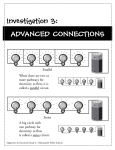
* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Grade 9
Survey
Document related concepts
Transcript
Grade 9 Word List Biological Diversity – the number and variety of organisms Species- a specific type of living organism Speciation – when a type of organism evolves into a variety of a similar species Structural Adaptation – when a species’ physical characteristics change (evolve) to better cope with its habitat. Behavioural Adaptation – when a species’ behaviour changes to better cope with its environment. (i.e. migrating or mating dances) Diversity Index – a way scientists measure an area’s biological diversity Environment – Competition – Niche – an organism’s ecological job Broad Niche – when an organism are able to adapt to a variety of environments and situations. Generalist – an organism with a broad niche Narrow Niche – when an organism has very specific needs and therefore has only adapted to one type of environment. Specialist – an organism with a narrow niche. (i.e. a koala only eats eucalyptus Symbiotic – a relationship two organisms have where they both gain benefits. Topic 3 Heritable – characteristic traits passed down from the previous generation Asexual Reproduction - occurs when only one parent passes genetic info Sexual Reproduction – when two parents pass genetic info. Binary Fission – asexual reproduction of one celled organisms Spores – a single celled reproductive structure of some asexually reproductive organisms (ferns, mushrooms, mould) Meristem – Rapidly reproducing cells at the growing tips of stems and roots Clones – offspring with the exact genetics as the parent Budding – when a piece of an organism (a bud) is released and grows into a complete organism. (sea sponges) Bacterial Conjugation – when genetic information in transferred between bacteria Zygote – the first cell of a growing organism Flower anatomy Pistil – female reproductive organ Stamen – male reproductive organ Ovule – contains the egg Pollen – male seed (sperm) Anther – part of stamen Stigma and Style – parts of the pistil Cotyledons – food source of seeds Embryo – multicellular form taken as zygote divides Topic 4 Genetics – study of heredity Continuous Variation – traits that have numerous possibilities (height, hand span) Discrete Variation – Traits with only a few possibilities (blood type, tongue rolling) Dominant Gene – characteristic trait that will always show up (black hair over blonde) Recessive Gene – trait often shadowed, but remains Mutation – changes in genetic info Mutagens – things that cause mutations (X-rays, chemicals) DNA – blueprint of genetic traits Chromosomes - bundled strands of DNA Nucleotide Pair – pairing in DNA that provides variety in the code Somatic Cell – any non-sex cell Sperm and Egg - male and female sex cells Genetic Engineering – altering DNA to gain desired traits Biotechnology – means to gaining desired traits Topic 6 Artificial Selection – gaining desirable traits (breeding dogs) Selective Breeding – actively choosing individuals to breed with desired traits Natural Selection – theory that desired traits evolve naturally within a species Topic 7 Bioindicator species – species used by biologist to assess habitat interference Grade 9 Terms Unit 2 Matter – anything that has mass and takes up space Solid – State of matter that has a definite shape and volume Liquid – state of matter that has definite volume but no definite shape Gas – state of matter with no definite shape or volume Fluids – liquids and gas because they flow Condensation, evaporation, melting (fusion), freezing (solidification), sublimation Topic 2 Pure Substance – object containing only one type of particle Mixture – contains two or more substances Element – contains one type of atom Compound – contains two or more types of atoms but still the same throughout (water, salt) Homogeneous – a mixture that is the same throughout (solutions) Solutions – the only homogeneous mixture (paint) Heterogeneous – mixtures containing two or more materials that are still visible Suspension – a heterogeneous mixture in which the particles settle slowly (oil/vinegar dressing) Colloid – heterogeneous mixture where particles do not settle, they stay dispersed (whip cream) Emulsion – colloids having an emulsifying agent (mayonnaise) Mechanical Mixture – heterogeneous mixture where the separate parts are easily seen (bird seed) Dissolving – mixing a solute into a solvent Solute – substance being dissolved Solvent – substance doing the dissolving Soluble – being able to be dissolved Agitation – shaken or stirred when dissolving Solubility – the mass of a solute that can be dissolve in a given amount of solvent to form a saturated solution Saturated Solution – one in which no more solute can dissolve Unsaturated Solution – one in which more solute can dissolve Supersaturated Solution – solution that has taken more solute than it normally would dissolve. Topic 3 and 4 Desalination – taking the salt out of water Flow Rate – the speed at which a fluid “runs” Viscosity – describing a fluids thickness Viscous – thicker liquids Topic 5 Density – the “crowdedness” of particles within a substance. How much mass per unit volume it takes up Mass – the amount of matter in a substance Volume – the amount of space a substance takes up