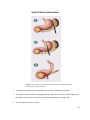
* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Sleeve Gastrectomy as a Surgical Tool for the Treatment of Obesity
Survey
Document related concepts
Adipose tissue wikipedia , lookup
Fat acceptance movement wikipedia , lookup
Saturated fat and cardiovascular disease wikipedia , lookup
Body mass index wikipedia , lookup
Epidemiology of metabolic syndrome wikipedia , lookup
Overeaters Anonymous wikipedia , lookup
Thrifty gene hypothesis wikipedia , lookup
Cigarette smoking for weight loss wikipedia , lookup
Human nutrition wikipedia , lookup
Food choice wikipedia , lookup
Abdominal obesity wikipedia , lookup
Childhood obesity wikipedia , lookup
Obesity and the environment wikipedia , lookup
Diet-induced obesity model wikipedia , lookup
Obesity in the Middle East and North Africa wikipedia , lookup
Transcript
TOPIC SELECTION Obesity has always been a fascinating subject to me. I have never been overweight nor have any of my family members been overweight. Therefore, I cannot relate to someone who is obese, however, I can empathize with his/her situation. I understand that it is not simply a matter of lacking self-control or being lazy. Obesity is a disease state just like cancer or diabetes, requiring a multidisciplinary approach to treatment. I couldn’t quite understand why obese individuals would choose to have major surgery when behavior and dietary habits need to be changed anyway. Why can’t they just change behaviors before hand and avoid surgery? By choosing to research a bariatric procedure, I had hoped to gain an understanding of this. The main reason I went back to school to be a registered dietitian was to work with the obese population. I originally wanted to become a weight management counselor at a local health club but have gained more and more interest in bariatrics. Part of why I chose this case study topic was to learn more about bariatric surgical procedures and to see if this truly is something I would like to specialize in (and it is). INTRODUCTION According to a recent study performed by the CDC, 34% of adults in the United States, aged 20 and over, were found to be obese in 2005-2006. This amounts to approximately 72 million Americans (1). What’s even more alarming than this is the global reach of obesity. In 2005, the World Health Organization (WHO) estimated that there were at least 400 million obese adults throughout the world and WHO further predicts that this number will jump to at least 700 million by the year 2015 (2). Obesity is no longer a disease seen only in industrialized nations. It is now prevalent in underdeveloped countries, especially in urban areas (3,2). Obesity is a very complex disease that still is not fully understood. What we do know is that obesity is influenced by many factors including environmental, biological, psychological, cultural, socioeconomic and genetic factors (4,5,6). It is a chronic disease associated with morbidity and mortality. The Centers for Disease Control (CDC), reports that there are approximately 400,000 deaths per year attributable to obesity related conditions (7). 1 There are several treatment options for obesity including behavior modification, diet therapy, exercise therapy, pharmacotherapy and bariatric surgery. Most non-surgical weight loss programs have proven ineffective at sustaining long-term weight loss (8,9). A recent systemic review of nonsurgical weight loss programs revealed high costs, attrition rates, and the probability of regaining at least 50% of the lost weight in 1 to 2 years (9). Research shows that bariatric surgery is the only effective treatment for morbid obesity and can result in improvement or complete resolution of obesity related co-morbidities (10,11,12). There are three types of bariatric surgical procedures: restrictive, malabsorptive and combined (restrictive and malabsorptive). Sleeve gastrectomy (SG) is a restrictive procedure that removes a large portion of the stomach, leaving a “sleeve” of a stomach that holds 2-3 ounces (13). Because there is no malabsorption involved, and the stomach functions normally, there are rarely any nutritional deficiencies and complications are minimized (14). Through the use of laparoscopic surgery, incisions are smaller, recovery time is quicker and hospital stay is reduced. SG was originally the first step in a two-stage procedure for the super-obese but has recently gained popularity among surgeons as a standalone procedure (15). Short-term studies of excess weight loss show promising results, however, there are no long-term studies to indicate efficacy beyond three years (16). WHAT IS OBESITY AND HOW IS IT DEFINED? As defined by WHO, obesity is an “abnormal or excessive fat accumulation that may impair health” (2). Excessive fat accumulation is the result of an imbalance between energy intake and energy expenditure (17). Excess energy is stored in fat cells of adipose tissue. As long as energy intake continues to exceed energy output, fat cells will grow in size. This is known as hypertrophic obesity (3). Fat cells are capable of expanding as much as 1,000 times (18). Once fat cells reach their maximum storage capacity, new fat cells are generated. This is known as hyperplasia obesity. (If an individual loses weight, he/she can reduce the size of the fat cells but cannot reduce the number of fat cells (3). Clinically, an individual is considered to be obese if he or she has a Body Mass Index (BMI) greater than or equal to 30 (19). BMI is a non-gender-specific measurement of a person’s weight in relation to height and is correlated with body fatness (3,17,20). Although it does not directly measure body fat percentage, it is a better marker of obesity than weight itself. BMI can be used to assess an individual’s 2 health risks associated with overweight and obesity; as BMI increases, health risks tend to increase (20). The National Institutes of Health (NIH) established the following guidelines to classify obesity (19): Table 1. NIH Classification of Overweight and Obesity CLASSIFICATION Underweight Normal Overweight Obesity, class I Obesity, class II Extreme obesity, class III BMI kg/m2 <18.5 18.5-24.9 25.0-29.9 30.0-34.99 35.0-39.9 >= 40 The location of fat on the body is also of great concern. Women typically collect fat in their hips and buttocks, giving them a “pear” shape, called gynoid obesity. Men usually build up fat around their bellies, giving them more of an “apple” shape, called central, abdominal or android obesity (14). Excess abdominal fat is an important, independent risk factor for disease. Research has shown that waist circumference is directly associated with abdominal fat and can be used in the assessment of the risks associated with obesity or overweight. If you carry fat mainly around your waist (central/abdominal fat), you are more likely to develop obesity related health problems (15). Central obesity is closely associated with heart disease, stroke, diabetes, hypertension, and some types of cancer (3). Women with a waist measurement of more than 35 inches and men with a waist measurement of more than 40 inches may have more health risks than people with lower waist measurements because of their body fat distribution (21). WHO ARE THE OBESE? Obesity has increased in men and women of all ages, races and ethnicity in the United States (Figure 1). However, minority populations experience the greatest prevalence of obesity (22). This appears to be the result of lower levels of education and income (17). Prevalence of obesity is higher among women than men. The oxidation of basal fat (the rate at which fat is broken down) is lower in women than men which could be a contributing factor to the higher incidence of obesity among women (3). 23% of white women, 31% of Hispanic women, and 37% of Black women are obese (22). Metabolic rates are lower in black women as opposed to white women, which could explain their increased prevalence of obesity (23). The prevalence of overweight and obesity in elderly adults in the US is high. 75% of men and 66% of women aged 60 or above are overweight or obese (24). 3 Figure 1. Source: Center for Disease Control (25) CAUSES OF OBESITY Simplistically, obesity is caused by an energy imbalance in which energy intake exceeds energy expenditure. For every 3500 kilocalories (kcals) consumed above individual needs, one pound of fat is created and stored in adipose and muscle tissue through the actions of the enzyme lipoprotein lipase (LPL). LPL activity in fat cells of obese individuals is higher than leaner individuals. As a result, even small increases in excessive intake results in greater storage of fat (3). This leads to hypertrophy and hyperplasia of fat cells. Unfortunately, obesity is much more complex than this. There are various influencing factors leading to excessive energy intake and limited or no energy expenditure. 4 Environment The evolution of humans created a biological adaptive response to starvation as a protective mechanism of body fat in times of energy scarcity (26). Hunters and gatherers of years long ago had a labor intensive existence. As time went on, technological advances were made that reduced the amount of manual work required for daily activities. The advent of industrialization paved the way for the proliferation of food availability, fast and convenience foods and energy-saving devices experienced today (22,26). Our biological response to store excess body fat is no longer essential for survival in modern-day society. Ironically, it is now counterintuitive to survival as high-calorie foods and inactivity lead to obesity and its associated diseases. The authors of Handbook of Obesity Treatment summed it up best: “biology permits obesity to occur in individuals, but the environment causes obesity in the culture (27).” Obesity prevalence has increased along with the changes in our food supply (17). We have seen a surge in high-fat and/or high-calorie snacks, meals, and beverages as well as larger portion sizes. This change does not support healthy eating habits (21). According to the surgeon general, only 3% of people in the US meet four out of five recommendations for intake of grains, fruit, vegetables, dairy products and meats (6). Food security and efficiency in food production has evolved at the cost of overall declining nutritional health (26). Over the last twenty years, portion sizes have increased significantly (Table 2). A typical hamburger in 1957 weighed 1oz and provided 210 kcals. Today, that same hamburger weighs 6oz and provides 618 kcals. Studies have shown that when people are given larger portions, they eat more. Compared to the 1970’s, average daily food intake has increased by at least 200 kcals per day (28). Assuming energy expenditure does not exceed intake that would lead to a twenty pound weight gain over the course of one year. 5 Table 2. Portion Comparison 20 Years Ago Additional Calories Consumed Today Portion Calories Portion Calories Hamburger 1 ounce 210 6 ounces 618 408 Bagel 3'' diameter 140 6'' diameter 350 210 Spaghetti w/meatballs 1 cup sauce 500 3 sm meatballs 2 cups sauce 1,020 3 lg meatballs 520 Soda 6.5 ounces 82 20 ounces 250 168 Blueberry muffin 1.5 ounces 210 5 ounces 500 290 Source: adapted from: http://www.nhlbi.nih.gov/health/public/heart/obesity/wecan/learn-it/distortion.htm The average American consumes over 3500 kcals per day (8). Most consumers have no idea how many calories they are actually consuming each day. It is very difficult to keep track when portion sizes are larger than recommended serving sizes. For example, a dinner at the Macaroni Grill could cost you 3,330 kcals (Table 3). Based on a 2,000 calorie diet, this is approximately 65% more than daily needs in just one meal! Table 3. Dinner at Macaroni Grill ITEM CALORIES 1 bowl pasta fagioli soup (appetizer) 760 Penne with oven roasted chicken (main entrée) 1330 Tiramisu (dessert) 1000 20 oz soda 240 Source: http://www.calorie-count.com/calories/manufacturer/1577.html There has been a shift in the types of beverages consumed. Our ancestors relied on two sources, water and breast milk. Today, there are numerous beverages on the market containing high-fructose corn syrup which have little or no nutritive value. These sweetened high-calorie beverages are partly responsible for the rise in obesity. Studies have shown consumption of these beverages do not reduce the amount of calories consumed from food in proportion to the amount of calories consumed from 6 beverages. In addition, intake of high-calorie beverages is actually increased as the portion size of food eaten is increased. Carbohydrates, the major component in sweetened beverages, do not stimulate as many “satiety factors” as fat or protein. One theory to explain this is that our evolutionary physiological adaptation to water and breast milk has made it difficult to process the carbohydrates found in beverages (29). Modern society has changed the types of foods we eat, how much we eat and where we eat. Fat in foods, which is characteristic of the American diet, increases the palatability of food. It is easy to overeat fatty food items because they do not produce the same feeling of satiety that carbohydrate rich foods do. Fats are easily hidden in foods. Calories can add up very quickly as calories derived from fat are denser than calories derived from protein or carbohydrates sources (30). Table 4. Caloric Value of Macronutrients Nutrient Calories/gram Carbohydrate 4 Protein 4 Fat 9 Many convenience food items are hydrogenated to preserve freshness. Foods that are hydrogenated contain trans-fatty acids (TFA). TFA’s are also found in hydrogenated vegetable oils used to fry many fast food items. The risk of weight gain increases with high intakes of TFA’s, possibly by producing a biological effect that promotes abdominal obesity (31). Food is available almost anywhere you go, not just in supermarkets or restaurants. In today’s competitive market, drug stores contain a full line of groceries. Even video stores sell candy, chips and beverages. Vending machines provide easy access to calorie dense drinks and snacks, which can be found just about anywhere: in schools, gas stations, office buildings, airports, and hotels. Several fast food restaurants, diners and supermarkets are open 24 hours a day. It’s no wonder that the average weight gain in the US is .5-1.0 kg/year (5). 7 A greater percentage of meals are now eaten or purchased outside of the home. Since the 1960’s, it has been reported that American households doubled their food budget spent on restaurant meals (8). These meals are generally rich in fat and simple carbohydrates which contribute to weight gain. Dietary fat is efficiently converted to body fat (17) and excess energy from carbohydrates leads to weight gain by suppressing fat oxidation (the break-down of fat) (18). Technology has created a very sedentary society. Cars, computers, television and video games all help to contribute to our “couch potato” lifestyles. Today, more people drive long distances to work instead of walking (21). And once at work, people may spend 8-12 hours a day in front of a computer screen. If people are not exercising daily to make up for sedentary behaviors, energy intake will be greater than energy expenditure, which will lead to weight gain over an extended period of time. Biology There are approximately 40 hormones that have been found to play a role in the regulation of intake and body weight (Appendix A). For the purpose of this paper, I will focus on two of the major regulators, leptin and ghrelin. Leptin is a protein that has been discovered to play a role in the regulation of intake and body weight (28). It acts like a hormone by signaling the brain to decrease food intake when energy stores are filled. Adipose tissue releases leptin into the circulatory system in response to energy stores. Leptin then travels through the blood brain barrier and into the brain, creating a feedback mechanism to suppress appetite and increase energy expenditure. As weight gain and body fat increase, leptin levels increase (3,28). Obese individuals tend to have high levels of leptin (3). So why don’t they lose weight? Researchers believe that obese people experience leptin sensitivity similar to that of a diabetic experiencing insulin sensitivity. It is postulated that leptin levels rise to compensate for this sensitivity (17). Ghrelin is also a protein that acts like a hormone, playing a role in the regulation of intake and body weight. However, ghrelin acts in opposition to leptin by stimulating appetite and decreasing energy expenditure (3,17). Lack of sleep produces ghrelin, which stimulates appetite and creates less leptin, which in turn, also increases appetite (32). Obese individuals may have trouble getting a good night’s sleep due to sleep apnea. Ghrelin levels are higher in females than males which may help explain why the prevalence of obesity is higher among females. A decrease in plasma ghrelin levels is seen post 8 gastrectomy (28). This gives an advantage to patients undergoing bariatric surgery involving gastrectomy. Socioeconomics Level of education and income play a role in the prevalence of obesity. Obesity rates are highest among those of a lower socioeconomic class. Low-income individuals are associated with lower levels of education. Knowledge of nutrition is inadequate. People are unaware of the harm they are doing to themselves with high-fat, high-calorie diets (they are likely not even aware of how many calories or grams of fat are in the foods they eat). Low-income families are at a disadvantage because they do not have the financial means to purchase gym memberships or healthy food items. It is often cheaper to purchase and prepare processed foods (21). Low-income families are more likely to take advantage of “supersizing” fast food items because it appears to be a better value financially. Access to nutritious food may not be available. Families may depend on the “corner store” for groceries because supermarkets are not within walking distance. “Corner stores” do not carry an array of nutritious foods. They mainly stock convenience food items that are high in fat and calories. The type of neighborhood you live in may contribute to the development of obesity. Neighborhoods can be without