* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Quantum effects in astrophysics
Corvus (constellation) wikipedia , lookup
Timeline of astronomy wikipedia , lookup
Observational astronomy wikipedia , lookup
Stellar classification wikipedia , lookup
H II region wikipedia , lookup
Stellar evolution wikipedia , lookup
Future of an expanding universe wikipedia , lookup
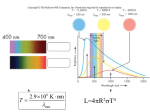
Quantum effects in astrophysics Donna Kubik May 7, 2003 Abstract Almost everything known about stars and galaxies is learned through spectroscopic observations. Their chemistry, physical properties as pressure, density and temperature, ionization state, magnetic properties, and motion can be determined from such observations. The interpretation of spectroscopic observations depends on an understanding of transitions that occur between atomic levels. How these transitions take place and what can be learned from them is considered. Quantum mechanical ideas, such as quantum statistical measurements, have been used to develop tools for the study of astrophysics. Two such tools, the hydrogen maser and the Hanbury Brown-Twiss interferometer are discussed As astronomers probe deeper and deeper into space they must detect fainter and fainter signals. Noise is the major limitation on detector performance. Two types of noise based on quantum effects, quantum 1/f noise and photon noise, are discussed. 2 Contents 1 Observing dynamic stellar parameters 2 Brief history of spectroscopy 3. Discovery of helium 4 Absorption and emission spectral and Kirchhoff’s laws 5 The information contained in spectral lines 5.1 Doppler broadening 5.2 Collisional broadening 5.3 Other types of broadening 5.4 Recombination lines 6 Hydrogen lines 6.1 Neutral hydrogen 6.1.1 H masers 6.2 Ionized hydrogen 6.3 Molecular hydrogen 7 H-R diagrams 7.1 Regions of the H-R diagram 7.2 Luminosity class 7.3 Stellar temperature 7.3.1 Stellar temperature via color 7.3.2 Stellar temperature via spectra 8 Spectroscopy in exotic environments 8.1 Forbidden lines 9 Quantum 1/f noise and photon noise 9.1 Quantum 1/f noise 9.2 Photon noise 10 Hanbury Brown and Twiss interferometer A1 Derivation of number of degenerate quantum states 11 Conclusion 3 1 Observing dynamic stellar parameters Every season we refer to the same stars. We refer to winter, spring, summer, and fall stars. Every winter we see Sirius, Betelgeuse, and Procyon, the stars of the Winter Triangle, high in the sky followed by spring stars that comprise constellations such as Virgo and Leo. The next season brings Vega, Deneb and Altair, forming the Summer Triangle, followed by the stars that form Pegasus, Andromeda, and Perseus, which are a few of the fall constellations. Then the stars of winter return and the sequence begins once again. The night sky appears to be unchanging year after year. More careful examination, however, reveals that, underneath this apparent unchanging permanence, the Universe is much more interesting. The only reason we can even see the stars is because they emit vast amounts of radiation created during the conversion of one element into another in their interiors. Sometimes the changes are even more dramatic, as seen in the sudden appearance of a supernova in a distant galaxy. The changes are not limited to chemistry. During the course of its life, stars also change in size, luminosity, temperature, color, pressure, and density. These changes can be observed via spectroscopic observations. 2 Brief history of spectroscopy Although the spectral nature of light can be seen in a rainbow, it was not until 1666 that Newton showed that the white light from the sun could be dispersed into a continuous series of colors. Newton introduced the word "spectrum" to describe this phenomenon. (The word spectrum is from the Latin for appearance, from specere, to look at) Some thought that the colors were somehow added to the light by the glass of the prism. Newton disproved this idea by refocusing the dispersed light through a second prism. Only white light emerged from the second prism, demonstrating that the second prism had reassembled the colors to give back the original beam, Newton's analysis of light was the beginning of the science of spectroscopy. In 2000, the European Space Agency decided to honor Newton by giving the name of Isaac Newton to the agency's XMM (Xray Multi-Mirror) spectroscopy mission. The xray space telescope, launched on 10 December 1999, was renamed the XMM-Newton observatory. XMM-Newton is expected to continue making observations until 2010. The wavelength band chosen for the Reflection Grating Spectrometer (RGS) component of the XMM-Newton mission (5 - 35 Ångstrom) contains the K-shell transitions of oxygen, neon, magnesium, aluminum, silicon, as well as the L-shell transitions of iron. Detailed study of these spectral features allows the physical characteristics (density, temperature, ionization state, element abundances, mass motions 4 and redshift) of the emitting region and its surrounding environment. XMM-Newton makes it possible to study these spectral features in many types of astrophysical objects like corona of stars, binary star systems, supernova remnants, clusters of galaxies and far away active galactic nuclei. In 1814 Joseph von Fraunhofer repeated Newton’s experiment, magnifying the resulting spectrum. He discovered that the solar spectrum contains hundreds of fine dark lines which became known as spectral lines. Fraunhofer detected more than 600 lines and today physicists have detected more than 30,000 lines. It gradually became clear that the sun's radiation has components outside the visible portion of the spectrum. William Herschel (1800) demonstrated that the sun's radiation extended into the infrared, and J.W.Ritter (1801) made similar observations in the ultraviolet. Over the years radiation from the low frequency radio through high frequency gamma radiation was discovered. By the mid-1800’s, chemists