Weight – the force of gravity acting upon an object Force – a push or pull acting on an object Gravity – a natural force causing one object to move toward another (or the Earth) Topic 6, 7 and 8 Buoyancy – the tendency for materials to rise or float in fluids Floating – when an object does not fall in air or sink in water Neutral Buoyancy – when gravity equals buoyancy Archimedes – Greek who related buoyant force and displacement of fluids Pressure – the amount of force acting on a certain area Pascal – unit of measure for pressure Incompressible – not being compressed (solids and liquids) Barometer – device used to measure air pressure Hydraulics – applying forces with liquids Pneumatics – applying forces with gases Grade 9 Chemistry and Exam Terms Chemistry – study of matter and how it changes Matter – anything that has mass and takes up space Caustic – materials that will burn, corrode or destroy organic tissue Organic – relating to living things Tissue – a group of cells that work together Mixtures – two or more pure substances together Homogeneous – mixtures where particles are uniformly scattered (solutions) Heterogeneous – mixtures where particles are not uniformly scattered (mechanical, suspensions and colloids) Pure Substances – elements and compounds Element – substance made of one type of particle and cannot be broken down Compounds – substances made of two or more elements and can be broken down Mass – amount of matter in a substance Definite – fixed or exact Proportions – parts of Conservation – with respect to reaction, amounts stay the same Chemical Change – occurs when a new substance forms Physical change – no new substance forms, only physical characteristics change Electrolysis – breaking apart compounds using electricity Atoms – Smallest form of matter that can exist on its own Sub-atomic particles – particles that make up atoms Electron – negatively charged particles that surround the nucleus of an atom Nucleus – centre of Proton – positively charged sub-atomic particle that partially makes up the nucleus Neutron – portion of an atom’s nucleus with no charge Electron Cloud – area above an atom’s nucleus where electrons encircle Atomic Symbol – letter symbol on Periodic Table that represents an element’s name Atomic Number – number given to an element on the Periodic Table to represent the amount of protons within its nucleus Metals – elements with the properties of: lustre, malleable, ductile and with the ability to conduct heat and electricity Non-metal – elements without these properties Metalloid – elements with some properties of a metal but act chemically as a non-metal Malleable – able to be hammered out into a sheet Ductile – able to be drawn out into a wire Conduct – able to have something (heat or electricity) move through it Chemical Family – group of elements with similar properties Alkali Metals – metals in group 1 with one electron in their outer shell and are very reactive Alkaline Earth Metals – metals in group 2 that not as reactive but still very reactive as they have two electrons in their outer shell Halogens – Non-metals in group17 wanting one more electron to stabilize and therefore are very reactive Noble Gases (aka Inert) – elements in group 18 with full outer electron shells that are not reactive Atomic Mass – average mass of an element’s atom Periodic Table – chart used to organize elements Ionic Compounds - compounds made from the bonding of a metal with a non-metal, that form ions and conduct electricity Molecular Compounds – compounds formed from the bonding of two non-metals that form molecules and are not good conductors Ion – a charged particle Chemical Formula – symbols and numbers used to represent the composition of pure substances Binary Compound – compound made of two elements Chemical Reaction – when two or more substances undergo a reorganization of atoms Reactants – Substances going through the reaction Products – substances produced by the reaction Chemical Equation – writing a chemical reaction using symbols to represent reactants and products Chemical Bond – forces keeping atoms together Exothermic – a reaction that gives off energy Endothermic – a reaction that requires energy Reaction Rate – how fast a reaction occurs Catalyst – a substance that speeds up a reaction without being used up itself Enzyme - a catalyst made by living things Corrosion – oxidation of metals or rocks Oxidation – corroding in the presence of air or water Rust – a particular type of corrosion dealing with iron Galvanization – coating metals with zinc to prevent corrosion Combustion – an exothermic reaction of burning a hydrocarbon in the presence of oxygen Hydrocarbon – a compound made primarily of hydrogens and carbons Electricity Vocabulary Electricity – a physical phenomena occurring from the behaviour of electrons and protons caused by attractions and repulsions. Mechanical Energy – energy provided by moving parts Chemical Energy – energy provided by a chemical reaction Thermal Energy – energy provided by heat Electrical Energy – energy provided by the movement of electrons Static Electricity – the build up of charge Current Electricity – the constant flow of electric charge Electric Charge – a build up of protons or electrons Balanced Charges – equal numbers of protons and electrons Unbalanced