sidewalks, parks, recreational facilities, after-school programs, and community centers, making it difficult to achieve an optimum level of physical activity (21). Unsafe neighborhoods may force residents to stay inside as often as possible, limiting their mobility and energy expenditure. Psychology Individual behaviors can lead to the development of obesity. Binge eating is a reported problem in 30% of obese individuals seeking treatment (17). It is often used as a coping mechanism for stress or depression. Binge eating disorder (BED) is characterized by the consumption of large amounts of food in a discrete period of time where the individual feels he/she has no control over eating during the episode (17). It is not associated with any other behaviors such as purging, fasting , or excessive exercise. Other behaviors that can lead to overeating and obesity are: Eating when you’re bored, nervous or not hungry Eating while watching television “Super-sizing” meals 9 “Cleaning your plate” at every meal Skipping breakfast Fad / restrictive diets Consuming high caloric beverages and/or snacks Not getting enough sleep Substituting food for other addictions (i.e. smoking) Intake of energy-dense, nutrient poor foods (18) Certain drugs used to treat depression, psychiatric conditions and seizures can cause weight gain by increasing appetite, causing fluid retention or lowering metabolic rate (Table 5) (15). Tricyclic and heterocyclic antidepressants may increase appetite and cravings for carbohydrates and sweets. These drugs can also cause side effects such as dry mouth and increased thirst which may cause an individual to seek relief from soft drinks and hard candy. Long-term use of steroids may lead to weight gain as well (17). Table 5. Drugs Associated with Weight Gain Antidiabetes agents o Insulin o Sulfonylureas o Thiazolodinediones Antihistamines o Cyproheptidine Antihypertensive agents o Beta adrenergic blockers (proprandolol) o Alpha-1 adrenergic blockers (terazosin) Psychiatric/neurological agents o Antipsychotic agents Thioridazine Olanzapine 10 (Table 5. Drugs Associated with weight gain cont’d) Clozapine Risperidone Quetiapine o Mood stabilizers – lithium o Antidepressants o Amitriptylinle nortriptyline imipramine mitrazapine monoamine oxidase inhibitors paroxetine Antiepileptic drugs gabapentin valporate carbamazepine Steroid hormones o Contraceptives o Glucocorticoids o Progestational steroids Source: ADA’s Nutrition Care Manual (33) Culture If you ask most people whether they eat to live or live to eat, they will tell you the latter. Our culture is centered on food. Go into any office and you are bound to find a jar of candy. We have cake for birthdays, assorted fat-laden appetizers and dessert for parties and holidays, buttered popcorn for watching movies (jumbo size, of course), salty snacks for watching television, and the list goes on. 11 People often eat out in celebration of a special occasion. Families no longer gather around the dinner table the way they used to. Meals are eaten at the computer, in the living room while watching television or at fast food restaurants. A majority of women now work outside of the home and do not have time to cook a nutritious meal. It is easier to pick up dinner at a fast food restaurant; you don’t have to shop, prep, cook or clean. Large portions have also become part of our culture. All of these extra calories add up quickly and can contribute to obesity if we’re not careful. Our current lifestyles are burdened with stress. Many Americans do not get the recommended number of hours of sleep each night and are sleep deprived. Even if we do get to bed at a decent hour, we have trouble sleeping because of stress. Although a causal link has not been found, sleep deprivation and stress have been associated with obesity (26). Genetics Obesity tends to run in families, suggesting a genetic cause. However, families also share diet and lifestyle habits that may contribute to obesity (21). Some people store more energy as fat in an environment of excess; others lose less fat in an environment of scarcity. The different responses are largely due to genetic variation between individuals (34). Separating genetic from other influences on obesity is often difficult. Even so, science does show a link between obesity and heredity (21). Family and twin studies have shown that genetic factors contribute 40-70% to the variation in common obesity (36). However, genes are not destiny (34). There are some genetic disorders that lead to obesity: Hypothyrodism – a condition in which the thyroid gland fails to produce enough thyroid hormone. It often results in lowered metabolic rate, which can lead to weight gain (21). Cushing’s syndrome – a hormonal disorder caused by prolonged exposure of the body’s tissues to high levels of the hormone cortisol. Symptoms vary, but most people have upper obesity, rounded face, increased fat around the neck, and thinning arms and legs (21). Polycystic ovary syndrome (PCOS) – an endocrine disorder characterized by hyperandrogenism and insulin resistance. Symptoms include irregular or missed menstrual cycles, multiple ovarian cysts, infertility, acne, hair growth and hair loss. PCOS is closely associated with obesity, mainly android obesity. People with PCOS often have eating disorders (18). 12 Praeder-Willi – a genetic disorder characterized by excessive appetite, massive obesity, short stature, and often mental retardation (3). HEALTH RISKS ASSOCIATED WITH OBESITY? Obesity is associated with numerous medical conditions such as cardiovascular disease (CVD), hypertension, pulmonary difficulties, certain types of cancers, chronic musculoskeletal problems, infertility, conditions resulting from intraabdominal pressure, fatty liver disease and type II diabetes (36,37). Data from NHANES III show that morbidity for a number of health conditions increases as BMI increases in both men and women (38). The degree of obesity increases the severity of co-morbidities (18). Some are debilitating and others can actually be life-threatening. According to the surgeon general, individuals who are obese have a 50-100% increased risk of premature death from all causes compared to normal weight individuals; and morbidity from obesity may be as great as from poverty, smoking, or problem drinking (6). Cardiovascular Disease (CVD) The risk of death in the morbidly obese is increased fivefold from cardiovascular disease. The risk of heart failure increases 5% for men and 7% for women with each increment of 1 kg/m2 in BMI (9). Inflammation, which can be caused by substances produced by excess body fat, may raise heart disease risk (39). Other obesity related risk factors contributing to heart disease are high blood pressure, high levels of triglycerides and low-density lipoprotein (LDL), and low levels of high-density lipoprotein (HDL) cholesterol (9). These risk factors, along with elevated blood glucose and central obesity, in combination are known as the “metabolic syndrome.” The National Cholesterol Education Project Adult Treatment Panel III has defined the metabolic syndrome as having at least three of the risk factors previously noted. The metabolic syndrome is a strong risk factor for CVD (9). According to a publication by the National Institute of health, a person with metabolic syndrome has approximately twice the risk factor for coronary heart disease and it is estimated that 27% of American adults have the metabolic syndrome (39). The heart of a morbidly obese person is forced to work harder due to an increased circulating blood volume that is associated with morbid obesity. As the heart works harder, the cardiac muscle gets larger. This causes structural changes leading to congestive heart failure which decreases the function of the heart. Structural changes can also lead to arrhythmias. Obesity is associated with a 50% increase 13 in the risk of developing atrial fibrillation secondary to left atrial enlargement (37). Structural and functional changes may also be the result of fat infiltration of the heart (9). This can alter the hemodynamic load or lead to metabolic abnormalities, inflammation or coronary artery disease. Obesity also increases the risk of stroke and peripheral vascular disease (40). Hypertension Prospective studies have shown that obesity increases the risk of developing hypertension. In the Framingham Offspring Study, 78% of cases of hypertension in men and 64% in women were attributable to obesity (41). Hypertension is defined as a persistently high arterial blood pressure, where systolic blood pressure is above 140 mm Hg or diastolic blood pressure is above 90 mm Hg (18). Because of the American diet, obese individuals are more likely to have high cholesterol levels. When the body has more cholesterol than it needs, cholesterol begins to build up and form plaque on artery walls, narrowing the passage of blood flow. The heart has to work harder to circulate blood throughout the body. This raises blood pressure, resulting in hypertension. Hypertension can also be caused pulmonary dysfunction, another obesity related co-morbidity (42). Pulmonary Dysfunction Obesity may compromise pulmonary function in the following ways: Excess fat externally and internally compresses the thoracic cavity Fatty infiltration of accessory breathing muscles decrease compliance of chest wall Increased intraabdominal pressure causes cephalad displacement of diaphragm, resulting in diminished lung volume and suboptimal pulmonary dynamics Increased pulmonary blood volume competes for space in chest cavity, further decreasing lung volumes (42) The functional issues listed above can lead to obstructive sleep apnea (OSA) and obesity hypoventilation (37). OSA is characterized by loud snoring, daytime sleepiness, and frequent night-time awakening. While asleep, people with OSA stop breathing for short periods of time. This is caused by a collapsing pharyngeal airway (43) from the pressure of fat around the neck when an individual lies down. The prevalence of obstructive sleep apnea in morbidly obese patients is as high as 71% (37). OHS, commonly 14 called the pickwickian syndrome, is defined as having a BMI greater than 30 kg/m2 and an awake PaCO2 greater than 45 mm Hg in the absence of a known cause for hypoventilation (42). Reactive airways in response to the pro-inflammatory state present in obese individuals may lead to asthma. Chronic aspiration associated with gastroesophageal reflux, a common condition among the obese, may also lead to asthma by creating hypersensitive airways (42). Cancer Obesity is associated with several types of cancers including prostate, breast (postmenopausal), esophageal, endometrial, colorectal, gall bladder and thyroid (in women) cancer (43). The surgeon general reports that: “women who gain more than 20 pounds from age 18 to midlife, double their risk of postmenopausal breast cancer, compared to women whose weight remains stable (44);” in women with a BMI of 34 or greater, the risk of developing endometrial cancer is increased by more than six times (6). The American Cancer Society found that in 2002, 51% of newly diagnosed cases of cancer among women and approximately 14% of newly diagnosed cases among men were linked to obesity. The etiologies are not fully understood. It is postulated that esophageal cancer arises from gastric reflux, which is often experienced by obese individuals. Since studies have shown that physical activity reduces the risk of colorectal cancer, it is hypothesized that physical inactivity, which is common among the obese, increases risk. Osteoarthritis The excess weight of an obese individual increases the stress placed on joint bones and cartilage. Overtime, the bone begins to wear down and cause pain due to the deterioration of cartilage which protects the bone (21,39). This is evident in weight-bearing joints such as the knees, hips, spine and ankles (37). According to the surgeon general, for every 2-pound increase in weight, the risk of developing arthritis is increased by 9 to 13%. The risk for osteoarthritis may be increased by inflammation commonly found in obese individuals (39). Reproductive Complications Obesity can cause reproductive problems in women such as menstrual irregularities and infertility. There are also several complications of pregnancy related to obesity. There is a higher risk of death for the unborn baby and mother. The maternal blood pressure can increase by as much as 10 times. High 15 blood pressure during pregnancy is called pre-eclampsia. If it is left untreated, it can cause severe problems for the mother and baby. Obese women who are pregnant are likely to develop gestational diabetes. Complications with labor and delivery are also increased. Infants born to obese women are at higher risk for birth defects such as neural tube defects and spina bifida (45) stillbirth, prematurity and being large for gestational age (39). Conditions related to intraabominal Pressure Intraabdominal pressure is caused by excessive abdominal girth (40). As pressure increases on the surrounding abdominal organs, risk of various associated conditions increases. Gastroesophageal reflux disease (GERD) is one such condition. When the lower esophageal sphincter does not close properly, contents of the stomach may leak back into the esophagus often causing heartburn (GERD can also be asymptomatic) (21). Overtime, GERD can erode the lining of the esophagus, causing a condition known as Barrett’s esophagus, which could potentially, although rare, develop into esophageal adenocarcinoma. There are several reasons why reflux may occur: a reduced lower esophageal sphincter (LES) pressure, increased frequency of transient LES relaxations, esophageal and gastric motility disorders or the presence of a hiatal hernia. Symptoms of GERD are more common in obese individuals than lean individuals (40). Other conditions associated with intraabdominal pressure are: Stress overflow urinary incontinence Venous stasis disease Nephritic syndrome Pulmonary embolism Pseudotumor cerebri Thrombophlebitis (38) Fatty Liver Disease (nonalcoholic steatohepatitis) Fatty liver disease occurs when fat builds up in the liver cells and causes injury and inflammation in the liver, which can lead to severe liver damage, cirrhosis or liver failure. Obese people are likely to develop diabetes; people who have diabetes are more likely to have fatty liver disease (39). Studies have found that obesity increases the risk for fibrosis and cirrhosis in patients with alcoholic liver disease (40). 16 Type 2 Diabetes Type 2 diabetes is a disorder of carbohydrate metabolism characterized by hyperglycemia and disordered insulin metabolism, usually resulting from inadequate or ineffective insulin (3). In a study by Hu, van Dam, and Liu (2001), findings indicate that a higher intake of saturated fat and trans-fat adversely affect glucose metabolism and insulin resistance (23). Over 80% of people with diabetes are overweight or obese (44). Obesity can directly cause some degree of insulin resistance (3). Being overweight puts added pressure on the body’s ability to properly control blood sugar using insulin and therefore makes it much more likely for an obese individual to develop diabetes (45). According to the National Health and Nutrition Examination Survey (NHANES, 1999- 2002), 51% of those with diabetes had a BMI of 30 or more (46). Data from NHANES III showed that the prevalence of diabetes increased from 2% in those with a BMI between 25 and 29.9 kg/m2 to 13% in those with a BMI of 35 kg/m2 or higher (40). The number of diabetes cases among American adults jumped by a third in the 1990’s. This rapid increase is due to the growing prevalence of obesity and extra weight in the US population (45). Diabetes by itself is a risk factor for the following medical conditions which can be life threatening: Diabetic retinopathy (eye disease) Diabetic nephropathy (kidney disease) Diabetic foot disease Neuropathy Heart disease Peripheral vascular disease Cerebrovascular disease Ketoacidosis Hyperosmolar syndrome Hypoglycemia (13) Other medical conditions associated with obesity include poor wound healing, gallstones, gout and increased risk for surgical complications. 17 Non-Medical Risks Associated with Obesity The quality of life of an obese individual is less than satisfactory. Obese individuals experience limited mobility and decreased physical endurance, difficulty with personal hygiene, limited selection in clothing (10), seats that do not accommodate their large size, low self-esteem, negative self-image, hopelessness (22) and social stigmatization (18). Obese individuals are thought to be lazy people who lack self control. They face discrimination in all areas of life such as relationships, education, and employment. Obese individuals face biased attitudes from medical personnel. This may lead to the avoidance of obtaining medical care as needed. Research has shown that obese patients are more likely to delay or cancel appointments to avoid negative experiences. Obese individuals who experience weight stigmatization have higher rates of depression, anxiety, social isolation, and poorer psychological adjustments (47). ECONOMIC COSTS ASSOCIATED WITH OBESITY Overweight and obesity, and the health problems associated with each, have substantial economic consequences for the US health care system. The increasing prevalence of overweight and obesity is associated with both direct and indirect costs. Direct health care costs refer to preventive, diagnostic and treatment services related to overweight and obesity. Indirect costs refer to the value of wages lost by people unable to work because of illness or disability, as well as the value of future earning lost by premature death (6). In 1995, the total costs attributable to obesity in the US amounted to an estimated $99 billion. In 2000, the total cost of obesity in the US was estimated to be $117 billion. Most of the cost associated with obesity is due to type 2 diabetes, coronary heart disease, and hypertension (6). The WHO estimates the total cost of obesity at 2%-7% of total health care costs worldwide (8). These increases can be felt in insurance premiums, deductibles and co-payments. It has been shown that obese adults face 37.4% greater annual medical expenditures than normalweight individuals (8). In 2002, the Medical Expenditure Panel Survey reported that individuals over the age of 55 who were obese incurred higher mean medical expenses ($7,235) when compared with normal weight individuals ($5,390) (7). 