discovered that they could produce spectral lines in colorless flame. Certain chemicals are easy to identify by the distinctive colors they emit when bits of the chemical are sprinkled into this flame. Bunsen’s colleague, Gustav Kirchoff, suggested that light from the flames could be better studied by passing it through a prism just as Fraunhofer had done with sunlight. They discovered that the spectrum from the flame consisted of a pattern of thin bright lines against a background. They found that each element produces its own characteristic pattern of spectral lines. This was the birth of spectral analysis, the identification of elements by their spectral lines. (Comins 2001) 3 Discovery of helium After Bunsen and Kirchoff recorded the prominent spectral lines of all the known elements, they began to discover other spectral lines in mineral samples. They found a new line in the blue portion of the spectrum of mineral water naming the unknown element responsible for the line cesium (from the Latin caesius, meaning gray-blue). They also found a new line in the red portion of the spectrum of a mineral, naming its source rubidium (from rubidus, for red). During a solar eclipse in 1868, astronomers found new spectral line in the light coming from the upper atmosphere of the Sun. This line was attributed to a new element, which was named helium (from the Greek, meaning Sun). 4 Absorption and emission spectra and Kirchhoff’s laws Kirchhoff sometimes observed dark spectral lines, called absorption lines and other times saw bright spectral lines, called emission lines against an otherwise dark background. By the early 1860’s, Kirchhoff had discovered the conditions under which theses different types of spectra are observed. 5 Law 1 A hot object or a hot, dense gas produces a continuous spectrum. A continuous spectrum is a complete rainbow of colors without any spectral lines. This is a blackbody spectrum Law 2 A hot, rarefied gas produces a series of bright spectral lines against a dark background called an emission line spectrum. Law 3 A cool gas in front of a continuous source of light produces a series of dark spectral lines among the colors of the rainbow referred to as an absorption line spectrum. FIG. 1 shows how absorption and emission lines are formed. The bright lines in the emission spectrum of a particular gas occur at the exactly the same wavelengths as the dark lines in the absorption spectrum. The relative temperatures of the gas cloud and its background determine which spectrum is observed: Absorption lines are seen if the background is hotter than the gas, and emission lines are seen if the background is cooler. Either can be used to determine the gas’ chemical composition. This link displays the emission and absorption spectra of all the elements: http://javalab.uoregon.edu/dcaley/elements/Elements.html 6 CONTINUOUS SPECTRUM GAS ABSORPTION SPECTRUM HOT SOURCE EMISSION SPECTRUM FIG. 1 Continuous, absorption, and emission spectra Bohr’s ideas help explain Kirchhoff’s laws. Each spectral line corresponds to one specific transition between energy levels of the electrons of a particular element. An absorption line is created when an electron jumps from a lower energy level to a higher level, extracting the required photon from an outside source of energy, such as the continuous spectrum of a hot, glowing object. An emission line is produced when and electron transitions to a lower orbit and emits a photon. Line spectra are also called atomic spectra, because the lines represent wavelengths radiated from atoms when electrons change from one energy level to another. Band spectra is the name given to groups of lines so closely spaced that each group appears to be a band. Molecular spectra, are produced by molecules radiating their rotational or vibrational energies, or both simultaneously. 7 5 The information contained in spectral lines Left to itself, an excited atomic system will spontaneously jump to a lower energy state. The mean time required for the transition depends on several factors including the symmetries of the states, the dimensions of the system. If the system has a total transition probability P, its total life in the excited state is t 1/ P . The energy of the state therefore cannot be determined with arbitrarily great accuracy. The limited time in the excited state implies that the energy can only be determined to an accuracy governed by the uncertainty principle, . t Improbable transitions therefore have a narrow natural line width. The accuracy to which the transition energy between two states i and k can be determined depends on the lifetime of both the upper and lower states. The total frequency width of the line , is the sum of the widths of the two levels, This total width is usually denoted by 2 1 2 . is called the natural line width for the transition. In astronomical sources, the natural line width is seldom observed, but deviations from this width can provide a great deal of information. The lines may be broadened by Doppler broadening, collisional broadening, and other effects due to interactions with neighboring atoms. 5.1 Doppler broadening Doppler broadening is due to random motions of emitting of absorbing atoms or molecules. For small velocities the frequency shift of the radiation is roughly proportional to the line of sight velocity component vr, 0 vr , vr c . c Two kinds of motion can contribute to Doppler broadening: the thermal velocities of emitting atoms within a cloud and the turbulent velocities peculiar to the clouds superposed along a line of sight. Sometimes these two effects can be resolved. Generally the width at half maximum for Doppler broadening, 2kT (ln 2) 0 2 mc 1/ 2 8 is much greater than the natural line width . However it drops off exponentially and therefore much faster than the natural line width, The observed wings of very strong lines, for example Lyman-generally are due to natural line width. 