookup
History of molecular theory wikipedia , lookup
Computational chemistry wikipedia , lookup
Isotopic labeling wikipedia , lookup
Mass spectrometry wikipedia , lookup
Size-exclusion chromatography wikipedia , lookup
Molecular dynamics wikipedia , lookup
Stoichiometry wikipedia , lookup
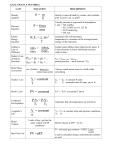
Culver City H.S. AP Chemistry Name __________________________________ Period ___ Date ___/___/___ 3 Stoichiometry STUDY LIST Atomic Mass Calculations: Hydrates: Know how a mass spectrometer is used for comparing the masses of atoms. Explain that a mole of any element is actually made up of various isotopes in a constant percentage abundance. Calculate the average atomic mass of an element using the percent abundance and mass of each isotope. Calculate the percent abundance of isotopes given the average atomic mass and isotopic masses of an element. Give examples of hydrates and anhydrous compounds. Calculate the formula of a hydrate from dehydration data. The Mole: State the significance of the mole. State the three mole facts for any substance (molar volume, molar mass, Avogadro’s number): 1 mole = 22.4 Liters @ STP (gases only) 1 mole = 6.02 x 1023 particles (particles = molecules, atoms, or ions) 1 mole = gram molecular mass of chemical Use dimensional analysis to convert between moles, mass, volume, and number of particles for a chemical. Molar Mass Calculations: Calculate the molecular mass or molar mass of any compound. State that the mass of a molecule is measured in amu’s and the mass of a mole is measured in grams. Percent Composition: Calculate the mass percent (percent composition by mass) of a substance based on its molecular formula. Give examples of empirical formulas and molecular formulas. Identify a formula as empirical or molecular. Calculate the empirical formula from mass percentages or mass data. Determine the molecular formula of a compound given its empirical formula and molar mass. Chemical Equations: Know how to represent a chemical change in a chemical equation with reactants and products. Be able to balance a chemical equation. Know how to indicate the physical states by symbols: (s), (l), (g), (aq). Stoichiometry: Know how to perform stoichiometric calculations based on the balanced chemical equation and given information. Know the concept of limiting reactants. Know how to determine the limiting reactant using reactant quantities or products formed. Know how to calculate how much excess reactant remains. Know how to calculate theoretical yield and percent yield.