18 TREATMENT OPTIONS FOR OBESITY Treatment options for obesity are just as complex as the disease itself. Most interventions focus solely on individual dietary consumption and physical activity (5), which is not an appropriate strategy for success. The treatment of obesity requires lifestyle modification which consists of a multidisciplinary approach to weight loss. Changing one’s lifestyle requires behavior therapy, diet therapy and exercise therapy in combination with one another. If these therapies prove unsuccessful, pharmacotherapy may be introduced. After this, surgical intervention may be considered. What is success? Obese individuals seeking to lose weight generally have unrealistic goals, which can lead to disappointment, frustration, self-blame and poor self-esteem, all of which can cause someone to give up, and possibly gain even more weight. The traditional goal of achieving one’s ideal body weight (IBW) is no longer considered acceptable (17). The goal should be one of health and not cosmetic. It is more important to reduce the medical risks associated with obesity than to look model thin which is unattainable and unhealthy. Studies show that as little as a 5-10% loss of initial weight can reduce risk factors of morbidity and mortality (21,39). Now that’s success! Behavior Therapy Behavior therapy provides methods for overcoming barriers to compliance with dietary therapy and/or increased physical activity (38). It is designed to promote long-term change (8) that will support weightloss goals and maintenance of weight lost. Behavior therapy can provide a structured lifestyle change, methods to modify diet and physical activity, cognitive techniques for attitude change, and strategies for increasing social support (17). Methods to modify eating and activity behaviors may include: Self-monitoring eating – involves recording the how, what, when, where and why of eating to provide an objective tool to help identify eating behaviors that need improvement. Also, the act of recording food eaten causes people to alter their intake. Self-monitoring activity, which includes the frequency, intensity, and type of activity performed, is also useful. Stress management – involves using strategies such as meditation and relaxation techniques to lower stress, which may improve eating behaviors. 19 Stimulus control – involves avoiding or changing cues that trigger undesirable behaviors (e.g., keeping “problem” foods out of sight or out of the house) or instituting new cues to elicit positive behaviors (e.g., putting walking shoes by the front door as a reminder to go walking). Problem solving – involves identifying eating problems or high-risk situations, planning alternative behaviors, implementing the alternative behaviors, and evaluating the plan to determine whether or not it reduces problem eating behaviors. Contingency management – involves rewarding changes in eating or activity behaviors with desirable nonfood dividends. Cognitive restructuring – involves reducing negative self-talk, increasing positive self-talk, setting reasonable goals, and changing inaccurate beliefs. Thoughts precede behavior; changing thoughts and attitude can change behavior. Social support – involves getting others to participate in or provide emotional and physical support of weight loss efforts. Source: adapted from “Nutrition Essentials for Nursing Practice, 5th edition (22) Behavior therapy requires gradual change toward realistic goals. Too many changes at once are overwhelming and ultimately lead to failure; unrealistic goals can cause frustration, also leading to failure. Behavior strategies need to be tailored to the individual. Behavior therapy will only be successful if the patient is motivated to change present undermining behaviors. It is the clinician’s responsibility to assess a patient’s level of motivation. Any barriers to change must also be assessed. 20 Table 6. Assessment of Motivation and Change Reasons and motivation for weight loss Previous history of successful and unsuccessful attempts at weight loss Support system Attitude toward physical activity Capacity to participate in physical activity Time available to commit Financial consideration Barriers to success Source: adapted from Kaplan & Sadock’s Comprehensive Textbook of Psychiatry, 8th Edition. (17) Participants of standard group therapy lose an average of 18 pounds in 15-20 weeks. Longer therapy can lead to weight loss of up to approximately 30 pounds (22). After one year of treatment, patient’s regain approximately one-third of weight lost. This can be reduced or avoided by keeping in contact with patients via follow-up visits, phone calls, or e-mails. Patients will also benefit from attending support groups. Diet Therapy Diet therapy should be used to safely achieve weight-loss goals. A reasonable goal is a loss of one to two pounds per week. A dramatic weight loss in a short period of time is hard to maintain. It may also result in the loss of lean muscle mass. A deficit of 500 kcals per day is required to lose one pound per week; a deficit of 1,000 kcals per day is required to lose two pounds per week. This can be done by reducing intake or by burning the equivalent number of kcals via physical activity. The latter is more 21 difficult to sustain long-term . It would be more beneficial to supplement a reduced calorie diet with exercise. Exercise Therapy Physical activity is a very important part of a weight loss program and should be used along with diet and behavior therapy. There are numerous benefits in addition to weight loss. Physical activity can: Raise HDL levels Improve cardiovascular function Release endorphins that make you feel good Relieve stress Help you sleep better at night Increase endurance Since most obese individuals are not used to an active lifestyle, they should start off with light physical activity and increase intensity over time as tolerated. A healthy and reasonable goal to strive for is at least 30 minutes of moderate-intensity physical activity on most or all days of the week. A good place to start is walking for just 10 minutes a day, 2-3 days per week. Behavior modification can be used to increase physical activity. For example, choosing stairs over an elevator or walking instead of driving. The important thing is to keep moving! Pharmacotherapy Pharmacotherapy should only be considered when an overweight or obese individual has not successfully been able to lose one pound per week in the first six months of a weight loss program that includes behavior modification, diet therapy and exercise (22). Prescription weight-loss drugs are approved only for those with a BMI of 30 and above or a BMI of 27 and above with any of the following co-morbidities: HTN, dyslipidemia, CHD, type II diabetes, OSA. Not all people benefit from or respond to drug therapy. Anyone who fails to lose 4.4 pounds from a weight-loss drug in 4 weeks is not likely to respond with subsequent use (48). Drug treatment should therefore be discontinued (22). Drug treatment by itself is not effective and should never be used as the only source of weight-loss. It should be used to help patients comply with their diet and physical activity plans while losing weight. Weight-loss drugs could potentially induce as much as a 10% reduction in initial body weight (17), 22 mainly within the first six months of treatment. However, weight loss drugs may only work for as long as they are being used; benefits may be lost upon cessation (38). Therefore, short-term use of antiobesity drugs are not very effective. It is not known how long drug therapy can be used; to date, there are no studies indicating the safety of long-term use (22). There are currently only two drugs that have been approved for long-term use by the Food and Drug Administration (FDA): sibutramine and orlistat. Their safety and efficacy have not been established for use beyond two years (46). Sibutramine is an appetite suppressant that works by inhibiting the reuptake of serotonin and norephinephrine. Clinical trials show daily use of 10-15 mg of sibutramine, in conjunction with diet therapy, will provide a weight loss of 4-10% of initial body weight, with losses maintained for up to 2 years (17). Reported side effects include increases in pulse rate, blood pressure, plasma epinephrine, plasma glucose and other more common side effects include headache 24,50), dry mouth, constipation and insomnia (30). Use of sibutramine is contraindicated in patients with uncontrolled HTN, narrow angle glaucoma, history of CAD, CHF, arrhythmias, stroke, severe renal impairment and severe hepatic dysfunction. Sibutramine should also not be used if patients are currently taking monoamine oxidase inhibitors (MAOI’s), or selective serotonin reuptake inhibitors (SSRI’s) such as antidepressants or migraine medications, or centrally acting appetite suppressant medications (17). Orlistat binds to gastric, pancreatic, and carboxyl ester lipases in the gastrointestinal tract and blocks the action of these lipases on dietary triglycerides and vitamin esters. The inhibition of fat digestion decreases micelle formation and the absorption of long-chain fatty acids, cholesterol, and certain fatsoluble vitamins (30). Approximately one-third of dietary fat is malabsorbed and excreted in stool (30,48). Studies show that a regimen of 120 mg of orlistat, three times a day, in addition to diet therapy, promotes a weight-loss of 5-13% of initial body weight, with losses maintained for up to two years (17). Studies also show that carbohydrate and protein intake is increased, possibly due to an indirect stimulation of the central hyperphagic signaling mechanism through lipase inhibition. This can lead to rapid regain of weight lost (49). Orlistat is generally well tolerated but it is not without side effects. If an individual consumes more than 30% of calories from fat, he or she may experience any of the following: abdominal pain, fecal urgency, liquid stools, flatulence with discharge, oily spotting, and incontinence (17,30). Use of orlistat may require supplementation of fat soluble vitamins that are lost through fecal excretion. Benefits of orlistat include: higher levels of HDL, drop in total cholesterol 23 levels, can be used in patients with HTN and cardiovascular complications (17), and helps minimize weight regain after weight loss (23). Certain drugs used to treat conditions other than obesity have been associated with short-term weight loss. They have been approved for use by the FDA for the conditions in which they were intended, but not for use as an anti-obesity drug. These drugs are being used “off-label” to treat obesity, meaning they are not being used for their intended purpose (48). These drugs include bupropion, topiramate, zonisamide and metformin. Table 7. Off-label Drugs Prescribed for the Treatment of Obesity Generic Name Intended Use of Treatment Bupropion Depression Topiramate Seizures Zonisamide Seizures Metformin Diabetes Common Side Effect Dry mouth, insomnia Numbness of skin, change in taste Drowsiness, dry mouth, dizziness, headache, nausea Weakness, dizziness, metallic taste, nausea Source: adapted from NIH Publication No.07-4191. (48) Bariatric Surgery The first bariatric procedure was performed in 1953 at the University of Minnesota. Although this endto-end jejunoileostomy procedure was associated with severe diarrhea, electrolyte imbalances and liver failure, which mandated the reversal of the procedure, it paved the way for future bariatric procedures. By the year 1990, approximately 16,000 bariatric surgeries were being performed annually in the US (50). In 2006, the American Society for Metabolic & Bariatric Surgery (ASMBS) reported that 177,600 people in the US had bariatric surgery; this represents less than 1% of people who actually qualified for surgery (46). It is estimated that by the year 2010, 218,000 bariatric surgeries will be performed (8), over a 1300% increase in just twenty years! Bariatric surgery is indicated for an individual with a BMI greater than or equal to 40 or a BMI greater than or equal to 35 with co-morbidities such as CVD, HTN and diabetes, and should only be consider if there have been multiple failed weight-loss attempts through an interdisciplinary weight-loss program. 24 Individuals should be highly motivated and willing to make changes for life. Bariatric surgery is only a tool to assist in weight-loss, it is not a “magic bullet. “ Without proper nutrition and exercise, weight loss from surgery will not be permanent. Candidates for surgery should be carefully screened preoperatively to ensure they are psychologically and medically stable. This requires an interdisciplinary approach including physicians, psychologists and dietitians. A complete medical history should be obtained along with a physical exam and laboratory tests (i.e. complete blood count; pulmonary, cardiac, and liver function tests). There are several bariatric surgical procedures being performed today, but the most popular procedures are the gastric bypass roux-en-y, which is the gold standard in the US (51), and the gastric adjustable lap band. Other bariatric surgical procedures include biliopancreatic diversion with duodenal switch and sleeve gastrectomy. These can be performed as open surgery or laparoscopically. Most procedures today are done laparoscopically, unless otherwise contraindicated (contraindications can include patients who are extremely obese, who have had previous abdominal surgery or complicated medical problems). Laparoscopic procedures require smaller incisions which reduces tissue damage and complications, resulting in a shorter hospital stay postoperatively (52). Surgical procedures can be categorized as restrictive, malabsorptive or combined. Restrictive procedures induce weight loss by combining a small gastric pouch with a narrow outlet, limiting oral intake and evoking early satiety (14). Malabsorptive procedures induce weight loss via by-passing segments of the gastrointestinal tract where various nutrients are absorbed (Figure 2). Nutrients that are not absorbed as a result of malabsorption are excreted during defecation. This can lead to vitamin and mineral deficiencies, including iron, vitamin B12, folate, calcium, and the fat-soluble vitamins (A, D, E, K) (53). Protein deficiencies are also of great concern following bariatric surgery. Restrictive procedures, although not malabsorptive, can also lead to deficiencies because of limited intake. Food intolerances that occur postoperatively can decrease intake as well. 25 Sites of Nutrient Absorption in the GI Tract Duodenum Jejunum Ileum Stomach Water Calcium Thiamin Calcium Ethyl alcohol Iron Riboflavin Phosphorus Copper Phosphorus Niacin Magnesium Iodide Magnesium Pantothenate Iron Fluoride Copper Biotin Zinc Vit D Molybdenum Selenium Folate Chromium Vit K Intrinsic Factor Thiamin Vit B6 Manganese Magnesium Riboflavin Vit C Molybdenum Niacin Vit A,D,E,K Amino acids Biotin Di-,tripeptides Vit C Folate Vit B12 Bile salts/acids Folate Vit A,D,E,K Figure 2. Source: Adapted from Clinical Nutrition for Surgical Patients (53) Roux-en-Y Gastric By-pass (RYGP): malabsorptive and restrictive RYGBP (Figure 3a) works by restricting food intake and by decreasing the absorption of food. Food intake is limited by a small pouch (36) that is about the size of an egg (1-2 ounces) (13). In addition, absorption of food in the digestive tracts is reduced by excluding most of the stomach, duodenum, and upper intestine from contact with food by routing food directly from the pouch into the small intestine (52). Patients experience an excess weight loss of approximately 70% (13). 