5.2 Collisional broadening In the relatively dense atmospheres of stars, atoms or ions often suffer a collision while they are in an excited state. Because any given collision may induce a transition to a lower state, the collective effect of such collisions is to increase the total transition probability. Thus, if the spontaneous transition rate were and the number of collisionsinduced transitions in unit time is , . The emitted line has the spectral density intensity distribution of the natural line shape except that is replaced by 5.3 Other types of broadening There are other effects due to interactions with neighboring atoms that can cause shifting and splitting of state through the influence of electric fields (Stark effect) and through resonance coupling between atomic states. These processes lead to line broadening but at low densities their effects are small. 5.4 Recombination lines A recombination line is an emission line arising from the recombination of a free electron with an ion. As the electron drops down through the energy levels of the atom, it emits recombination lines at wavelengths that depend on the difference in energy between the levels. For hydrogen, these lines range from radio (caused by transitions between the outer, lower-energy transitions) to optical to the second energy level. Recombination lines to the ground state produce an ultraviolet photon which itself causes further ionization. The gas temperature and density of ionized nebulae can be estimated from recombination lines. 6 Hydrogen lines Hydrogen is the most abundant element in the Universe. It occurs in many forms, including neutral hydrogen (HI), ionized hydrogen (HII), and molecular hydrogen (H2). The spectra from each of these forms can be used to learn about the motion and extent of galaxies, interstellar and intergalactic magnetic fields, and the location of star forming regions. 9 6.1 Neutral hydrogen (HI) The energy of a spectral line is the energy difference of the atomic levels between which the transition occurs. The energy, , of an atomic level n, is n Z 2 e 4 , 2n 2 2 where Z is the atomic number, is the reduced mass, e is the charge of an electron, and is Planck’s constant. Since the energy depends on the reduced mass, it has a somewhat different value for normal hydrogen than it does for deuterium. The extra neutron makes the nucleus about twice as massive as in normal hydrogen so that the reduced mass of deuterium is D me mD 2me mP m me 1 e me mD me 2mP 2m P while P me 1 me mP . Subscripts e, D, and P, represent electrons, deuterons, and protons. The differences in reduce mass means that the energy levels of deuterium will lie further apart than the levels of ordinary hydrogen by about one part in 2mP / me 3700 . In the visible part of the spectrum, this corresponds to a line shift on the order of 1.5 angstroms. This shift could be easily detected, yet, for many years, no deuterium was ever detected in any astronomical object. In contrast, the terrestrial abundance of deuterium is readily measured and is roughly 2x10-4 by fraction of atoms. If such abundances existed elsewhere in the Universe, deuterium should have been readily detected. However, in the interstellar medium, deuterium appears to be fractionated; it binds more readily to molecules than ordinary hydrogen and therefore appears underabundant. Most of the existing deuterium may be primordial and any deuterium that has even been cycled into a star has been destroyed. If all interstellar matter in the Galaxy had undergone such cycling at least once, planets formed early in the Galaxy’s life and clouds containing gases that have never undergone nuclear processing in a star might be the only places where deuterium can now be found. There 2n2 quantum states are associated with the nth energy level (Appendix A). The lowest (ground) state of hydrogen consists of two distinct components, corresponding to two different orientations of the electron’s spin relative to the nuclear spin. These two configurations have slightly different energies, so a transition from the higher to the lower state can occur spontaneously. This transition occurs at a frequency 10 of 1420 MHz corresponding to a wavelength of 21 cm and an energy of ~6x10-6 eV (FIG. 2). 1420 MHz FIG. 2 Hydrogen spin flip transitions An individual hydrogen atom excited into the higher energy level typically waits 12 million years to make the energy jump spontaneously. However, the radiation is observed because of the vast numbers of hydrogen atoms in galaxies and because collisions can trigger the transitions. The distribution of hydrogen in the Galaxy was first mapped by 1420 MHz observations. This was possible because radio waves are not absorbed by intervening gas and dust that bocks visible light. It is interesting that we now have very good maps of the distribution of gas in the Galaxy but no comparable map showing the distribution of stars. This is because stars do not emit enough radiation in the radio or far infrared (which can also penetrate gas and dust). 6.1.1 H masers Hydrogen masers are now used as frequency standards at many astronomical observatories. They provide accurate frequencies for signal processing (as references for local oscillators) at radio and millimeter-wave observatories and provide the very accurate timing required for interferometric observations (to get accurate phase information). However, the first atomic clock was an NH3 gas maser. It was based on the first maser, which was an ammonia- beam maser (Gordon, Zeiger, and Townes 1955). The two energy levels used in the ammonia maser are vibrational states of the ammonia molecule (FIG. 3). The hydrogen atoms can be considered to rotate. The nitrogen atom oscillates between two positions, above and below the plane of the hydrogen atoms. These two arrangements do not represent exactly the same energy. The wave functions of the hydrogen and nitrogen atoms are not quite symmetrical. Therefore the molecule exists