Charges – an excess of either protons or electrons causing a positive or negative charge Atoms – smallest form of matter that exists on its own Sub-atomic Particles – particles making up atoms Protons – positively charged particles found in the nucleus of atoms Electrons – negatively charged particles found orbiting an atom’s nucleus Neutrons – sub-atomic particles with no charge Negative Charge – an excess of electrons Positive Charge – an excess of protons Neutral – an equal amount of protons and electrons Laws of Charges (3) – 1) opposite charges attract one another 2) like charges repel one another 3) charged particles attract neutral ones Attract – bring closer Repel – push away from Conductor – a substance that allows electricity to flow through it (give examples) Insulator – a substance that does not allow electricity to flow through it (give examples) Superconductor – a substance that allows electricity to flow freely with little to no resistance Semiconductor – a substance with resistance that reduced the flow the electricity Grounding – connecting an object to Earth to safely rebalance a charge Electrostatic Buildup – accumulation of charges Circuit – a closed circular line consisting of a source and conductors Load – a device on a circuit that changes electricity into another source of energy Source – device on a circuit that provides the electric current Circuit Diagram – an illustration of a circuit using specific symbols Cell – a device that delivers electric current due to a chemical reaction Battery – a current producing device containing more than one cell Switch (or control) – a device that breaks and closes a circuit Lamp (light) – a device that transforms electric energy into light energy Resistor – a device that resists the flow of electricity, often changing it into another form Conducting Wire – drawn out metals that allow electricity to flow in a circuit Nichrome Wire – wire often used in labs that allow some electricity to flow but produces much resistance Current – the amount of electric charge flowing past a specific circuit point per unit time (also give its symbol) I Potential Difference – the amount of energy per unit charge needed to move a charged particle from a specific point on a circuit to another (also give its symbol) V Resistance – restricting the flow of electric charge (also give its symbol) R Time – a measurement of “how long” it takes to do something Ammeter – a device used to measure larger currents Galvanometer – a device used to measure weak current Amps – unit used to measure current Volts – unit used to measure potential difference (voltage) Ohms – unit used to measure resistance Ohm’s Law – resistance is the ratio of potential difference over current R=V/I Home Voltage Value – 120 V Resistance Math Equation – R = V / I Problem Solving Triangle for Resistance Equation – V R I Rheostat – a continuously variable resistor used to regulate current Variable Resistor – a resistor that changes – regulating current Polygraph – electrical device used to determine if a person is lying Series Circuit – a circuit where electricity has only one path to take - the current stays the same throughout but the voltage can chage Parallel Circuit – a circuit with more than one branch for electricity to take - the voltage stays the same but the current can chage Factors Affecting a Wire’s Resistance – length of wire, cross section, temperature, material used Energy – the ability to do work Thermocouple – a device often using two different metals at two different points that measures a temperature difference and when one side is heated, produces current Thermo-electric Generator – based on a thermocouple, a device that converts heat energy into electric energy Piezoelectric crystal – a crystal that when squeezed or pulled causes vibrations that produce a current. Light Emitting Diode (LED) – electronic components that glow when electricity paces through them. They use semiconductors and therefore use less power. Photovoltaic Cell – a source of electricity that uses light energy to excite electrons and move them, thus creating electricity. Electrochemical Cell – a source of energy that converts chemical energy into electric energy (battery). Electrode – metal conductor in a cell or battery. Electrolyte – substance that conducts an electric current (liquid or paste) Primary Cell – a non-rechargeable cell. The amount of chemicals dictates the amount of energy it can output. Secondary Cell – a rechargeable cell. These use chemical reactions that can be reversed. Wet Cell – an electric energy source (cell/battery) that uses liquid as its electrolyte. Dry Cell – an electric energy source that uses a paste as its electrolyte Fuel Cell – another term for an energy source (battery) Generator – a device that converts mechanical energy into electric energy Motor – a device that produces motion St. Louis Motor – a motor that uses a changing magnetic field to move its parts Magnetism – physical phenomena involving the attracting and repelling of various metals Electromagnet – a device that uses electric current to create a magnetic field Coil of Wire – wire that has been curled Alternating Current (AC)- current that moves back and forth Direct Current (DC) – current that moves in one direction only Dynamo – a generator that produces