26 Adjustable Gastric Band (AGB): restrictive AGB (Figure 2b) works primarily by decreasing food intake. Food intake is limited by placing a small bracelet-like band around the top of the stomach to produce a small pouch that is similar in size to the RYGB. The outlet size is controlled by a circular balloon inside the band that can be inflated or deflated with saline solution to meet the needs of the patient (51). Generally, a weight loss of approximately 5560% of excess weight is achieved (13,55). Biliopancreatic Bypass Diversion(BPD) with a Duodenal Switch(DS): malabsorptive BPD, (Figure 3c) usually referred to as a “duodenal switch,” is a complex bariatric procedure that includes removing the lower portion of the stomach and creating a gastric sleeve with the small pouch that remains. That pouch is connected directly to the small intestine, completely bypassing the duodenum and upper small intestine from contact with food. BPD produces significant weight loss; approximately 80% of excess weight) (13,55). However, the mortality rate is higher than with other bariatric operations and there are more long-term complications because of decreased absorption of food, vitamins and minerals (52). Figure 3 (a), (b), (c). Source: Reprinted with permission from the Bariatric Institute of Wisconsin; http://www.bariatricwi.com/surgery_types.asp 27 Sleeve Gastrectomy (SG): restrictive SG is usually performed as the first stage of BPD with DS on GBP in patients who may be at high risk for complications from more extensive types of surgery. These patients’ high risk levels are due to body weight and/or existing medical conditions. A SG operation restricts food intake and does not lead to decreased absorption of food. However, most of the stomach is removed, which may decrease production of a hormone called ghrelin. A decreased amount of ghrelin may reduce hunger more than other purely restrictive operations, such as gastric banding (52). (See section Laparoscopic Sleeve Gastrectomy for a more in-depth look at this procedure.) Approximately 10% of bariatric patients may have unsatisfactory weight-loss or regain most of the weight lost. Inadequate weight loss can be caused by a stretched pouch (from dilatation or dietary noncompliance) or separated stitches (52). Complications that may occur from bariatric surgery include (13,52): EARLY STAGE COMPLICATIONS Bleeding Infection Vomiting Dumping syndrome (malabsorptive procedures) Anastomosis leaks Blood clots in legs (which can travel to heart of lungs) LATE STAGE COMPLICATION Malnutrition Strictures Hernias o Incisional o Internal The overall mortality rate from bariatric surgical procedures is approximately 0.5% (37). The leading cause of death postoperatively is pulmonary embolism (52). But surgery can be well worth the risk. It can resolve or greatly improve type II diabetes in the majority of patients almost instantly (9). Many other co-morbidities associated with obesity may be resolved or improved including CVD, OSA, GERD, 28 asthma, gout, hypercholesterolemia, and hypertension (Figure 4). Bariatric surgery significantly improves the quality of one’s life. Figure 4. Source: http://www.asbs.org/Newsite07/patients/benefits.htm 29 LAPAROSCOPIC SLEEVE GASTRECTOMY Approximately 2% of the bariatric surgeries performed in the US are the laparoscopic sleeve gastrectomy (LSG) (56), which is quickly gaining popularity among bariatric surgeons as a safe procedure for weight-loss. This restrictive procedure got its start in 1988 as a modification to the biliopancreatic diversion (BPD) with duodenal switch (DS) (57). About 8 years ago, some surgeons who were performing the BPD with DS operation began to do the SG as a first-stage for super obese and poor-risk patients, intending to do the second-stage later. They discovered that some patients lost enough weight with the SG that they did not need to undergo the DS (56). Surgeons began using SG as the sole bariatric operation. Mechanism of Action LSG is a purely restrictive procedure in which 60-85% of the stomach, including the fundus, is removed, creating a stomach that measures 1-5 ounces. The fundus is the main production site of the hormone ghrelin, which stimulates appetite (15). A reduction of ghrelin should theoretically decrease food intake. Although this has not been proven, many surgeons believe this is one of the reasons for the success of LSG. The fundus is also the portion of the stomach that stretches the most. Removal of the fundus leaves a long, vertical, tube shaped stomach that is least likely to expand over time (59). Some surgeons believe that distention of the gastric antrum due to food accumulation in this area after excision of the fundus, may be the dominant mechanism of action by decreasing hunger and increasing post prandial sensations of satiety, thereby reducing food intake (57). Increased satiety may also be related to the increased pressure within the lumen of the sleeved stomach with its pylorus intact (15). Since the pylorus remains intact after surgery, the stomach functions normally, without malabsorption. Indications/Contraindications As previously noted, SG was initially used as a first-step in BPD with DS. It can also be used as the first step to RYGBP. It is generally indicated as a first-step procedure for patients with a BMI greater than 50 (super obese). When an individual’s size is of this magnitude, risk of surgery is much greater. LSG is a less complicated procedure than RYGBP and BPD with DS. The goal is for the patient to lose some weight before undergoing a riskier bariatric operation. Research recommends a wait time of 6 months to 18 months, given adequate weight loss, before performing the second-stage surgery. The super 30 obese may also require a second-stage procedure because weight loss from LSG in this population may not be significant enough. As with any bariatric surgery, according to the 1991 NIH Consensus Statement, surgery is indicated for anyone with a BMI >= 40 or a BMI >= 35 with one or more co-morbid conditions (16). The criteria for LSG is widening (57); it is now being offered to patients with a low BMI and low risk as an alternative to laparoscopic gastric banding. LSG may also be indicated for the following individuals (59,11,15): Those who are concerned about the potential long term side effects of an intestinal bypass such as intestinal obstruction, ulcers, anemia, osteoporosis, protein deficiency and vitamin deficiency Those who are considering laparoscopic gastric banding but are concerned about a foreign body or worried about frequent adjustments or finding a band adjustment physician Those who have experienced a failed gastric banding Those with severe medical illness such as cirrhosis, Chrohn’s disease, and anemia (contraindications for GBP) Those needing long-term anticoagulation Those requiring the use of anti-inflammatory medications (in GBP, these medications are associated with a very high incidence of ulcer) Those requiring endoscopic surveillance for chronic gastric diseases Those with volume eating disorders Those with a low BMI needing restrictive surgery LSG is contraindicated in morbidly obese individuals who have sweet-eating disorders (13). Remember, LSG is a restrictive procedure only; there is no malabsorption and no dumping syndrome involved. LSG for a person with a sweet-eating disorder would be counter-intuitive. Technically, an individual post LSG could eat small amounts of sweets throughout the day with no adverse side effects, other than weight gain. This person would benefit most from a malabsorptive surgical procedure. Preoperative Testing/Evaluation There are several tests and evaluations a patient will undergo prior to surgery at the request of the primary care physician, surgeon, insurance company and/or hospital where the surgery is to be 31 performed (13). The purpose is to assess the current health of an individual to ensure that he or she has no medical problems that would increase the risk of surgery. However, if there are medical problems related to obesity, it increases the need for surgery (60). But by knowing what medical conditions exist ahead of time, the surgeon can plan a course of action and be prepared. Testing prior to surgery may include any of the following: Physical exam Blood work o Complete blood count (CBC) – determines if anemia is present o Metabolic panel – determines how well kidney and liver work and how well body absorbs nutrients o Liver function tests – determines specific aspects of liver (bile draining, tissues) o Prothrombin time (PT) partial thromboplastin time – measures how well blood clots o Hemoglobin A1C – determines if diabetic patient has well-controlled blood sugars o Free T4, thyroid stimulating hormone (TSH) – determines function of thyroid Urinalysis – determines pregnancy status or if presence of infections, complications of diabetes or nicotine exists Cardiac Tests o Electrocardiogram (EKG) – determines if heart is working properly o Echocardiogram – an ultrasound of heart; determines how well heart is working o Stress test – determines ability of heart to stand up to stress Pulmonary Tests o Chest x-ray – shows the health of lungs o Chest CAT scan – a more detailed picture of lungs (if chest x-ray is abnormal) o Spirometry – measures the function of lungs and airways o Oximetry – measures percentage of blood that contains oxygen 32 o Arterial blood gas (ABG) – determines ability of lungs to get oxygen into blood and remove waste produced o Sleep studies – determines if sleep apnea is present Gall Bladder Tests o Ultrasound of right upper abdomen – determines if gallstones are present Upper Gastrointestinal Series – used to look at esophagus, stomach and duodenum to determine if there is any inflammation ,growths, hernias or ulcers Vaginal ultrasound – determines abnormalities of ovaries and uterus in women Psychiatric evaluation – determines patient understanding of risks and complications of surgery and ability to comply with postsurgical guidelines Nutritional evaluation – determines patient’s past weight-loss attempts and harmful eating behaviors as well as the ability to change behaviors postoperatively (13,59) Patients should be strongly encouraged to attend bariatric support group meetings prior to surgery. This will give them an opportunity to hear first-hand what to expect and to realize that there is a light at the end of the tunnel. Surgical Procedure It is important to have an understanding of the anatomy of the stomach to understand the surgical procedure. The diagram below illustrates the areas of the stomach (Figure 5). Figure 5. Source: http://www.nature.com/nrc/journal/v2/n1/images/nrc703-f2.gif 33 There are multiple technical variations to LSG (56) including the number of trocars used, bougie size, division of the antral area (which can vary from 2 cm – 10 cm proximal to the pylorus) (11,15,56,61) and whether or not to construct the sleeve first or mobilize the greater curvature first. A trocar is “a hollow cylinder with a sharply pointed end, often three-sided, that is used to introduce cannulas and other similar implements into blood vessels or body cavities (62) (Figure 6).” Trocars may also be bladeless. In laparoscopic surgery, trocars are access ports that are inserted into the abdominal cavity to allow entry of the required laparoscopic instruments. The first trocar inserted, or primary trocar, is used to place a cannula through which a laparoscope is inserted to view internal structures (63). Other trocars are used for laparoscopic hand instruments such as a liver retractor or stapler (62). Research indicates the use of five or six trocars as common practice (11,56) but use of seven trocars is also noted in the research literature (Figure 7). Use of fewer trocars may decrease cost, pain and the number of scars associated with LSG (15). Trocar/cannulae system: Figure 6 Source: http://www.righthealth.com/Health/trocars/-od-definition_wiki_Trocar-s (62) 34 Figure 7. Source: http://www.coastalobesity.com/images/Incisions-for-lapar.jpg A bougie is “a thin cylinder of rubber, plastic, metal or another material that a physician inserts into or through a body passageway, such as the esophagus, to diagnose or treat a condition. A bougie may be used to widen a passageway, guide another instrument into a passageway, or dislodge an object (64).” In LSG, a soft, plastic bougie is used as a guide to create the new, smaller gastric pouch. It is inserted down the esophagus and into the stomach and pushed against the wall of the lesser curvature of the stomach. A stapler is then used to cut away the remainder of the stomach to the left of the bougie. The bougie size is an indicator of the gastric volume that will be created. For example, a 40-Fr bougie creates approximately an 8 oz pouch. A bougie greater than 40-Fr can cause stretching of the new stomach (this leaves part of the fundus in tact, which is the stretchable portion of the stomach). However, bougie size may not always indicate gastric volume. For example, if a smaller sized bougie is used but moves away from the lesser curvature and toward the greater curvature before stapling (and this is not corrected), the new pouch will be larger than predicted because of the extra space provided. 35 Likewise, a surgeon may leave too much space to the right of the bougie while stapling, creating a larger stomach. Bougie sizes for LSG range from 32-Fr to 60-Fr. The 60-Fr is indicated when LSG is used as a first step in a two-stage procedure such as RYGBP (56). The First International Consensus Summit for Sleeve Gastrectomy, New York City, October 25-27, 2007 published the surgical procedure of LSG in Obesity Surgery, 2008 as performed by Dr. Gagner (56): Five or six trocars are used, with the surgeon standing between the patient’s legs. Gagner uses an open technique for the first trocar, establishing a pneumoperitoneum of 15 mm Hg. Then, two right trocars, a left trocar, and a midline trocar are inserted, for vision to the upper right. The right subcostal trocar is used to insert the fan retractor for the liver. The camera is placed between the umbilicus and xiphoid and has to be high in position. An orogastric tube may be passed to initially decompress the stomach, and it is then removed. Some surgeons commence with an opening through the gastrocolic ligament to lesser sac, and initially cut-staple the vertical channel. Gagner first mobilizes the greater curvature outside the epipoic arcade, close to the gastric wall, which will be removed. With light traction between two atraumatic forceps, and starting below the midpoint of the greater curvature, using LigaSure, SonoSurg, Harmonic Scalpel or coagulating hook, and the patient in slight reverse Trendelenber, the posterior stomach is visualized. Fine adhesions to the pancreas are divided and the lesser sac totally freed. Fat must be cleared off the left side of the GE junction, so that later stapling would not be compromised. Exposure must be high, defining the complete left crus. Most surgeons commence the dissection 5-10 cm proximal to the pylorus, but some European surgeons start the resection closer to the pylorus. If the dissection commences too close to the pylorus, the antral pumping mechanism will be defective, the antrum will not empty properly, and the patient may have some nausea. The linear stapling division is generally from a right trocar towards the left shoulder, with or without buttressing material, and leaves about 1 cm of fat pad along the lesser curvature (~3 cm width). This assures adequate blood supply on the lesser curvature for the sleeve. Dr. Gagner starts transecting the stomach 6 cm proximal to the pylorus and then the anesthesiologist inserts a 36-40-Fr bougie down to pylorus, if the SG is intended as the sole operation. The sleeve is started at the lower end of the crow’s foot. The procedure requires five 36 to six firings of the linear cutting stapler (60 cm long, 4.8-mm staple-height, green cartridge) to divide the entire stomach. It is important to remove all fundus to avoid regain of weight. The vagus nerves anteriorly and posteriorly are preserved for normal gastric emptying. The greater curvature portion may be extracted in a bag via a right paramedian or epigastric trocar-site enlarged to two-fingers diameter. The specimen has the shape of a comma with the fundus at the top. The staple-line is variously oversewn, and many workers do intraoperative testing via an 18-Fr Argyle tube with diluted methylene blue or air under saline using a gastroscope, with the prepyloric area compressed. With the bougie removed, Gagner reinforces each crossing-overlapping site from the stapler with an absorbable monofilament figure-of-eight suture (56). Most surgeons will insert a drainage tube into the abdominal area called a Jackson Pratt (JP) drain. This tube comes out of the body, attaches to a plastic bottle, and drains fluid from inside the stomach. If there is any infection in the abdominal area, the drain removes the fluid, keeping the patient healthy. The JP drain may be removed before the patient leaves the hospital, or the patient may go home with it (13); the surgeon determines the length of time the JP drain remains in the abdomen. 37 Vertical Sleeve Gastrectomy Figure 8. Source: Reprinted with permission from the Bariatric Institute of Wisconsin: Bariatricwi.com/surgery_types.asp 1. The gastrocolic omentum is dissected off of the stomach, freeing up the stomach 2. The sleeve is being created by stapling/removing a large portion of the stomach, beginning in the antral area, proximal to the pylorus, and firing up preceding in the angle of His. 3. The new stomach pouch is created. 38 Some surgeons may require a patient to wear leg compressors during surgery to prevent the risk of blood clots. These may also be worn postoperatively while in the hospital. An upper GI contrast study is performed by many surgeons the day after surgery (56). However, some surgeons may not perform this test until postoperative day 2. The upper GI contrast study is used to determine if there are any leaks in the new stomach pouch. A blue dye is ingested by the patient, which can be tracked and viewed by x-ray as it works its way down from the esophagus into the stomach, to see if any liquid leaks out of the abdomen. Once it is determined that there are no leaks, the patient may begin a clear liquid diet as tolerated. Advancement of the diet is based on the bariatric program guidelines and the patient’s tolerance to the diet. The surgeon or dietitian will let you know when you may advance to the next stage. Generally, the diet is advanced to full liquids within a day or two, pureed foods for the next few weeks, soft foods the next few weeks thereafter, until the patient is healed and ready for a regular diet. It could be several months before a patient advances to a regular diet. There is no set rule for advancement of the diet, each individual is different in what he/she can tolerate. For example, some individuals cannot tolerate meat for the first six months whereas others may be able to eat meat after the first month. (see the Medical Nutrition Therapy for Gastric Sleeve section for a more in-depth look at the postoperative diet). Some surgeons may prescribe proton pump inhibitors (PPI) to prevent any acid reflux (15). A medication, such as Actigall, may also be prescribed prophylactically to reduce the risk of gallstones. Research has shown that rapid weight loss can cause the development of gallstones. Most surgeons recommend that patients take a multivitamin, calcium and possibly a B12 vitamin after surgery (65). Cost of Surgery Laparoscopic sleeve gastrectomy generally costs between $20,000 and $25,000 in the US. However, this procedure is approximately 50-60% cheaper abroad. Medicaltourism.com lists the following prices outside of the US: Mexico - $11,990 Costa Rica - $8,400 India - $7,500Puerta Vallarta - $11,000 39 Postoperative Complications There are several complications that may occur if extreme caution and precision are not undertaken by the surgeon during surgery. The risk of bleeding exists the moment dissection of the gastrosplenic ligament begins. If dissection of the stomach begins too close to the pylorus, the staple line can crack and predispose the stomach to leaks. Leaks have a reported incidence of 0-1.4% up to 5.3% (66). Dissection too close the pylorus can also lead to improper emptying of the antral area, which may cause some patients to experience nausea. Early vomiting may occur if a firm bougie is not used. Dr. Greg Jossart reports a 30% incidence of early vomiting among his patients (56). Vomiting may also occur as a result of overeating. It takes time for patients to adjust to their new pouch. Creation of a long, narrow sleeve can cause stenosis; especially if there is tightness in the construction at the angularis (56). A gastric fistula can develop but is preventable with the use of an absorbable buttressing material. Another complication of LSG (and bariatric surgeries in general) is rhabdomyolosis. Rhabdomyolysis is a clinical and biochemical syndrome caused by destruction of skeletal muscles which can occur in patients with a BMI greater than 40 due to an abnormal pressure that disrupts internal cellular structures. It is a crushing injury from a prolonged stable position. Muscle enzymes should be checked postoperatively (67). There can be a delay in the emptying of the proximal gastric pouch if the excision line is swollen. If this occurs, it needs to be treated with intravenous hyperalimentation for two weeks (54). Intravenous hyperalimentation is a form of nutrition that is delivered into a vein. It is prescribed when an individual cannot use his or her digestive system. In the case of a swollen gastric pouch, it gives the stomach time to heal instead of aggravating the condition with oral intake. 