in two energy states. The difference in energy between the states corresponds to a frequency difference of 23.87 GHz, or ~24 GHz. 11 24 GHz N H H H H H H N FIG. 3 NH3 vibrational states It is no coincidence that “laser action” was first produced in the microwave region. There is no need for pumping. This can be seen from the distribution function for transitions that occur in the microwave region. The Boltzman distribution may be used because the first maser operated with a gaseous system, neither molecular state influences the other state, and the system is in thermal equilibrium. The population ratio of the higher energy state to the lower energy state is about 1:1 in the energy range that corresponds to microwaves: E for microwave (maser) transitions is ~10-5 eV, while for optical (laser) transitions, E is only a few eV. So thermal energy (kTroom = 0.0258eV) is enough to generate a large population of atoms in the higher energy state. The population ratio vs. energy is shown in FIG. 4. population ratio 1.00 0.90 0.80 n2/n1 0.70 0.60 nE2 0.50 nE1 e E / kT 0.40 0.30 0.20 0.10 0.00 0.00E+00 masers 2.00E-01 4.00E-01 6.00E-01 8.00E-01 1.00E+00 delta E 1.20E+00 1.40E+00 (eV) FIG. 4 Population ratio vs. 1.60E+00 1.80E+00 2.00E+00 lasers 12 Another reason it is no coincidence that “laser action” was first produced in the microwave region is that there is no need use 3 or 4 level schemes (as used in lasers) to populate a metastable state. The ratio of the spontaneous emission coefficient to the stimulated emission coefficient varies with frequency as 3, A21 8h 3 . B21 c3 This ratio is much smaller in the microwave part of the spectrum than in the optical. Spontaneous emission can therefore be neglected compared to other important processes such as stimulated emission and absorption. There is no need to take advantage of an intermediate metastable state. The states are inherently long-lived. So all you have to do is physically separate the high energy state from the low energy state to achieve a metastable population inversion. The operation of the ammonia-beam maser shown in FIG. 5 is described below. 6 1 2 3 5 4 FIG. 5 Simplified diagram of the essential parts of the NH3 maser 1. A heater gives energy to molecules of ammonia (NH3) in the source. At this point about half of the molecules are in an excited state, the other half are not. 2. The ammonia molecules stream into the focuser (also called a separator), which is evacuated. 3. The focuser removes molecules in the lower quantum state from those in the upper quantum state (for these would absorb rather than emit photons at the desired frequency) while focusing those in the upper state. The energy states can be separated by a system of focusing electrodes. The electric dipole moments induced in the NH3 molecules 13 interact with the non-uniform electric field produced by the electrodes. The internal energy of an upper state molecule is increased and that of a lower state molecule is decreased so that, in the non-uniform electric field, the lower state molecules move towards the higher field region and the upper state molecules move to the lower field region. 4. The ammonia molecules that pass into the resonant cavity (tuned to 24 GHz) are almost all excited constitute an inverted population. The cavity has a very high Q, so there is sufficient noise power to initiate transitions from the upper state the lower state. Photons from these transitions can then stimulate emission from other molecules. 5. When it is used as an amplifier, the signal to be amplified is injected into the cavity via an input waveguide. This radiation leads to even more rapid stimulated emission by the excited molecules. 6. The resultant coherent radiation detected at the output waveguide is an amplified version of the input signal. Masers were used in radio astronomy as very low noise amplifiers. Since molecules are uncharged, the usual shot noise in electronic amplifiers is missing, and essentially no noise in addition to thermal noise is present in maser amplifiers. (Gordon, Zeiger, and Townes 1955). The radiation reflects back and forth inside the cavity, whose size is specially chosen and regulated to reinforce waves of just this frequency. If the NH3 can supply sufficient power to make up for losses in the cavity, the maser may function as a selfoscillator. Such masers are extremely selective as amplifiers. They will not amplify signals that are as little a 5000 Hz away from 24 GHz. They do not shift by more than one part on a billion or more over long periods, so the early masers were used as atomic clocks. An NH3-beam maser served as the first atomic clock standard by NIST (National Institute of Standards and Technology). H masers have superceded NH3 masers as frequency standards. They are also based on a two-state system, in this case the 1420 MHz transition described above. The operation of the H maser is very similar to that of the NH3 gas maser. The selector is based on an inhomogeneous magnetic that focuses upper energy atoms into a Tefloncoated quartz storage bulb located in a TE01-mode cavity. The cavity is tuned to the 1420 MHz hyperfine transition frequency of hydrogen. The hydrogen atom spends most of its time in free space in the storage bulb, and, unlike NH3, the effects of wall collisions are small because its electric polarizability is low. The effect of first order Doppler shift is nearly eliminated because the average velocity of the atom in the storage bulb is close to zero. The long interaction time of the confined hydrogen atom produces stabilities near one part in 1014 for averaging times from 1 to 105 seconds. (Weber 1987) But was the NH3 maser really the first maser? Naturally occurring molecular masers and lasers have been oscillating for eons in interstellar space, on comets, and in 14 planetary atmospheres. In 1962, molecular lines detected in radio emission from interstellar clouds had huge intensities (equivalent to blackbody