direct current Commutator – circular metal of a machine connected to the current carrying coil Armature – rotating part of a dynamo Brush – sliding connection completing a circuit between a fixed and a moving conductor. DC Motor – a motor working in direct current AC Motor – a motor working in alternating current Rotor – the rotating core of an AC motor Stator – stationary component surrounding the rotor Transformer – devices that either “step up” or “step down” voltage when it travels Circuit Breaker – a safety switch that can cut off power to the home if the current exceeds a safe level. Fuse – a device that contains a metallic conductor that melts when excessive current heats it up Transistor – electronic switches in digital devices Electric Power – energy per unit time (provide symbol) P Mathematical Equation for Power – P = IV Problem Solving Triangle for Power Equation – P I V Watt – unit measuring power Kilowatt – one thousand watts. Generally what is used to measure power in houses. Kilowatt Hours – unit of electric energy; the amount of energy transmitted by one thousand watts of power over one hour Electric Efficiency – the relationship of energy inputted to outputted Efficiency = energy output -----------------energy input Flare Gas – waste gas from natural gas facilities Incandescent Bulb – light bulb that uses a metal (tungsten) that glows when heated Halogen Bulbs – filled with gas that contains iodine that helps the filament from evapourating Fluorescent Bulbs – uses gases such as mercury vapour that emits energy when heated. Short Circuit – occurs when two bare wires touch resulting high current flowing between them and often sparking Renewable Resource – energy sources that can be replenished (wood, biomass) Non-renewable Resource – resources that are consumed faster than nature can replenish them (fossil fuels) Open Pit Mining – mining for fuels by digging a large hole Scrubbers – used in factories to clean emissions, usually sulfates Greenhouse Gases – gases that contribute the greenhouse effect (carbon dioxide) Global Warming – the consistent increase in the globes average temperature. Sulfates – pollutant emissions with sulfur as its main element Nitrates – pollutants with nitrogen as its main element Carbonates – pollutants with carbon as its main element Hydro-electricity – producing electricity using the movement of water Nuclear Fission – splitting uranium into two smaller atoms Nuclear Fusion – joining smaller atoms to make larger ones Thermonuclear – using heat in a nuclear power plant Thermo pollution – occurs when warm water from a power plant is released into a lake/river altering the temperature of the environment Cogeneration – generating stations that produce electricity and thermal energy. Geothermal Energy – thermal energy from the Earth Solar Energy - energy from the Sun. (light energy) Space Terms Frame of Reference – your viewpoint based on your location in the universe Celestial Body - anything in space Constellations – a group of stars that make a pattern. Used for early navigation. Stars – large balls of gases that are exploding. Our sun is one example. Navigation – mapping or orienteering Planets – rocky or gassy – they orbit stars to make Solar Systems Moons – rocky or gassy - smaller bodies that orbit planets. Earth has one moon. Satellites – ANYTHING that orbits something else. Our moon is Earth’s satellite, Earth is a satellite of the Sun and the FOX network has TV satellites orbiting Earth. Azimuth – the angle of measurement measured clockwise from north (N, E, S, W) Altitude – a measurement of a body in degrees above the horizon Altitude-Azimuth Co-ordinates – co-ordinates that determine the placement of a celestial body in the sky based on your frame of reference. Horizon – imaginary line in the distance when you look across Earth Astrolabe – an upside down protractor type tool that measures angles of altitude Compass – a tool used to find direction Geocentric – Earth centred Heliocentric – Sun centred Telescope – a tool used to view things far away Refracting Telescope – a telescope that uses lenses to bend light to see Reflecting Telescope – a telescope that uses mirrors to bounce light Ocular Lens – the eyepiece of a telescope Aperture – area of a telescope that allows light to enter Resolving Power – allows for greater detail Ellipse – “oval” shape of planetary orbit Orbit – moving around a larger body due to gravity Universal Gravitation – all bodies in the universe have gravity and are attracted to all other bodies Gravity – force that pulls bodies toward each other Astronomical Units (AU) – measurement of distance equal to the distance from the sun to the Earth Diameter – distance across a circular object at its equator Spectrum (Electromagnetic Radiation Sepectrum) – variety of waves from radio through visible light to gamma rays Spectroscope – took used to get a spectral analysis of stars Spectral Lines – bar code that tell you what elements are in stars Spectroscopy – study of spectral lines Diffraction Grating – produces the spectral lines Spectral Analysis – interpreting the lines of a spectrum Astronomers – people who stud space Doppler Effect – when observed frequency of waves change as the source moved toward or away from the observer Red Shifted – when spectral lines move