40 Table 8. Risks and Complications of Laparoscopic Sleeve Gastrectomy Esophageal reflux / gastroesopageal reflux Stapling of gastric tube Bleeding liver Failure to position nasogastric tube properly Failure of staple line (leaks/bleeding) Gastric fistula Hemorrhage of spleen Ventral hernia Rhabdomyolysis Defective antral pumping mechanism Stenosis of gastric sleeve Gastric fistula Temporary hair loss Dehydration Gastric dilatation Insufficient weight loss or weight regain – may require another surgery Nausea/vomiting Delay of gastric emptying due to edema of excision line Deep vein thrombosis Dehydration Pulmonary embolism (11,13,56,57,61,67) 41 Postoperative Testing/Evaluation Careful follow-up is essential to the patient’s health and success. For the first year, patients should be seen by a member of a multidisciplinary team including surgeons, dietitians, psychologists, and primary care physicians at least every three months (68). A CBC should be completed within a few months of surgery to check for nutritional deficiencies. Weight should be monitored at each visit. If weight loss is too rapid, the patient may develop gallstones. Rapid weight loss may also cause dehydration. The clinician should be aware of the signs for dehydration and also monitor the patient for signs of depression, nausea, and vomiting. Patients may experience depression as they mourn the loss of food. They may also have trouble dealing with their new found “thinness.” Nausea and vomiting can be an indication of a more serious complication that may need to be treated. Follow-up should continue once a year after the first year to continue to monitor progress and any complications or nutritional deficiencies that may develop. Patients should strongly be encouraged to continue attending bariatric support group meetings. Keeping in touch with patients will help them adhere to the program and maintain a sense of responsibility. Results There are no long-term studies greater than 3 years that show sustained weight loss. However, shortterm results are encouraging. The American Society for Bariatric Surgery reviewed the outcomes of 775 patients from 15 published reports and found that patients with a preoperative BMI of 35 to 69kg/m2 had an excess weight loss ranging from 33-83% (67) (Appendix B). (Recall that an excess weight loss of just 5-10% improves overall health.) The review also showed resolution or improvement in type 2 diabetes, hypertension, hyperlipidemia, sleep apnea, degenerative joint disease, gastroesophageal reflux disease, peripheral edema and depression (Appendix C). So, how does LSG compare to the other bariatric surgical procedures? comparison chart of the various bariatric surgical procedures) (see Appendix D for a Research indicates a comparable percentage of excess weight loss from LSG to RYGBP at 2 and 3 years postoperatively (56). The lack of intestinal bypass, which is present in RYGBP, avoids potentially costly long-term complications such as marginal ulcers, vitamin deficiencies and intestinal obstructions (70). 42 Compared to the laparoscopic gastric banding (LAGB), the restrictive effect of food intake with LSG is similar and does not require a foreign object in the body. There are no adjustments that need to be made, unlike the band. One surgeon reported that his reoperation rate at 5 years, after 2,000 gastric banding operations, was 50%. In addition, weight loss and hunger sensation were better after LSG than LAGB. There was also significantly less vomiting with LSG than LAGB (56). Table 9. Advantages and disadvantages of laparoscopic sleeve gastrectomy ADVANTAGES Technically an easier procedure in complex/high risk patients Avoids anastomotic complications No increased risk of ulcers, strictures, intestinal complications, internal hernias Do not have to alter medications previously taken Ability to “re-sleeve” or perform a second procedure if weight loss is inadequate Less risk of vomiting No re-routing of intestines, avoids: Marginal ulcers Intestinal obstructions Anemia Osteoporosis Vitamin deficiencies Deep vein thrombosis and pulmonary embolism may be less likely to occur because it is a quicker procedure Function of the stomach remains intact (should eliminate potential for dumping syndrome) Level of ghrelin is much lower Can be used instead of GBP if multiple intraabdominal adhesions are present Can be used in patients weighing over 500 pounds Very effective as first-stage procedure for BMI > 55 kg/m2 DISADVANTAGES Lack of published evidence for sustained weight loss beyond 3 years Soft calories, such as ice cream and milk shakes, are easily absorbed and may slow weight loss Irreversible procedure Difficulty in finding good, qualified surgeons Usually not covered by insurance companies Sleeve may be stretched out weight loss may not be sustained due to: Stomach dilatation Non-dietary compliance Eating disorders Alcohol intoxication occurs faster than with other surgeries may produce GERD due to the surgical division of ligaments around the abdominal esophagus and destruction of the cardioesophageal junction May not produce anticipated weight loss results since there is no intestinal bypass may cause weight regain because there is no intestinal bypass 43 MEDICAL NUTRITION THERAPY for SLEEVE GASTRECTOMY As noted in the medical section of sleeve gastrectomy, the function of the stomach remains intact and since there is no bypass of the small intestines, there is no incidence of malabsorption. As a result, nutritional deficiencies postoperatively are rarely seen (55). However, this restrictive procedure does decrease dietary intake and has a tendency to cause intolerance towards nutrient-rich foods (53), mainly red meat, poultry and leafy green vegetables, thereby reducing macronutrients and micronutrients (14). Nutrition care for patients is controversial and recommendations vary widely (33). Most surgeons do agree on a daily multivitamin (65). Some surgeons will also recommend a calcium supplement and/or vitamin B-12 supplement (14,65). Nutritional Deficiencies Morbidly obese individuals are generally malnourished in the sense that they do not consume a wellbalanced diet and may be deficient in certain vitamins and minerals preoperatively (14). This should be addressed during the initial screening either by a physician who can have blood serum levels tested to determine deficiencies or by a dietitian who can review a food frequency questionnaire or 24-hour recall to evaluate deficiencies. A multivitamin should be prescribed preoperatively if deficiencies are present. Deficiencies of micronutrients including vitamins A, K, C and E, zinc, arginine, glutamine, copper, essential fatty acids, bromelain, and bioflavanoids may interfere with wound healing after surgery (14). Greg Dakin of Cornell, NY, found 85% of his patients to be deficient in vitamin D preoperatively. He recommends the use of calcium supplements post-operatively for replacement (56). Postoperative complications, such as excessive vomiting, although a rare occurrence in patients who undergo sleeve gastrectomy, can lead to: dehydration, protein-calorie malnutrition, and thiamine deficiency. This can result in neurologic sequelae (14). Greg Dakin had only one reported incidence of Wernicke’s syndrome due to thiamine deficiency from vomiting. In the experience of Dr. Jossart, 30% of his patients experienced early vomiting (56). Thiamine deficiency can be treated with a daily vitamin B complex with thiamine tablet in addition to the prescribed daily multivitamin. Protein-calorie malnutrition can be treated with high protein foods and/or protein supplements (i.e. protein shake). If dehydration is severe, it may require intravenous fluids to restore intravascular volume (54). Vomiting may be a sign of dysfunctional eating habits including overeating, eating too fast, or not chewing food 44 well. It is important that the root cause of vomiting be found and addressed immediately to prevent further nutritional deficiencies. Other causes of vomiting may include: Overdistention of the pouch by fluid Large volume meals Food intolerance GERD Symptomatic gallstones medications Vitamin B-12 deficiency may occur due to the smaller gastric pouch created by the sleeve gastrectomy. A smaller gastric pouch reduces acid and pepsin digestion of protein-bound cobalamines in food, incomplete release of R binders, and decreased production of intrinsic factor (14). B-12 deficiency may also be caused by inadequate intake of foods rich in B-12. A daily oral or sublingual vitamin B-12 supplement is used to correct this deficiency (a monthly intramuscular injection may be used as well). Iron deficiency may be the result of a reduced intake of red meat and poultry due to postoperative food intolerances. Menstruating females may be more susceptible to iron deficiency (33). A daily iron supplement should be taken along with vitamin C for increased absorption. Folate deficiency is also possible due to a reduced intake of green leafy vegetables; another postoperative food intolerance. A multivitamin is generally all that is needed to prevent this deficiency. Calcium and vitamin D deficiencies are also possible due to reduced dietary intake. Since the stomach’s holding capacity is much smaller and an individual reaches satiety quickly, foods containing calcium and vitamin D may not be consumed in sufficient quantities to avoid deficiencies. The American Dietetic Association (ADA) recommends 1200-1500 mg of calcium citrate daily, which should be taken in doses of 500 mg each to maximize absorption. To avoid interference with absorption, calcium supplements should not be taken within two hours of any iron containing supplements (33). Protein deficiency may occur as a result of inadequate ingestion of protein (33). This can be due to food intolerance, smaller stomach capacity, vomiting or dietary non-compliance. Supplemental protein (I.e. protein isolate powder in milk or other liquid) and consumption of protein-rich foods can help prevent this deficiency. Exercise can help reduce the occurrence of protein deficiency by building lean muscle 45 tissue. Excessive weight loss too soon can deplete protein stores by using lean muscle tissue for energy. To avoid protein deficiency, the ADA recommends a goal of 1.5 g of protein/kg of ideal body weight (33). Most physicians recommend a daily protein intake that falls within a range of 60-80 grams. Mild dehydration is common in the early postoperative period. Patients have difficulty drinking the necessary amount of fluid as they adapt to very small gastric capacities (54). There are no mathematical equations that accurately estimate fluid needs in the obese. Individuals should be encouraged to a carry around a water bottle (or other non-caloric, clear, non-carbonated fluid source) and sip throughout the day (54). Bariatric programs differ on the amount of water that should be consumed daily, ranging from 32 oz to 64 oz. The ADA recommends at least 6 cups of fluid per day, with 3 cups from high-protein liquid supplements and 3 cups from sugar-free, noncarbonated soft drinks (33). There are several guidelines that should be followed for the safety and efficacy of patients (Table 10). 46 Table 10. Dietary Don’ts of Sleeve Gastrectomy DO NOT…… Because….. Drink liquids with meals Liquids will fill the small stomach before adequate nutrition can be consumed; liquids can also move food along the GI tract more quickly, reducing a longer period of satiety and leading the individual to eat again sooner than expected. Drink liquids 30 minutes before or after meals Liquids will fill the small stomach and produce satiety before adequate nutrition can be consumed. Drink beyond fullness Drinking beyond satiety can cause nausea, vomiting and can stretch the pouch. Stretching the pouch can lead to increased intake. Straws increase swallowed air which can cause the pouch to Use straws distend and create an uncomfortable feeling. Drink caffeinated beverages Caffeine can contribute to dehydration. Drink alcoholic beverages Alcohol has no nutritive value and can contribute to dehydration. Drink sweetened, high-caloric beverages Beverages high in calories can leave little room for needed nutrients such as protein and can impede weight loss efforts. Highly sweetened beverages may also cause nausea and possibly vomiting or diarrhea Source: Adapted from the ADA’s NCM (33) 47 Nutritional deficiencies can occur from non-compliance of prescribed nutritional supplements and/or recommended dietary intake. The degree of nutritional deficiency following sleeve gastrectomy is dependent upon: Preoperative nutritional status Occurrence of complications Ability to modify eating behavior Compliance with regular follow-up Compliance with prescribed vitamin and mineral supplementation (38) Preoperative Diet Not all bariatric programs require a preoperative diet. However, there are some programs that require an individual to lose weight prior to surgery. The Center for Advanced Weight Loss at St. Francis Hospital in Trenton, New Jersey, requires patients to lose a minimum of 10-15 pounds prior to surgery or the surgery will not be performed. Weight-loss decreases the risk of surgical complications. St. Francis, as well as other bariatric programs, requires a liquid protein diet two weeks prior to surgery to improve fatty liver conditions, commonly found in morbidly obese individuals. It also helps to improve protein stores, which is important for healing postoperatively. The ADA recommends that individuals sample different protein supplements prior to surgery so they are aware of what is palatable and what is not. It may be difficult for individuals to move around the first few weeks after surgery. It would benefit the patient to stock his/her pantry with the necessary food items before surgery (33). Dietary behavior modification should also begin prior to surgery. Lifestyle changes take time and cannot be learned over night. In order to be successful, each change should be added one at a time and subsequent changes should not be added until the prior change has a become a new habit. 48 Postoperative Diet As you can imagine, the postoperative diet also varies among programs. Generally, the types of diet are as follows: Clear liquid Full liquid Pureed Soft foods Regular foods These diet types are categorized into postoperative stages. The stage and duration is determined by the surgeon. Each stage is designed to give the patient what he/she needs nutritionally, as well as to help him/her lose weight safely and effectively. It also gives the patient a chance to heal internally while getting familiar with a new, smaller stomach (13). All diets begin with a clear liquid diet day one, provided there are no leaks or other complications detected after surgery. A barium swallow is usually performed the day after surgery to determine if there are any leaks. Advancement of diet to the next stage depends on the guidelines of the program as well as the individual. If a patient does not tolerate advancement to the next stage, he/she will regress back to the prior stage until the surgeon clears him/her to advance again. To gain an understanding of the stages and advancement of diet, this paper will provide guidelines taken from the ADA’s Nutrition Care Manual for bariatric surgery (33). (Note: it is not specific to sleeve gastrectomy but is specific to laparoscopic bariatric restrictive type surgery and assumes a four day hospital stay.) Day 1: 30 cc per hour sugar-free, noncarbonated, clear liquids Crystal light or sugar-free Kool-Aid type products Low-fat, clear broths Water Patient should sip, no straws (place a 30 cc medicine cup by bedside) 49 Day 2: 60 cc per hour low-fat, low-sugar, high-protein liquids o Skim milk o Strained soup made with skim milk o Sugar-free yogurt and pudding o Bariatric liquid protein supplement no ice cream or sherbet Day 3: semi-liquid, high-protein foods o Skim milk o Strained soup made with skim milk o Sugar-free yogurt and pudding o Low-fat strained meats thinned with broth o Mashed potatoes with protein powder 1 Tbsp every 15 minutes (2 to 3 oz per hour) Day 4: Discharge nutrition (first 2 weeks) o Pt should have 3 small feedings (1/2 cup per meal) plus high-protein liquids o Pt should have High protein, low -sugar, low-fat bariatric liquid protein supplement Liquid or chewable multivitamin and mineral supplements Liquid or chewable calcium supplement Regular follow-up with the dietitian after discharge Weeks 1 & 2: Limit volume to ¼ cup (4 tbsp) at each meal Total of 5 cups fluids per day including: o 3 cups high-protein, low-fat, low-sugar Bariatric Liquid Protein Supplement Sip 1 oz (30 cc) every 15 minutes between meals 50 o 2 cups other liquids Water Sugar-free noncarbonated drinks: Crystal light Sugar-free Kool-Aid Low-calorie cranberry juice Sugar-free popsicles Broth o ½ cup yogurt or sugar-free pudding throughout the day o Vitamin and mineral supplements daily o No alcohol o No chewing gum (if swallowed, may block stomach pouch outlet) Weeks 3 & 4: Progress to a Pureed Bariatric Surgery Nutrition Therapy. o All foods must be blended to the consistency of applesauce o Wait at least 30 minutes after a meal to start fluids Eat pureed protein foods first. Protein sources include: o fish o Tender, low-fat meats and poultry o Cottage cheese o Eggs or egg whites cooked without added fat o Tofu o Avoid high fat meats: o Sausage Bacon Luncheon meats Regular cheese Meats should be: Baked Broiled 51 Braised Stop eating or drinking when full. Overfilling stretches the stomach pouch and hinders weight-loss. Being overfull may lead to nausea and vomiting, which can pull out staples or sutures. Drink 6 cups fluids daily. This should include the following: o 3 cups high-protein, low-fat, low-sugar bariatric liquid protein supplement o Sip 1 oz every 15 minutes between meals 3 cups other liquids: Water Sugar-free noncarbonated drinks: Crystal light Sugar-free Kool-Aid Low-calorie cranberry juice Sugar-free popsicles Broth o No alcohol. o Sip sugar-free, noncarbonated fluids. No straws. o No chewing gum. o Continue bariatric liquid protein supplement. o Continue to avoid fat and sugar. o Continue supplements. 