temperature of 10121015 K) but at the same time had very narrow doppler linewidths (corresponding to kinetic temperatures below 100 K). An explanation is that these emissions represent naturally occurring masers. Many types of astrophysical masers have been detected as OH, SiO, and H2O. In colliding galaxies and near black holes, astronomical masers can be a million times stronger than regular masers. These masers, named megamasers, were discovered in 1982. Masers whose detailed modeling has been most successful are the OH masers in late-type stars that are pumped by infrared radiation resulting from the reemission of the stellar radiation by the dust particles that permeate the stellar wind. Detailed models of the H2O masers in these sources show that pumping is controlled by collisions. (Harwitt 1998) 6.2 Ionized hydrogen (HII) The formation of a cluster of stars can be detected by a red glow in a nebula. Because these nebulae are predominantly ionized hydrogen, they are called HII regions. The most massive pre-main-sequence stars (developing stars before the onset of fusion) are of type O and B. Pre-main-sequence O and B stars are exceptionally hot, emitting ultraviolet radiation, so they can ionize distant hydrogen atoms. While some hydrogen atoms are being knocked apart by UV photons, some of the free protons and electrons recombine. As these reassemble, energy is released. The transition of the electron from the n=3 to n=2 level produces H-alpha photons at 656 nm in the red portion of the visible spectrum (FIGS. 6 & 7). FIG. 6 Hydrogen transitions 15 FIG. 7 H-alpha is seen at optical wavelengths High energy light from the bright young stars NGC 2244 ionizes the surrounding hydrogen gas clouds to create the red emission nebula (FIG. 8). FIG. 8 NGC 2244 and emission nebula 6.3 Molecular hydrogen (H2) Molecular clouds are dense concentrations of primarily H2 gas that span tens to hundreds of light years and weigh as much as a million suns. These objects are of great interest to astronomers, because they contain roughly half of all the interstellar gas and dust and give birth to nearly all of the stars in spiral galaxies such as ours. Star-forming molecular hydrogen is difficult to detect in space. The hydrogen molecule is symmetric, with two atoms of equal mass joined together. Such molecules do not emit many photons at radio frequencies. Asymmetric molecules, like CO, consist of two molecules of unequal mass. CO is easily detected at radio frequencies. The ratio of CO to H2 is reasonably constant: 1:10,000. So CO is an excellent tracer for H2. 7 H-R diagrams . Although stellar evolution occurs on scales too slow for us to see, each star is at a different point in its evolution. By measuring the temperature and brightness of many stars and arranging the data, ideas of stellar evolution have been formulated. 16 Around 1905, Ejnar Hertzsprung pointed out that a regular pattern appears when the luminosites (or absolute magnitudes) of stars are plotted against their surface temperature (or spectral type). Within 2 years, Henry Norris Russell independently discovered the same result. Such plots are now referred to as Hertzsprung-Russell diagrams, or H-R diagrams. H-R diagrams show that stars do not have random surface temperatures and luminosities (FIG. 9). Temperature and luminosity are correlated. H-R diagrams are valuable tools to organize stars by their physical properties (temperature and absolute magnitude), but the diagrams do not explain the physical mechanisms that produce these characteristics. The locations of stars on the H-R diagram provide clues that astronomers use in developing and testing theories of stellar evolution. Theories of stellar evolution attempt to explain the distribution of stars in the H-R diagram, not only why some areas are populated and others are not, but why some are heavily populated and others are not. FIG. 9 H-R diagram 7.1 Regions of the H-R diagram Main sequence stars are those that are fusing hydrogen into helium in their cores at a constant rate. The location of a main sequence star depends primarily on its mass. The least massive stars are the least luminous. The most massive stars are the most luminous. Massive O and B stars may consume all of their hydrogen in only a few million years, because gravitational forces are greater on a more massive star’s core. The resultant high pressures speed up fusion. Also, the CNO cycle which dominates over the p-p chain in high-mass stars, is a faster method hydrogen fusion. Low mass stars may take hundreds of billions of years to consume all of their hydrogen. But, no matter if it’s high or low mass, a main-sequence star takes a long time to consume all its hydrogen compared to the time it spends in the other stages of stellar evolution. That is why the majority of stars represented on an H-R diagram are on the main sequence. 17 To the right of the main sequence is a group of stars that are bright but cool. From the Stefan-Boltzman law, a cool object radiates much less heat per unit area then a hot object does. In order to be so bright, these cool stars must therefore be huge compared to main sequence stars of the same temperature. Since they are huge, they are called giants, and by contrast, main sequence stars are often called dwarfs The cooler members of the giants are called red giants, because they appear reddish (Aldebaran, Arcturus). Some stars are bigger and brighter than typical giants. These are called supergiants (Betelgeuse, Antares) The stars in the lower left are hot, yet dim, therefore must be tiny. These are called white dwarfs. 