toward the longer red section of the spectrum Blue Shifted – when spectral lines move toward the shorter blue side Adaptive Optics – technology that adjusts the images in telescopes Triangulation – using three points to determine the distance away an object is Parallax – the angle created by the space between your eyes – creates imperfections in measurements Light year – the distance light will travel in one year Radio Waves – shorter waves sometimes used in imagery Interferometry – technology connecting two or more telescopes Very Long Baseline Interferometry (VLBI) – inferometry over long distances Rocket – a tube with combustable material at one end and payload at the other Atmosphere – invisible barrier surrounding Earth Payload – the device a rocket is carrying Exhaust Velocity – speed at which exhaust leaves a rocket Staged Rocket – a rocket with more than one part that separates at different times in the launch Ballistic Missile – a rocket with a bomb as a payload Astronaut – person traveling is space Cosmonaut – term for a Russian astrnaut Sputnik – first satellite to make it to space (Russian) Charged Couple Devices (CCD) – converts light signals to electric signals Hubble Telescope – large telescope out in Earth’s orbit Artificial Satellites – orbiting bodies put there by humans (TV satellites) Geosynchronous – orbiting around Earth Geosynchronous orbit – same Remote Sensing – taking measurements of Earth from space Global Positioning System (GPS)- using satellites to determine positions on Earth Solar System – a group of planets and star(s) Solar Wind – high energy particles streaming off the Sun Voyager – a specific space mission (shuttle) Apollo – another mission (shuttle) International Space Station – artificial body in space, for astronauts, for many countries Canadarm – robotic arm used in space and develop by Canadians Microgravity – zero gravity Environmental Chemistry Terms Chemistry – the study of matter and how it changes. Nutrients – molecules or elements required by an organism Molecules – two or more elements boded together Elements – a pure substance made of one type of particle Organic Molecules – carbon-based molecules (often can biodegrade and are ‘natural’) Inorganic Molecules – not carbon-based (often metals) Carbohydrates – organic molecules used as energy sources in nutrition. Proteins – organic molecules used for structure and repair. Lipids – organic molecules used as energy storage. Vitamins – organic molecules used in enzyme function Minerals – inorganic molecules that make enzymes among other functions Enzymes – natural chemicals that speed up the metabolic reaction Metabolism – the bodies processing and using of food. Macro – large [as in marcromolecule (large molecule) or macromineral (large mineral)] Micro – small Nitrogen – elemental nutrient. In plants used for Potassium – elemental nutrient. In plants used for Phosphorus – elemental nutrient. In plants used for Fertilizer – chemicals and nutrients added to soil to enhance plant growth. Pesticides – Chemicals used to kill of unwanted organisms Herbicides – chemicals used to specifically eradicate unwanted plants Insecticides – chemicals used to specifically eradicate unwanted insects Fungicides – chemicals used to specifically eradicate unwanted fungae Ppm / ppb – parts per million / billion – amount of a chemical found in an ecosystem. Equivalent to 1mg / 1000L DDT – specific example of a pesticide having drastic affects on the environment. Biological Magnification or Biomagnification – the increase of concentration of chemicals as it moves through a food chain. Food Chain – a pathway showing “what eats what” and the direction of energy flow. Pesticide Resistance – the ability of pests to resist chemicals over time. Organic Agriculture – growing crops or maintaining livestock without the use of chemicals. Acids – chemicals with a pH lower than 7 and produce hydrogen ions Base – chemicals with a pH higher than 7 and produce hydroxide ions. Neutral pH – exactly 7 on the pH scale pH Scale – measuring scale that determines chemicals as acidic or basic Indicators – tools used to highlight pH indicators – indicators determining acids and bases Litmus – plant compounds used in acid/base indicating (blue means base) Acid Precipitation – when sulfate, nitrate and carbonate molecules combine with water in the atmosphere and fall as precipitation with a lower pH than normal precipitation. Neutralization – combining acids and bases and gaining a pH of 7. Catalyst – Molecules used to speed up chemical reactions Oxidation – when oxygen combines with of chemicals to produce other molecules Catalytic Converter – mechanisms in vehicles to reduce pollution Scrubbers – in factory stacks used to clean pollutants Sorbent – substances that absorb or capture oxides. Exam Terms Differences - variations Variation - differences Interdependence – relationship between Graphical Representation – showing stats in graph form Advantages – benefits Disadvantages – drawbacks Rate – speed – how fast Dissolves – mixed within Excerpt – piece of Unrelated – not related Prevent – resist Stimulant – provides a rush Inorganic – not living Concentrated – most intense Yield – production (crops) Responding variable – the one that the experiment depends on Approximate – about or rough estimate