4 weeks post-surgery Progress to Bariatric Soft Nutrition Therapy o This nutrition therapy has small feedings that will be followed forever. o Wait at least 30 minutes after a meal to start fluids. o Continue to eat protein foods first: fish Tender, low-fat meats and poultry Cottage cheese Eggs or egg whites cooked without added fat 52 Tofu Avoid high fat meats: o Sausage Bacon Luncheon meats Regular cheese Meats should be: Baked Broiled Braised After protein foods eat: Vegetables Eat only cooked vegetables avoid those with o excessive fiber (celery) o tough hulls o skins (peas, corn) then soft fruit canned in its own juice or bananas. o No salads or other raw vegetables. o Last, eat starchy foods including: o Breads Cereals Grains Pasta Avoid soft bread, doughnuts, and pastries (they may form a dough ball and become lodged in stomal outlet) o It is better to take small bites of toast or crackers. Stop eating or drinking when full. Overfilling stretches the stomach pouch and hinders weight loss. o Continue bariatric liquid protein supplement. o Continue to sip fluids frequently. 53 Aim for 6 cups or more of sugar-free, noncarbonated fluids Do not use a straw. o Continue vitamin and mineral supplements. o Continue to avoid sugar and sweets. These foods have many calories. o No chewing gum. o No alcohol. After 6 months: The physician may allow the patient to reintroduce other items as tolerated. In general, patients will consume 400-800 calories per day in the first few months. Because the pouch is swollen and sore from surgery, it is difficult to eat and appetite tends to be poor. Usually within four to twelve months, appetite improves and intake will approximate 1,000 to 1,200 calories per day. It is important to try and keep a calorie level no greater than 1200 per day until goal weight is attained. After this time, calories can be increased gradually until weight is stable. This calorie level will be different for everyone, depending on factors such as height and activity level (13). In order to maintain weight loss, individuals must continue to follow a nutritious diet that is low in fat and rich in lean protein sources, whole grains, fruits and vegetables. Remember, weight loss surgery is only a tool to weightloss; proper nutrition, exercise and behavior modification are key. Nutrition Assessment The nutritional assessment of a bariatric patient begins before surgery. Most bariatric programs (and insurance companies) require patients to consult a dietitian at least once preoperatively. Insurance companies want to see documentation of prior weight-loss attempts. It is also important for the dietitian to assess the ability of an individual to make necessary diet/behavior changes postoperatively. Surgery is only a tool for weight loss and should not be performed on individuals who are not willing to make lifestyle changes. 54 The ADA recommends using the Bariatric Assessment and Presurgical Education Report form to collect the data needed for assessment and to provide a guide for nutrition education (Appendix E). It is a comprehensive form that provides the following information (33): Client history Medical history related to obesity Pertinent medications/nutrition supplements Diet history 24 hr recall/food frequency questionnaire (FFQ) Disordered eating Environmental issues affecting weight Activity level Support system Anthropometrics and weight history Patient’s estimated needs Nutrition education Assessment of understanding of expected dietary/behavior changes Postoperative follow-up plans Dietitian assessment/evaluation of patient’s ability to undergo surgery The ADA provides a Nutrition and Eating Habits Questionnaire (NEHQ) (Appendix F), containing a 24 hour food recall and FFQ, which can be sent to the patient to be completed and sent back prior to the initial dietary consultation. This will give the dietitian a good understanding of the current nutritional status of the individual, provided that the information given is accurate. An important part of the nutrition assessment is to educate the patient about the nutrition guidelines that he or she will have to follow after surgery. As written in the ADA’s NCM, patients need to verbalize understanding of the following points: They will need to be on a liquid nutrition therapy while in the hospital. When they go home, they will not be able to eat anything that is not blended or pureed for approximately 1 month while the area that has been stapled heals. 55 Their new stomach pouch will be about the size of a chicken egg and they will be able to eat only about a ½ cup of food at a meal. After surgery, they will need to drink about 3 cups of high-protein liquid supplement. Because of the small size of the pouch, they will need to sip the supplement in portion of about 2 oz per hour (1 Tbsp every 15 minutes at first). They should not drink during the meal or for 30 minutes afterward. Sweets and high-fat foods are going to be off-limits. [These high calorie foods can lead to weight gain and leave little room for needed nutrients.] After eating or drinking a small amount (1/2 cup), they will feel that the small pouch is filled. They must not keep eating or drinking. To do so will stretch the pouch. Over time, their stomach will hold more and more. Unfortunately, people who do this do not get the maximum weight-loss from the procedure. In addition, some who ignore the full feeling and stretch the pouch regain weight they lost. No alcohol. It is dehydrating and has no nutrients. It is not wise to consume carbonated drinks. The bubbles may cause the pouch to stretch. It can also cause uncomfortable bloating. Straws are also discouraged because the person using a straw tends to swallow air, which leads to bloating. After a month, patients will be able to eat a soft meal plan. This means tender meats, cooked vegetables, and fruits canned in water or juice. No salads, raw vegetables, soft bread, meat with tough fibers or gristle that might block the outlet of the stomach pouch. Small bites of toast or crackers are acceptable. They will have to take nutritional supplements for the remainder of their life. o Liquid protein drink – because it is difficult to get enough protein, many people need to continue to supplement long term. o Multiple vitamin and mineral liquid or 2 chewable tablets per day. o Calcium – most people need this mineral to promote strong bones. o Vitamin B-12 – may be needed if deficiency is present. o Iron – given to menstruating women or if the patient becomes anemic. 56 Calculations for Nutrition Assessment Use the information obtained in the Bariatric Assessment and Pre-surgical Education Report to calculate: BMI IBW % IBW Resting Energy Expenditure (REE) BMI – to calculate BMI, you will need the patient’s height and weight (you should physically measure and weigh the patient). You may use the BMI table in the appendix to obtain the BMI (Appendix F). Find the patient’s height in inches and weight in pounds on the chart and find the BMI value on top that corresponds with these parameters. Note: the patient’s body weight may fall in between two BMI values. For a more accurate measure, calculate BMI mathematically (33,38): weight (kilograms) BMI = height2 (meters) If pounds and inches are used: weight (pounds) x 703 BMI = height2 (inches2) IBW – to figure out a patient’s ideal body weight, you need to obtain his/her height and use one of the following formulas based on the patient’s gender: Men: IBW = 106 lbs for first 5 feet + 6 lbs for each inch over 5 feet Women: IBW = 100 lbs for first 5 feet + 5 lbs for each inch over 5 feet For the individual under 5 feet, subtract 2 pounds for each inch under 5 feet 57 % IBW – to figure out a patient’s % ideal body weight, you need the patient’s ideal body weight and current weight. Use the following mathematical equation to obtain % IBW: Current weight (pounds) % IBW = IBW REE – use the Mifflin-St. Jeor (1990) equation to estimate REE (kcal/day) (33). You will need the patient’s weight in kg, height in centimeters (cm) and age in years. To convert height in inches to height in cm, multiply inches by 2.54. To calculate, use one of the following based on gender: Men: (10 x wt in kg) + (6.25 x ht in cm) – (5 x age in years) + 5 Women: (10 x wt in kg) + (6.25 x ht in cm) – (5 x age in years) – 161 Multiply the REE (kcal/day) by an activity factor of 1.3 for sedentary individuals. If needed, use a higher activity factor to correct for active individuals engaging in exercise or purposeful activity (33). After assessing the patient, determine the diagnosis, etiology and supporting documentation to create a PES statement that accurately reflects the current nutrition problem. Always look at the intake domain first. This statement may change with each subsequent nutrition consultation. Nutrition Intervention The ADA’s NCM categorizes nutrition intervention for bariatric surgery into preoperative, hospitalization and postoperative intervention as follows: Preoperative Intervention Give the patient a copy of the pureed bariatric surgery nutrition therapy at the initial assessment. Before the surgery, the patient may want to stock up on items allowed on the discharge eating plan. Encourage patients to test various high-protein liquid supplements to find one they like. 58 Encourage patients to purchase and try other items they will need. These include pureed meats, canned tuna, cream of wheat, and cream soups. Discuss the importance of vitamin and mineral supplements after the surgery. Discuss the importance that physical activity will play in both losing the maximum amount of excess weight and maintaining weight loss. Lead the patient to start making plans for physical activity after surgery. Provide the patient with recipes for protein supplement use. At the second preoperative visit with the patient discuss potential failure of the surgery. Some reasons patients have failed to maximize weight loss include the following: o Did not make lifestyle behavior changes o Drank liquids with meals (stretched stomach pouch) o Did not stop eating when felt full (stretched stomach pouch) o Emotional issues associated with eating (afraid to be thin) Give the patient a list of behavior strategies for avoiding overeating. Ask the patient to write his or her own list of things that may be helpful. It is important for the patient to know that he or she will need help for a couple of weeks after surgery. The patient should get up and move around as soon as possible. Reinforce the recommendations about volume of foods and fluids. Intervention During Hospitalization: Postoperative Bariatric Surgery Nutrition Therapy Monitor for nausea and vomiting. Nursing is responsible for dispensing the 1 oz fluid/hour on postoperative day 1 and 2 oz fluid/hour on postoperative day 2; reinforce the importance of sipping the fluid between meals so as not to become dehydrated. Reinforce no fluids with meals or for 30 minutes after meals. Reinforce the discharge eating plan and be sure the patient has copies of education materials. 59 Postoperative Interventions: Bariatric Surgery Nutrition Follow-Up Form Advance eating plan to pureed bariatric surgery nutrition therapy Regularly assess weight loss Always ask the patient to bring in a 3-day food record While the patient is in the office, conduct a 24-hour recall. Assess nutritional adequacy of patient’s intake for protein and fluids. Ask the patient if he or she is continuing to take supplements regularly. Reinforce the importance of the vitamin and mineral supplements, hydration, plenty of protein, stop eating when full, and making lifestyle changes. Assess current physical activity. Reinforce the importance of regular physical activity. It will aid in maximum loss of excess weight and will help with maintenance of lower weight. Use the assessment tool to formulate nutrition diagnoses. Using the PES statements, work with the patient to plan and implement specific interventions as needed. Concentrate on behavior changes. After 4 weeks, the stapled areas should be healed and the patient may progress to the bariatric soft nutrition therapy. (Note: this is from the ADA’s NCM. Advancement of diets will vary among bariatric programs. You may need to adjust information given to a patient based on this.) Nutrition Monitoring and Evaluation The ADA recommends using the Gastric Surgery Nutrition Follow-Up form to use as a guide for questions to ask at every follow-up visit. 60 The NCM divides nutrition monitoring and evaluation into 4 parts: 1. Nutrition Assessment o o 24-hour food intake recall Intake of water or other non-caloric beverages (what kind, how much) o Consumption of liquid protein supplement (what kind, how much) o Estimated total protein intake/day o Assess adequacy of supplement use (ask when and how much) o Query the patient about the following: Drinking while eating (remind to wait 30 minutes after meals to start liquids) Carbonated drinks (remind that the gas bubbles will stretch the pouch and may cause discomfort) Straws (remind that straws cause swallowing of excess air that can stretch pouch) o o Chewing gum (swallowing gum can plug up stomach pouch outlet) Monitor any nausea or vomiting Hair loss (may be sign of protein deficiency) Ethanol use Weight Current weight Weight at last visit Pre-surgical weight Total weight loss to date Percent of excess weight that patient has lost Identify any needed education Reinforce as needed based on interview questions and responses Advance nutrition therapy if indicated 2. Write nutrition diagnoses using PES statements 3. Plan nutrition intervention - lead patient in setting goals and strategies 4. Schedule follow-up appointment 61 PATIENT PRESENTATION KS was thin for most of her life. Her weight troubles began when she quit smoking fourteen years ago and was then diagnosed with hypothyroidism. Two years later, she married and became pregnant. Four months after giving birth to her first child, KS was pregnant again. Despite numerous attempts at weight-loss through the years, KS never achieved more than a 30 pound weight-loss. With each weightloss, KS regained more than she lost. KS states that she is not an overeater but craves wings, salty foods and chocolate. She is not an emotional eater and does not eat out of boredom. However, KS eats outside the home a total of 3-6 times per week (breakfast = 1-2x , lunch = 1-2x, dinner = 1-2x,). Her physical activity prior to surgery consisted of tasks at work, such as transporting patients, and keeping up with her children. About a month before surgery, KS began bike riding twice a week. I first met KS the day after surgery. She was extremely motivated to lose weight. I asked her why she had chosen sleeve gastrectomy as her surgical tool to weight-loss. She responded that she “did not want her intestines moved,” and didn’t want a malabsorptive procedure. She also did not want to experience “dumping syndrome.” KS spent 2 days in the hospital. 