7.2 Luminosity class Because stars can have the same temperature but different luminosities, astronomers usually describe a star by both its spectral type and its luminosity class. By studying absorption lines in detail (as the width of the lines), astronomers can categorize stars into luminosity classes (FIG. 10). This is because absorption lines are also affected by density and pressure of a star’s atmosphere, which depends on the size of a star. Luminosity class corresponds to different stages of stellar evolution. Ia : Brightest supergiants Ib : Less luminous supergiants II : Bright giants III : Giants IV : Subgiants V : Main-sequence stars FIG. 10 Luminosity class 7.3 Stellar temperature The surface temperature of a star may be measured either from the color of the star or by analysis of the stellar spectrum. 18 7.3.1 Stellar temperature via color A star’s blackbody curve reveals its temperature, as seen from Planck’s function, 2h 3 1 . B (T ) 2 h / kT c e 1 Starlight is passed through different color filters. The intensity for each wavelength is measured. The intensity of light from a cool star peaks at long wavelengths, making the star look red. A hot star’s intensity curve looks blue. A star of intermediate temperature, such as the Sun, occurs near the middle of the visible spectrum, giving the star a yellowish color (FIG. 11). FIG. 11 Blackbody curves reveal stellar temperature Photometry is used to measure the color accurately. A light-sensitive device (such as a CCD) measures the intensity of light that passes through a set of standardized filters, the most common being UBV filters. Each of the three UBV filters is transparent in one of three broad wavelength bands: the ultraviolet (U), the blue (B), and the central yellow (V, for visible) region of the visible spectrum (FIG. 12). FIG. 12 UBV bandwidths 19 To do photometry, the intensity of the starlight that passes through each of the filters is measured. This gives three apparent magnitudes for the star. The intensity of the starlight in neighboring bands is compared by subtracting one magnitude from another to form the combination (B-V) and (U-B), which are called the color indices. The color index indicates how much brighter or dimmer a star is in one wavelength than another. The color index is directly related to the star’s surface temperature. If a star is hot, its radiation is skewed toward the short-wavelength ultraviolet, which makes the star bright through the U filter, dimmer through the B filter, and dimmest through the V filter (FIG. 13). FIG. 13 Color indices Interstellar gas and dust cause stars to appear redder than they really are, so this reddening must be taken into account whenever a star’s temperature is determined from its color indices. A star’s spectrum provides a more direct measure of a star’s surface temperature. 7.3.2 Stellar temperature via spectra Stars are classified according to the appearance of their spectra in a way that reveals their surface temperatures. Each element has a specific signature at a specific temperature. For example, produce certain spectral lines, a star must be hot enough to excite the electrons out of the n=2 state but not hot enough to ionize a significant fraction of the atoms. In ionized atoms, the e- are removed, so do not have e- to excite, so no lines. Astronomers have classified stars according to their spectra into spectral classes (also called spectral type) (FIG. 14). The major spectral classes are type O, B, A, F, G, K, M. Each type is subdivided into 10 finer divisions (0-9), as A8 or F0. 20 Class O B A F G K M 8 Color Surface Temperature Blue-white 35,000°C Blue-white 21,000°C White 10,000°C Creamy 7,000°C Yellow 6,000°C Orange 4,500°C Red 3,000°C FIG. 14 Spectral classes Strong lines Ionized helium Helium Hydrogen Ionized calcium Calcium Titanium oxide Titanium oxide Spectroscopy in exotic environments Astronomical objects provide exotic environments to study physics. Stars, nebulae, planets, etc, may serve as laboratories in space providing conditions beyond those possible in experiments built on earth. For example, the “exotic” temperatures, pressures, and magnetic fields in pulsars allow astrophysics to study aspects of plasma physics, fluid dynamics of super fluids, general relativity, solid state physics, and magnetohydrodynamics not possible on earth. Other examples include the observation of forbidden lines and the discovery of helium via solar spectroscopy. 8.1 Forbidden lines Under normal conditions, not all transitions are equally likely. Quantum mechanics provides certain selection rules which predict the more commonly observed transitions between different energy levels. These allowed transitions generally represent energy that one photon can carry off easily (in the emission case) and still conserve the total angular momentum of the system. Other transitions are mathematically possible but are considered “forbidden” because chances are good that, under terrestrial conditions, where gases are much denser, that before the atom can radiate by such a forbidden transition, a collision with another atom or molecule will deexcite the atom collisionally (Shu 1982). However, in the low densities of interstellar space and the regions around hot stars, collisions are extremely rare and there is time for the spontaneous decay to occur. Forbidden lines disappear above a critical density (about 108 atoms/cm3), so their existence is an indicator of density in the interstellar gas. Forbidden lines are denoted by square brackets, such as the [O III] lines of doubly ionized oxygen. A semi-forbidden line, designated with a single square bracket, such as C III], occurs where the transition probability is about a thousand times higher than for a forbidden line (Ridpath 1997). 21 9 Quantum 1/f noise and photon noise As astronomers want to probe deeper and deeper into space they must detect fainter and fainter signals. Noise is the major limitation on detector performance. Noise sources may be separated into four main types: intrinsic noise, i.e. noise originating in the detector, signal noise, i.e. noise arising from the character of the incoming signal, particularly its quantum nature, external noise such as spurious signals from cosmic rays, etc, and processing noise, arising from amplifiers, etc, used to convert the signal from the detector into a useful form (Kitchin 1991). Some of these noise mechanisms will be discussed below, elaborating upon those with quantum mechanical considerations. 