62 Patient Presentation Outline Pt: KS Sex: Female Age: 38 Occupation: radiology technician at Kennedy Health Systems (KHS) since September, 2007 Hours worked: 8 hrs / day, 40 hrs / week Family hx: married for 12 years has two children: 10 year old boy, 9 year old girl has one older brother parents still married; father is an alcoholic Social Hx: occasional alcohol use Quit smoking approximately 13 years ago (1 pack per day for 6 years ) Current dx: morbid obesity Past medical hx: Small hiatal hernia Gastroesophageal reflux disease (GERD) elevated cholesterol back pain hypothyroidism hx of kidney stones bladder incontinence Past surgical hx: laparoscopy in 1995 & 2000 laser eye surgery – 1999 63 tubal ligation – 1999 thermal balloon ablation – 2007 D&C – 2007 Family medical hx: maternal grandmother – obesity, diabetes maternal aunts – obese Paternal grandfather – heart disease Supplements: MVI Medications: synthroid, stopped approximately one year ago Nexium, stopped approximately 1.5 years ago Diet / Weight Hx: Highest weight: Lowest weight: 247# / 112.3kg 130# / 59.1kg Table 11. Patient reported weight hx from 1/04-1/08 DATE WEIGHT (lbs.) 1/2/04 227.5 /27/04 230.5 11/30/04 237.9 1/3/05 233 2/2/05 228.5 3/11/05 228 7/29/05 227 8/1/05 227 2/9/06 235 12/14/06 226 2/5/07 236.5 1/16/08 236 64 Wt loss drugs tried over the last 10 years: phen-phen phentermine hoodia relacore achieved 30# weight-loss; regained upon cessation Table 12. Patient reported weight-loss program attempts Year 1990’s & Type of Diet Weight watchers Duration of Diet 6 mo-1yr Wt lost 20# Duration of weight lost Couple of weeks 2007 Nutrisystem 4 months 8# n/a n/a Gym Membership n/a n/a n/a n/a Trim-Spa n/a n/a n/a n/a “The Healthy Weight” at Cooper n/a n/a n/a 2007 Hospital n/a = information not available Table 13. Anthropometrics PRE-OP 1 DAY POST-OP 2 WEEKS POST-OP 1 MONTH POST-OP 2/19/08 5/6/08 5/20/08 6/4/08 HEIGHT 5’6” 5’6” 5’6” 5’6” WEIGHT 242 # 238* 222# 219# 117-143# 117-143# 117-143# 117-143# %IBW** 170.6 166.4 155.2 153.1 BMI 39.1 38.5 35.9 35.4 WASIT-TO-HIP RATIO (WHR) *pt reported .85 N/A .85 .827 IBW **calculated using upper end of IBW (143#) 65 MEDICAL / SURGICAL COURSE Surgical Consultations 1/30/08 – initial, discussed available surgical tools to weight loss 4/16/08 – follow-up (f/u), wt. taken 5/21/08 – first post-op visit, wt. and measurements taken, resolution of obesity related conditions discussed (no reflux, no urinary incontinence), current activities evaluated (jumping rope, using 5# weights) 6/4/08 – f/u, wt. and measurements taken, review of pt’s current health ( denies n/v/d) Next f/u scheduled for August; labs will be drawn to check for vitamin/mineral deficiencies Multidisciplinary consultations for preoperative evaluation: gastroenterology assessment (2/13/08) o Physical assessment revealed: Blood pressure = 124/84 Pulse = 68 Wt = 240# Oropharynx clear No adenopathy or palpable tumors normal active bowel sounds, non-tender, non-distended pulmonary assessment (2/18/08) o pulmonary function tests – normal o may have mild sleep apnea but not enough to warrant sleep study cleared for surgery from GI perspective cleared for surgery from pulmonary perspective psychiatric assessment (2/21/08) o mental status revealed: mood was euthymic oriented to time, place, and person speech relevant and coherent no evidence of flight ideas, hallucinations, delusions, or paranoid ideation general knowledge and judgment intact 66 insight is adequate cleared for surgery from psych perspective KS was made aware by psychiatrist that surgery could lead to depression, warning signs and symptoms were discussed cardiac assessment (2/27/08) EKG – normal Echocardiogram – normal Cleared for surgery from a cardiac perspective 67 Table 14. Lab Values TEST NAME RESULT RESULT RESULT RANGE UNITS April 19, 2008 May 5, 2008 May 6, 2008 Glucose 85 132 132 70-105 mg/dL Sodium 139 134 138 133-145 mmol/l Potassium 4.0 3.8 3.9 3.3-5.1 mmol/l Chloride 104 101 105 96-108 mmol/l CO2 25 25 28 22-32 mmol/l 14.0 11.8 8.9 10.0-20.0 8 9 10 6-20 mg/dL Creatinine 0.7 0.8 0.8 0.4-1.1 mg/dL Calcium 9.3 9.3 9.0 8.4-10.0 mg/dL WBC 7.6 11.8 12.2 4.8-10.8 X10-3 RBC 4.52 4.57 4.1 4.00-5.2 X10-6 HGB 13.7 14.1 12.9 12.0-16.0 g/dL HCT 39.8 40.5 36.4 34.9-44.9 % MCV 87.9 88.6 88.7 81.0-99.0 um-3 MCH 30.3 31.0 31.5 27.3-33.1 pg MCHC 34.5 35.0 35.5 33.0-37.0 g/dL RDW 13.6 13.1 13.6 11.5-14.5 % MPV 9.3 8.7 9.2 7.4-10.4 um-3 Platelet Count 287 271 267 130-400 X10-3 Anion Gap BUN 68 Hospitalization Summary Table 15. Medication Administration Record Medication May 5, 2008 May 6, 2008 Caefazolin 2 gm/D5W 50 mL IVPB q8 X D5/RL 1000 mL IV @ 150 mL/hr q6.75 hrs X Famotidine/Pepcid 20 mg IV q12 X Heparin 5000 unit subcut q8 X Hydromorphone/Dilauded 2 mg IV q3prn Ketorolac/Toradol 30 mg IV q6 X Metoclopramid/Reglan 10 mg IV q6 X May 5, 2008 o Admitted to hospital Diet: NPO o Prepared for surgery o Surgery performed (see appendix H for surgical report) May 6, 2008 o Diet: NPO o Barium swallow performed No leaks or complications discovered Pt felt painfully full after two small sips of barium solution Dietetic assessment (see section on Nutrition Care of Patient for full evaluation) 69 o Diet advanced to bariatric clear liquids May 7, 2008 o Discharged from hospital NUTRITION CARE OF PATIENT 2/29/08 – initial pre-op assessment o Assessment: Reason for surgery – health, wt loss, keep up w/children Current motivation – children are self-sufficient, has the time to devote, good support system Client hx Medical hx related to obesity Meds/supplements Diet hx Watching portion size # of meals eaten away from home - ~6x/week Cravings – wings, chocolate salt Usual food intake Activity level Current: physical at work, keeping up with kids and 2 dogs Plans post-op: walking, purchased abdominal machine and workout video Previous attempts at weight loss (Table 12) Anthropometrics (Table 13) Weighed patient Obtained height and usual body weight (UBW) Calculated: o BMI o IBW o % IBW 70 o Diagnosis: obesity related to food and nutrition-related knowledge deficit and physical inactivity as evidenced by BMI = 39 (grade II), patient reports eating out approximately 6x/week, low-intensity physical activity o Intervention Nutrition education for sleeve gastrectomy o Changes to the stomach/GI tract o Capacity of post-op stomach o Anticipated weight loss o Pre-operative diet recommendations o Post-op diet stages/progression (see appendix I for detail of stages used at KHS for sleeve gastrectomy) o Nutritional considerations after surgery o Complications of surgery and what to look for/what to do o Exercise o Lactose intolerance o Meal duration o Chewing thoroughly o Fluid and protein needs o Prevention of dumping syndrome o Vitamin/mineral supplementation o Portions sizes o Label reading o Dining out o Food records o Recipes o Hair loss Set goals for dietary/behavior changes post-op eat at home before going out Cook breakfast at home Plan meals ahead of time Stick with diet plan/don’t get discouraged 71 Encourage follow-up visits postoperatively for diet reinforcement and to facilitate healthy wt loss o o Monitor Weight Diet Behavioral changes as stated in goals Activity level Commitment to lifestyle changes Evaluation Patient’s expectations of post-op eating habits are realistic Anticipate ability to comply with necessary changes; good family support system 3/10/08 –2nd pre-op assessment o Assessment: Current diet General diet Attempting to wean caffeine Nutritional needs 1650 kcals (based on 16 kcals/kg body weight for weight loss) 52-65 g protein (.8-1.0 g/kg IBW) Activity level (sedentary) Anthropometrics o physical at work, keeping up with kids and 2 dogs Measurement taken from prior visit to calculate weight, BMI, WHR Diagnosis: obesity related to food and nutrition-related knowledge deficit and physical inactivity as evidenced by BMI = 39 (grade II), patient reports eating out approximately 6x/week, low-intensity physical activity o Intervention Nutrition education for sleeve gastrectomy o Principles of Good Nutrition/Food Guide Pyramid o Portion control o Meal plan = 1600 kcals 72 o Behavior modification tips o Label reading o Provided “Lifesteps” referral (a 12-week weight management program at KHS) Set goals for dietary/behavior changes Continue to wean caffeine Decrease meat portions and increase fruit and vegetable consumption Record meals 3 days prior to next visit Ride bike 20 minutes, 2 days per week Encourage follow-up visits postoperatively for diet reinforcement and to facilitate healthy weight loss o o Monitor Weight Diet Behavioral changes as stated in goals Activity level Commitment to lifestyle changes Evaluation Patient will continue to comply with dietary/behavior changes 4/2/08 –3rd pre-op assessment o Assessment: Current diet 1600 kcals Increased fruit and vegetable consumption 1 meal very high fat Activity level (moderate) physical at work, keeping up with kids and 2 dogs attempting to bike 20 minutes, 2x per week Anthropometrics Measurement taken from prior visit to calculate weight, BMI, WHR 73 o Diagnosis: obesity related to food and nutrition-related knowledge deficit and physical inactivity as evidenced by BMI = 39 (grade II), patient reports eating out approximately 6x/week, low-intensity physical activity o Intervention Nutrition education for sleeve gastrectomy o Supermarket tips o Recipe modification o Tips for dining out Reviewed progression of post-op diet Discussed pre-op preparation at home Set goals for dietary/behavior changes Continue to wean caffeine Decrease high fat foods/meals Increase fruit and vegetable consumption Continue to ride bike 20 minutes, 2 days per week Encourage follow-up visits postoperatively for diet reinforcement and to facilitate healthy wt loss o o Monitor Weight Post-op Diet Behavioral changes as stated in goals Activity level Commitment to lifestyle changes Post-op complications Evaluation Patient will continue to comply with dietary/behavior changes 5/6/08 – post-op day 1 assessment (in hospital) o Assessment: Current diet NPO Reports poor appetite but is very thirsty 74 Nutritional needs 1622 kcals (based on 15 kcals/kg body weight) 52-65 g protein (based on .8-1.0 g/kg IBW) Denies n/v/d Activity level Currently patient is encouraged to move around without strenuous activity o o No lifting o No driving Anthropometrics (Table 13) Weight – pt. reported IBW %IBW BMI Diagnosis: no nutrition problems at this time (first day post-op patient is on clear liquids with no prior history of nutritional deficiencies, patient is well-nourished). o Intervention Reviewed post-op diet requirements o Progression of diet o No straws o No caffeine o Hydration (consuming at least 32 oz of fluids) o Protein shakes Set goals for dietary/behavior changes Comply with dietary guidelines Continue exercising (as approved by physician) Encourage follow-up visits postoperatively for diet reinforcement and to facilitate healthy wt loss o Monitor Weight Tolerance to diet/progression of diet 75 o labs Activity level Commitment to lifestyle changes Post-op complications Evaluation Patient is very motivated to lose weight Plans to adhere to guidelines Plans to follow-up with dietitian Expect good compliance 5/20/08 –1st follow-up assessment (2 weeks post-op) o Assessment: Current diet Puree (Stage II); began one week post-op No appetite Feels full after eating food the size of a quarter (approximately 1 oz) Consuming protein shake daily Doesn’t tolerate mayonnaise well anymore Meeting fluid needs Taking children’s chewable MVI 2x/day 24-hour food recall: o Breakfast – 1-1.5 oz cottage cheese with fruit or shake OR 6 oz Protein shake made with 1 scoop whey protein (provides 20 g pro) and 1/3 cup skim milk powder (provides 8 g pro); takes a few hours to finish shake OR o Yogurt Lunch/Dinner 1-2 oz chicken puree with broth, 1 oz potato or spinach OR 76 o Estimated protein intake = 45 g Denies n/v/d Loose stools Back pain – pt attributes this to surgical table GERD and urinary incontinence resolved o 1-2 oz tilapia/tuna or egg Urinates 4-5x/day instead of 4-5x/hour Activity level (sedentary/moderate) Started walking yesterday Bought a jump rope Bought 2 – 5# dumbbells Discontinued all pain meds Anxious to return to work; feels good Trouble sleeping 1st and 2nd night due to drain tube (drain tube removed 3rd day) Anthropometrics (Table 13) Lost 16 pounds since surgery Weight IBW %IBW BMI Diagnosis: inadequate protein intake related to s/p gastric sleeve as evidenced by 24 hour food recall revealing approximately 45 g pro/day. o Intervention Reviewed post-op diet requirements o Stage II o Protein intake/sources of proteins Set goals for dietary/behavior changes 60 g protein/day Talk to surgeon regarding calcium supplementation Begin exercise 30 minutes, 3x/week with surgical clearance Requested 3-day food record 77 o o Encouraged pt to attend bariatric support group meeting Scheduled next appointment (June 4) Monitor Weight Tolerance to diet/progression of diet Protein and fluid intake labs Activity level Commitment to lifestyle changes Post-op complications Evaluation Patient still highly motivated Expect good compliance and continued weight loss 6/4/08 –2nd follow-up assessment (4 weeks post-op) o Assessment: Current diet Stage II Consuming protein shake daily Meeting fluid needs (water/crystal light) Supplements: o 2 children’s chewable MVI o Actigall Patient reported going out to eat for breakfast; had 3 bites of scrambled egg and 1 bite of pancake and had to stop due to satiety 24-hour food recall: o Breakfast 6 oz protein shake, ¼ cup banana (consume ½ and then finishes other ½ 30 minutes later) OR o Cottage cheese Lunch 78 ½ scrambled egg OR o 2 oz tuna/tilapia/chicken Dinner 2 oz tuna/tilapia/chicken with spinach and mashed potato o Snack o 3 oz sugar-free pudding Estimated protein intake = 50 g Patient did not provide 3-day food record Denies n/v/d Normal bowels – no more loose stools Incision sites achy and tingly (pt reports this is due to rain) Back pain improved – pt had adjustment by chiropractor Activity level (moderate) Bike riding approximately 5 days per week for 30-45 minutes Bought a jump rope Bought 2 – 5# dumbbells Returned to work May 25th Anthropometrics (Table 13) Lost 19 pounds since surgery Weight IBW %IBW BMI Diagnosis: inadequate protein intake related to s/p gastric sleeve as evidenced by 24 hour food recall revealing approximately 50 g pro/day. o Intervention Reviewed post-op diet requirements o Stage III and Stage IV o Protein intake/sources of proteins 79 o o o Fiber sources Set goals for dietary/behavior changes 60 g protein/day Talk to surgeon regarding calcium supplementation Continue exercise, 30 minutes 3x per week Continue to increase fluid intake – long-term goal of 64oz/day Encouraged patient to attend bariatric support group meeting Scheduled next appointment (August) Monitor Weight Tolerance to diet/progression of diet Protein, fluid, fiber intake labs Activity level Commitment to lifestyle changes Post-op complications Evaluation Pt still motivated Expect good compliance and continued weight loss 80 CRITICAL COMMENTS KS made an informed decision to undergo sleeve gastrectomy. She attended a presentation given by a bariatric surgeon at Kennedy Health Systems (KHS), discussing the available surgical procedures. She then discussed her prior weight, medical and personal history with the surgeon at her initial consultation. She knew she didn’t want a malabsorptive procedure because she had learned about the side effects and did not want to experience dumping or nutrient deficiencies. However, given that KS has a “sweet tooth,” roux-en-y gastric bypass would have been more appropriate (sweets can still be consumed with sleeve gastrectomy with little to no consequences other than weight gain). KS’s surgeon took all the necessary precautions to ensure that she was physically and mentally able to withstand surgery by having her consult with a cardiologist, pulmonologist, dietitian, psychiatrist and gastroenterologist. KS met with the anesthesiologist a few days prior to surgery. While she was hospitalized, she was prescribed pain medications, anti-gerd medications and an anticoagulant. The use of an anticoagulant was a good, precautionary step to help reduce the incidence of blood clots postoperatively (recall that the number one cause of death among bariatric surgical patients is pulmonary embolism). KS She was treated with respect and dignity throughout her hospital stay. KS’s first post-op visit with the surgeon occurred two weeks after surgery. I think this should have occurred one week after surgery just to make sure there were no major problems such as dehydration or hyperemisis. KS was prescribed two children’s multivitamins and actigall daily. Since rapid weight loss is associated with gallstones, I think it was an excellent precautionary measure to prescribe actigall. She was not prescribed a calcium supplement. Some surgeons prescribe this postoperatively, some do not. Given that initial intake is anywhere from 400-800 calories, I think it would have been wise to prescribe one. She is scheduled for a follow-up visit with the surgeon in August (3 month post-op visit) where she will have blood drawn to check for vitamin and mineral deficiencies. Since KS’s serum Calcium was within normal limits before surgery, perhaps the surgeon felt supplementation was not needed at this time (and would be re-evaluated when serum calcium levels are checked in August). 81 KS had several pre-op dietary consultations. She was well educated on the dietary and lifestyle changes that would be necessary postoperatively. Small, achievable goals were set to begin behavior and dietary modification. However, KS set a weight loss goal of 77 pounds, to achieve a weight of 165 pounds. KS most likely will not achieve this goal. Current research indicates short-term excess weight loss at approximately 60-70%; this would equate to a 60-70 pound weight loss. Of course there have been individuals who have achieved greater excess weight loss, but this is not the norm. KS is potentially setting herself up for failure by creating an unrealistic weight loss goal. There was poor communication between the surgeon and dietitian. A program that houses the surgeon and dietitian under the same roof would be beneficial to the patient. The dietitian thought KS should be taking a daily calcium supplement postoperatively. The dietitian instructed KS at the first post-op visit to mention this to the surgeon. At the second post-op visit, KS was still not taking a calcium supplement. I am unsure as to whether or not this was mentioned to the surgeon by KS. The dietitian again instructed KS to mention a calcium supplement to the surgeon. I believe it would have been best for the dietitian to telephone the surgeon and discuss supplementation. Correspondence consisted of faxing nutrition assessment forms to the surgeon’s office. Dietitians and surgeons should be working closely together with bariatric patients. Although the dietitian working with KS has a basic understanding of bariatric surgery and diet progression, she is not specialized in the field of bariatrics. Patients deserve dietitians who are specialized and have experience working with this population. The dietitian meeting with patients should be present at support group meetings to keep continuity of care. Currently at KHS, the dietitian attending the support group meetings is not the same as the dietitian assessing bariatric patients. There are only a few things I would have done differently: weigh KS at all pre-op nutrition consultations, set a more achievable weight loss goal, improve communication with the surgeon, and attend bariatric support group meetings. I am not sure if the dietitian will request a copy of the lab results in August – this is something I would request; I would also ask for a lipid panel to see if KS’s cholesterol level has improved as a result of the surgery. The dietitian assessing KS had an excellent rapport with her. She was compassionate, understanding and supportive. 82 SUMMARY Obesity has reached epidemic proportions over the last twenty years. The causes are not completely understood but contributing factors include the environment, society, genetics, biology, and psychology. Behavior, dietary, exercise and pharmacotherapies have proven ineffective for maintaining weight loss. Bariatric surgery is the only proven method; the restrictive procedure, sleeve gastrectomy, shows promising results for reducing excess weight and co-morbid conditions but long-term studies are needed to prove its efficacy. 