9.1 Quantum 1/f noise Many detectors used for astrophysical observations are solid state devices. In solid state devices, intrinsic noise comes from four sources: thermal noise, shot noise, g-r noise (generation–recombination) and flicker, or 1/f or low-frequency, noise. Thermal noise, also known as Johnson or Nyquist noise, arises in any resistive material and is due to the thermal motion of the charge carriers. These motions give rise to a current whose mean value is zero, but which may have non-zero instantaneous values, observed as noise. Shot noise occurs in junction devices and is due to variation in the diffusion rates in the neutral zone of the junction because of random thermal motions g-r noise is caused by fluctuations in the rate of generation and recombination of thermal charge carriers which leads to fluctuations in the device’s resistivity. 1/f noise occurs when the signal is modulated in time either because of its intrinsic variations or because it is being chopped (i.e. the source and back ground are alternately observed). The mechanism of 1/f noise is unclear and is an active research topic. Its amplitude follows an f n power spectrum where f is the chopping frequency and n lies typically in the range 0.75-2.0. This noise can be minimized by increasing f, however, faster operation may be limited by the response time of the detector. Two features of 1/f noise are especially interesting. First, its near ubiquity suggests that at least some feature of the explanation should not depend on the detailed models of particular materials. The form of the noise spectrum is about the same in metals, semiconductors, semimetals, and Josephson junctions (Weissman 1988). Second, the spectral form is nearly scale invariant. 22 Based on measurements on a number of materials, Hooge (Hooge 1981) postulated the following empirical relationship: Si ( f ) I 2 H fN where S i is the spectral intensity of the 1/f noise current I is the average current N is the average number of charge carriers, and H is the Hooge parameter. One theory for the production of 1/f noise is that it is the superposition of g-r noise and quantum 1/f noise (FIG. 15) (Van der Ziel 1986). FIG. 15 Superposition of a 1/f noise spectrum, two g-r spectra, and thermal noise Open circles represent experimental data (Van der Ziel 1986) g-r noise may be due to electrons being randomly absorbed and reemitted by traps in the semiconductor, either at the surface or in the bulk. If a number of traps exist with an appropriate distribution of time constants, a frequency dependence with is close to 1/f may result (FIG. 15) It has been shown that mobility fluctuations may result in 1/f noise. The carrier mobility will show random variations due to the scattering process which the carriers undergo. This phenomenon is central to the quantum 1/f noise theory. Quantum 1/f noise is generated whenever charged particles are accelerated or decelerated. Some of the particles emit very low frequency radiation, bremsstrahlung 23 radiation, of frequency F 10 4 10 4 Hz (Van der Ziel 1986). Since the energy per photon is hf, and the energy per unit Hz is a constant for bremsstrahlung radiation, the number of photons per unit Hz will be proportional to 1/f. This bremsstrahlung part of the beam modulates the rest of the electron beam producing a 1/f noise spectrum. The Hooge parameter can be derived in terms of the average change in velocity, v, for the scattering event the electrons go through, H 4 v 2 , 3 c 2 where is the fine-structure constant ( 1/ 137 ). For an elastic scattering process, one can show that v 2 2v 2 . Since v 2 3kT / m* , H 4 6kT , 3 m * c 2 where m * is the effective mass, k is Boltzman’s constant, T is the absolute temperature, c is the speed of light. For a given material, the strength of the 1/f noise is inversely proportional to the effective mass. This result agrees with the observation that Si devices have much lower 1/f noise than GaAs devices, for the effective mass of GaAs is smaller than the effective mass of Si (Van der Ziel 1986). Since traps may be controlled by material processing, quantum 1/f noise is often regarded as the lower limit for 1/f noise. 9.2 Photon noise Noise also arises from the quantum nature of light. This is sometimes called “photon noise”. At low signal levels, photons arrive at the detector sporadically. The probability of arrival is given by a Poisson distribution, and this has a standard deviation of n , where n is the mean number of photons per unit time. Thus the signal will fluctuate about its mean value. In order to reduce the fluctuations to less than x%, the signal much be integrated for 10 4 /( nx 2 ) times the unit time. At high photon densities, photons tend to cluster more than a Poisson distribution would suggest, because they are 24 subject to Bose-Einstein statistics. This latter noise source may dominate at radio wavelengths, but is not normally of importance over the optical region (Kitchin 1991). 10 Hanbury Brown and Twiss interferometer Bose-Einstein statistics are used to astronomers’ advantage to measure stellar diameters using the Hanbury Brown –Twiss interferometer. A property of a stream of particles obeying Bose-Einstein statistics (which includes photons) is that the particles tend to bunch in clusters. In the 1950’s, Hanbury Brown and Twiss determined the size of a radio star by measuring this coherence of the observed radiation (Henny, et. al. 1999). Quantum mechanically two particles are considered identical if their momenta and positions are identical within values p xx h, p y y h, p z z h, and if their spins are identical. In this description each particle is characterized by a position (x, y, z, px, py, pz) in a six-dimensional phase space. It occupies a sixdimensional phase cell whose volume is xyzp xp yp z h 3 . Particles within one phase cell are identical, physically indistinguishable, while those outside can be distinguished. The fact that two photons sometimes occupy the same phase cell is used to measure the angular diameter of stars (Harwitt 1998). Two photon counters are placed a distance D apart, transverse to the direction of the star (FIG. 16). 25 d R D FIG. 16 The Hanbury Brown-Twiss interferometer If D is small enough, there is the possibility that one photon from a cell will hit one detector while the other photon hits the other detector, the simultaneous arrival detected by a coincidence counter. The angle a star subtends is d / R , where d is the diameter of the star at distance R. The photon pair entering either detector has a distribution in momentum, along the direction of D, pD p (h / c) where is the frequency to which the detector is sensitive. The nonzero value of pD makes it necessary that D itself be small so that photons reaching either detector may be from the same phase cell. It is necessary that Dp d h, Dh h, c D , c / is the wavelength of the radiation. By increasing D, a decreasing coincidence rate is observed, and for values of D at which coincidences no longer occur the angular diameter is / D . The stellar angular diameter is d / R / D. 26 11 Conclusion The interpretation of many astrophysical observations depends on an understanding of quantum mechanical processes, as atomic and molecular energy transitions. Quantum mechanical ideas have also been used to develop tools for astrophysics, such as the hydrogen maser and the Hanbury Brown-Twiss interferometer. 27 Appendix A Derivation of number of degenerate quantum states The Schrödinger equation in three dimensions has the following form: 2 2 2 2 2m x 2 y 2 z 2 U ( x, y, z ) E , (A-1) where is a function of x, y, and z. This equation is generally solved by using spherical coordinates ( r , , ) instead of (x, y, z) (Eisberg and Resnik 1985). After the change of variables, those solutions that are separable and can be factored as (r , , ) R(r )( )( ) (A-2) are considered, where the radial function, R (r ) , the polar function, ( ) , and the azimuthal function, ( ) , are each functions of a single variable. This procedure gives three differential equations, each of a single variable ( r , , or ). When solving such a three-dimensional equation, three parameters emerge in a natural way as indices for the solutions. These indices (n, l, ml), are the three quantum numbers that label the solutions. The solutions can be written as: n,l ,m (r , , ) Rn,l (r ) l ,m ( ) m ( ) . l l l (A-3) The three quantum numbers and their allowed values are shown in TABLE A1: Quantum number n l ml principal quantum number angular quantum number magnetic quantum number Allowed value 1, 2, 3, … 0, 1, 2, … , n-1 0, 1, 2,..., l TABLE A1 Allowed values of the three quantum numbers 28 n l ml Number of degenerate eigenfunctions for each l Number of degenerate eigenfunctions for each n 1 0 0 2 0 0 1 -1, 0, +1 0 0 1 -1, 0, +1 2 -2, -1, 0, +1, +2 1 1 3 1 3 5 1 3 4 9 TABLE 2 Possible values of l and ml for n = 1, 2, 3 Inspection of the allowed values shown in TABLE 2 indicate that: 1. For each value of n, there are n possible values of l 2. For each value of l, there are (2l+1) possible values of ml. 3. For each value of n, there are a total of n2 degenerate eigenfunctions Another way to see that the number of possible quantum states with principle quantum number n is proportional to n2 is to consider that the nth radial state of an atom has phase space volume proportional to 4p n2 p n and 4rn2 rn . Then the 4p n2 p n 4rn2 rn phase space volume Number of states volume of unit cell xyzp xp y p z (A-4) Quantum mechanically it can be shown that two particles are considered to be identical if their momenta and positions are identical within values p xx h, p yy h, p zz h (A-5) and if their spins are identical. In this description each particle is characterized by a position in a six-dimensional phase space. It occupies a six-dimensional phase cell whose volume is xyzp xp yp z h 3 . (A-6) Particles within one phase cell are identical, while those outside can be distinguished from one another. 29 To distinguish states having different radial positions and momenta, the product pr for these state would have to differ by , otherwise they would not be distinguishable in Heisenberg’s sense: p n rn n (A-7) Also the smallest possible size of an electrostatically bound atom must be related to the uncertainty in momentum through p 2 r 2 p 2 r 2 2 . (A-8) Using A-6, A-7, and A8, the number of states (A-4) can be written as 16 Number of states 2 n23 h 3 n 2 . (A-9) 30 References Comins, N. F. 2001, Discovering the Essential Universe (W.H, Freeman and Company New York) Eisberg, R, AND Resnick, R 1985, Quantum Physics of Atoms, Molecules, Solids, Nuclei, and Particles, (John Wiley & Sons, New York) Gordon, J. P., Zeiger, H. J., and Townes, C. H. 1955, “The Maser – New Type of Microwave Amplifier, Frequency Standard, and Spectrometer”, Physical Review, Vol. 99, No. 4, pp. 26-36. Harwitt, M, 1998, Astrophysical Concepts, (Springer, New York) Henny, M., Oberholzer, C., Strunk, C., Heinzel, T., Ensslin, K., Holland, M., Schonenberger, C., “The Fermionic Hanbury Brown and Twiss Experiment”, Science, April 9, 1999, Vol. 284. Kitchin, C. R. 1991, Astrophysical Techniques, (Adam Hilger, Bristol) Ridpath, I. 1997, A Dictionary of Astronomy (Oxford University Press, Oxford) Shu, F. H. 1982, The Physical Universe (University Science Books, Sausilito, CA) Van der Ziel, A. 1986, Noise in Solid State Devices and Circuits (John Wiley & Sons, New York) Weber, M. J. 1986, Handbook of Laser Science and Technology, Volume 1 Lasers and Masers, (CRC Press, Inc., Boca Raton, Florida) Weissman, M. B. 1988, “1/f Noise and Other Slow, Nonexponential Kinetics in Condensed Matter”, Review of Modern Physics, Vol. 60, No. 2, pp. 537-571.