83 MEDICAL BIBLIOGRAPHY GENERIC NAME BRAND NAME DRUG USE FOOD/DRUG INTERACTION SIDE EFFECTS ursidiol Actigall to prevent or dissolve gallstones take with food nausea, abdominal pain, diarrhea cefazolin Ancef/Kefzol antibiotic take 2 hrs before or after antacid, magnesium supplement, iron supplement or MVI with iron (otherwise absorption of drug will be decreased) diarrhea famotidine Pepcid anti-gerd: increases gastric pH, decreases acid secretions take drug at least one hour after iron supplement; decreases iron and vit B12 absorption; magnesium or aluminum/magnesium antacids decrease drug’s absorption nausea, vomiting, diarrhea, contsipation heparin Heparin sodium anticoagulant none noted bleeding, hemorrhage, dizziness, headache, abdominal pain, GI bleeding, constipation hydromorphine Dilaudid analgesic insure adequate fluid intake/hydration anorexia, decreased gastric motility, nausea, vomiting, constipation ketorolac *Toradol anitinflammatory: used to treat pain after surgery none noted long-term use can damage kidneys or cause bleeding metoclopramide Reglan anti-gerd avoid alcohol - increases effects of alcohol; may alter insulin requirements (caution with diabetes) nausea, diarrhea, restlessness, drowsiness, fatigue, dizziness Source: Food Medication Interactions, 13th Edition (70). *source: http://www.drugs.com/toradol.html 84 APPENDIX A – Appetite Regulatory Peptides HORMONE/PEPTIDE/MESSENGER RESEARCH FINDINGS APPETITE EFFECT Agouti-related protein (AgRP) Overexpression of central AgRP results in obesity; intracerebroventricular administration of AgRP increases feeding with long-lsating effects while inhibiting the action of alpha-MSH increase Alpha-melanocyte stimulating hormone (alpha-MSH) Alpha-MSH is an important melanocortin that inhibits food intake via opioid pathways. decrease AMP-activated protein kinase (AMPK) Pharmacological activation of AMPK in the hypothalamus is shown to increase food intake. The molecule is a component of a protein kinase cascade that acts as an intracellular energy sensor maintaining the energy balance within the cell. increase Amylin Amylin (amyloid polypeptide) reduces food intake and the amylin agonist pramlintide has been shown to reduce body weight in teyp-1 and type-2 diabetics. decrease Bombesin intraperitoneal injections of bombesin are shown to elicit behavioral satiety under condition in which gastric, intestinal and postabsorptive mechanisms are minimally activated by ingested food. decrease Brain derived neurotrophic factor (BDNF) BDNF induces severe, dose-dependent appetite suppression and weight loss through central mechanisms. decrease Cholecystokinin (CCK) CCK is raapdily released from the gastrointestinal tract postprandially and stimulates pancreatic secretion, gut motility and gall bladder contraction; it also inhibits food intake via the brainstem in humans and rodents. decrease Cocaine- and amphetamine-related transcript (CART) CART is an hypothalamic endocrine signaling protein. When injected intracerebroventricularly into rats, recombinant CART inhibits both normal and starvation-induced feeding, and completely blocks the feeding response induced by NPY. decrease Corticotrophin-releasing hormone (CRH) CRH inhibits feeding and increases metabolic rate when injected into the brains of animals; it has the opposite effect to NPY. CRH ameliorate obesity through stimulation of sympathetic nerve-mediated mechanisms and inhibition of vagus nerve-mediated mechanisms. decrease Dopamine The neurotransmitter dopamine modulates motivation and reward circuits. Availability of dopamine D2 receptors is found to be decreased in the brains of obese patients, suggesting dopamine deficiency may cause pathological eating to compensate for decreased activation of these circuits. increase/decrease Dynorphin Dynorphin is shown to produce highly specific increase in food ingestion. increase Endocannabinoids (CB-1) Endocannabinoids are involved in appetite, eating behavior and body weight regulation: CB-1 cannabinoid receptor stimulate appetite and ingestive behaviors. increase 85 Enterostatin Enterostatin, the pancreatic polypeptide, has been shown to chronically reduce fat intake and boy weight, suggesting that it may attenuate the appetite for fat. decrease Gamma-aminobutyric acid (GABA) Studies show that GABA stimulation of hypothalamic GABAA receptors increases food intake and body weight. Decreasing neuronal GABAergic tone is shown to inhibit feeding. increase/decrease Galanin The brain peptide galanin is shown to preferentially increase carbohydrate/fatrich nutrient intake in animals. increase Ghrelin Grhelin is synthesized in the stomach and expressed in brainstem and hypothalamic nuclei. Ghrelin causes hyperphagia in rodents; and plasma levels are shown to be increased during juman starvation and fall after eating, indicating its orexigneic potential. increase Glucagon-like peptide-1 (GLP-1) GLP-1 is synthesized via the proglucagon gene in the gastrointestinal tract and brain where the active peptide GLP-1 amide is released into the circulation after food ingestion. GLP-1 inhibits food intake in healthy individuals, diabetics and non-diabetic obese men. decrease Histadine/Histamine hypothalamic histadine suppresses food intake through its conversion to histamine; dietary histamine has also been shown to decrease food intake and body fat via activation of histamine neurones. decrease Huntingtin-associated protein-! (Hap1) Hap 1 is highly expressed in the hypothalamus and increases activity at GABAA receptors; mice lacking Hap1 are found to be hypophagic. increase/decrease Insulin Insulin promotes the conversion of glucose to fat and storage of fat in adipose tissue. Hypothalamic injections of insulin inhibit feeding, stimulate brown adipose tissue (BAT) thermogenesis, and causes weight loss. Insulin is believed to affect food intake by reducing NPY expression in the hypothalamus. decrease Leptin Leptin is secreted by adipocytes and is the gene product of the OB gene. Leptin deficiency and leptin resistance lead to sever obesity in mice, suggesting that it might be crucial to the normal control of food intake and body weight. However, only a few cases of congenital leptin deficiency associated with severe earaly onset obesity have been documented. Paradoxially, most obese patients present with hyperleptinaemia, but this has been interpreted as evidence of leptin resistance, suggesting reduced sensitivity to leptin’s physiological effects. increase Mammalian target of rapamycin (MTOR) MTOR, a serine-threonine kinase, regulates cell-cycle progression and growth by sensing changes in energy status. Research suggests mTOR signaling plays a role in the brain mechanisms that respond to nutrient availability, regulating energy balance. Central administration of leucine increases hypothalamic mTOR signaling and decreases food intake and body weight. decrease Melanin-concentrating hormone (MCH) MCH found in the hypothalamus increases food intake in a dose-dependent manner and lowers plasma glucocorticoid levels through a mechanism involving adrenocorticotrophic hormone. increase Melanocortin-$ receptor (MC4R) The key signaling protein melanocortin-4 receptor (MC4R) is found to be decrease 86 important in modulating obesity: mutations in the MC4R genes are shown to be a frequent and heterogenous cause of morbid obesity. Nesfatin-1 Nesfatin, corresponding to NEFA/nucleobindin 2 (NUCB2), a secreted protein, is expressed in the appetite-control hypothalamic nuclei in rates. iNtracerebroventricular injections of NUCB2 are shown to reduce feeding. decrease Neuromedin B, C, U and S Neuromedin peptides are known to inhibit feeding. Neuromedin C microinjected into the amygdale has been shown to decrease feeding. decrease Neuropeptide Y (NPY) NPY is highly concentrated in the hypothalamus and induces feeding through receptor subtype interaction that binds NPY. Paraventricular injections of NPY reduce energy expenditure by inhibiting the sympathetic nerves that innervate and stimulate BAT, casuing hyperphagia. increase Neurotensin Intracerebroventricular administration of neurotensin produces a dose-related decreased in food intake. decrease Noradrenaline (NA) Pharmacologic manipulations that elevate noradrenaline can increase or decrease food intake, depending on the site and type of noradrenaline manipulation. increase/decrease Oleoylethanolamide (OEA) OEA, and endogenous lipid produced primarily in the small intestine, indirectly mediates the effects of appetite suppression, reduces blood lipid levels, and enhances peripheral fatty acid catabolism. OEA also regulates body weight by altered peripheral lipid metabolism, including increased lipolysis in adipocytes and enhanced fatty acid uptake in enterocytes. decrease Orexin A/Orexin B Central administration of orexins A and B stimulate food intake and upregulation of prepro-orexin mRNA levels on fasting. increase Oxyntomodulin (OXM) OXM is released from the gut postprandially in proportion to energy intake, and circulation levels of OXM are increased in several conditions associated with anorexia. Infusion of OXM is shown to significantly reduce ad libitum energy intake at a buffet meal and causes a significant reduction in scores for hunger. decrease Oxytocin (OT) Intracerebroventricular administration of OT and an OT agonist is shown to significantly decrease food intake in a dose-related manner. decrease Pancreatic polypeptide (PP) Intravenous infusions of PP restore normal serum PP levels, and a regimen of morning and afternoon PP infusions is found to significantly reduce food intake in Prader-Willi subjects who are characteristically hyperphagic and obese. decrease Peptide YY (PYY3-36) PYY, secreted by the L-cells of the gastrointestinal tract, is released proportionate to calories ingested. Administration of the active form, PYY3-36, causes marked inhibition of food intake in rodents and man. decrease (cont’d on next page) POMC is the precursor for melancortins (e.g. alpha-MSH) which cleave other 87 Proopiomelanocortin (POMC) proteins in the appetite regulation pathway. Homozygous mutations in the POMC gene cause early onset obesity, adrenal insufficiency and red hair pigmentation in humans, whereas heterozygous mutations cause subtle defects contributing to inherited obesity. decrease Protein tyrosine phosphatase-1B (PTP-1B) PTP-1B plays a role in the pathogenesis of insulin resistance in obesity and type 2 diabetes mellitus. PTP-1B activity is found to be significantly reduced in obese and diabetic individuals compared with controls. increase/decrease Satietin Satietin is blood-borne anorectic glycoprotein that is found to have a longlasting satiatory effect. decrease Serotonin (5-HT) The neurotransmitter serotonin is a well-established anorectic and its hypophagic effect requires activation of 5-HT1A and 5-HT1B receptors. decrease Triiodothyronine (T3) Peripheral and hypothalamic triidothyronine directly stimulates feeding: peripheral administration of T3 has been found to double food intake in ad libitum-fed rats over 2h, whereas injection of T3 directly into the hypothalamus is shown to produce a fourfold increase in food intake in the first hour. incresae Uncoupling proteins (UCPs/thermogenin) Mitochondrial membrane proteins (e.g. UCP-1, 2 and 3) usually found on brown adipocytes play a role in energy balance and weight gain. Transgenic ablation of adipocytes that express UCP1 and UCP2 results in obesity in mice suggesting the UCPs in BAT metabolism are important in energy balance and obesity. increase Source: reprinted with permission from T.J. Atkins (50) 88 APPENDIX B 89 APPENDIX C 90 APPENDIX D 91 APPENDIX E 92 APPENDIX e 93 APPENDIX E 94 APPENDIX E 95 APPENDIX E 96 APPENDIX F 97 APPENDIX F 98 APPENDIX F 99 APPENDIX F 100 APPENDIX F 101 APPENDIX F 102 APPENDIX G 103 APPENDIX H– Surgical Report of Patient FINDINGS: Dense adhesions between the posterior aspect of the stomach and the anterior surface of the pancreas. ESTIMATED BLOOD LOSS: 20 mL. BRIEF HISTORY OF PRESENT ILLNESS AND INDICATION FOR PROCEDURE: The patient is a 38-year-old female with morbid obesity. Medical complications related to her morbid obesity refractory to nonoperative weight loss attempts now for definitive surgical procedure. Discussed with the patient at length the risks, benefits, possible alternatives, possible complications of the procedure including the possibility of leak, blood clots, intractable bleeding. Consent obtained. PROCEDURE: The patient was brought to the operating room theater, prepped and draped in the normal sterile fashion after the success of general anesthesia. Foley and OG tube were placed. Suitable sites for a skin incision were selected in the right upper quadrant. A 5-mm skin incision was made with the #15 scalpel blade with dissection through soft tissue with the bladeless trocar. The abdomen was entered without difficulty. The abdomen was insufflated to 15 mmHg. The abdominal contents were inspected with no damage to the underlying structures. A 12-mm port was placed in the supraumbilical position, a 5-mm port was placed in the epigastrium, two 12-mm ports were placed in the left hemiabdomen, a 5-mm port placed in the left anterior axillary line, a 15-mm port placed in the right hemiabdomen. The stomach was fully decompressed with the G-tube and the G-tube was removed. The gastric sac was entered by dividing the omentum along the greater curvature of the stomach with the ligature device. This was carried up to the angle of His and then down along the greater curvature of the stomach to a point approximately 6 cm from the pylorus along the antrum. The stomach was then elevated up. Dense adhesions were encountered between the posterior aspect of the stomach and the pancreas suggesting a prior inflammatory process. These were taken down with the use of blunt and sharp dissection with the use of the ligature device. At this point due to the dense posterior adhesions the decision was made to only proceed with the gastric restricted portion of the procedure. A vertical gastrectomy was then performed. Starting approximately 6 cm from the pylorus two 60-mm green cartridges were fired along a 40-French bougie and then the remaining fires were 60-mm blue cartridges preceding in the angle of His. The gastrectomy specimen was removed through the 15-mm port site. The anastomosis was tested, submerged under saline solution with intraoperative EGD and there was no evidence for a leak. The staple line was covered with Tisseel. A #19 Blake drain was positioned through the 5-mm port site in the left hemiabdomen adjacent to the staple line. The liver retractor in the epigastrium was then removed. The 12 and 15-mm port sites were closed with 0 Vicryl suture and an Endoclose device. The abdomen was desufflated. The skin incisions were irrigated and closed with staples. The patient tolerated the procedure well, was extubated and brought to the recovery room in stable condition 104 APPENDIX I – Kennedy Health Systems Diet Progression for Sleeve Gastrectomy STAGE I – CLEAR LIQUIDS Clear liquids generally begin after an upper GI “swallow test” is performed. Your surgeon will order the diet, usually the day after surgery, and it will continue for the next few days. In the hospital you will get diluted juice (2oz juice & 2oz water), broth, and diet gelatin on each tray Try to sip fluids at the rate of 1-2oz per hour. A “medicine” cup or shot glass may assist in measuring this portion size Goal: consume 32oz (4 cups) fluid per day in order to prevent dehydration Sipping fluids out of bottles or straws may cause gas, which may lead to indigestion, bloating and abdominal discomfort, so avoid using them. Food to Choose Guide to Clear Liquid Choices Foods to Avoid Diluted fruit juice: Fruit nectars or juices with pulp Apple Cranberry Grape Water milk /milkshakes Crystal light or other sugar-free drink mixes cream soups Sugar-free popsicles sweetened drink mixes Diet gelatin cocoa Broth *Carbonated beverages Sugar substitutes Soda Sparkling water seltzers alcohol Splenda Equal Sweet-n-low coffee tea *carbonation may cause gas, bloating, and stretch the pouch 105 STAGE II – SMOOTH CONSISTENCY The next step of the diet progresses to puree foods, which are low in fat and contain no sugar. Foods should be pureed to a smooth consistency without any lumps. During this stage, only a few tablespoons will be consumed at meals and snacks. This will last approximately 2-3 weeks. Foods include: skim milk, pureed poultry and meat, mashed potatoes and unsweetened applesauce, unsweetened yogurt, sugar-free puddings, cream of rice, cream of wheat May utilize protein powder or shakes Begin vitamin/mineral supplementation (1 month after surgery) Eat every 3-4 hours Sip liquids all day long, and wait 30 minutes after a meal Prepare food ahead of time and freeze in ice-cube trays, which can later be reheated Each meal should initially be ½ cup (8 Tbsp) and progress gradually to ¾ cup A healthy, well balanced diet should include food sources from the following groups: o Protein: 6-8 servings per day o Vegetable: 2 servings per day o Bread/Grains (starches): 2 servings per day o Fats: 3 servings per day Stage II Sample Menu Breakfast: ¼ cup pureed cottage cheese with 1/8 cup pureed peaches Lunch: 2 ounces (about ¼ cup) pureed tuna fish and 1/3 cup puree vegetable Snack: ¼ cup sugar-free pudding (suggestion: add protein powder) Dinner: ¼ cup pureed chicken with ¼ cup smooth mashed potatoes and 1/3 cup pureed vegetable Snack: ½ cup skim milk or applesauce with protein powder 106 STAGE III – MINIMAL TEXTURE After 2-3 weeks following surgery, progress to soft, cooked foods as tolerated. Initially a “cottage cheese” consistency, still pureed but not as smooth Next, increase to finely ground meats, soft fruits, cooked vegetables as tolerated and unsweetened cereals Finally, advance to diced poultry (without skin) Still follow low-fat and low-sugar choices Continue to sip fluids all day long Begin vitamin and mineral supplements (1 month after surgery) Each meal should be ¾ cup to 1 cup in size This stage lasts approximately 2-3 weeks. Check with your physician. Stage III Sample Menu Breakfast: 1 egg poached or scrambled, and ¼-1/2 cup oatmeal with protein powder. Take multivitamin Fluids: 1c decaffeinated coffee or sugar-free lemonade, sip slowly; 2oz every 15 minutes Snack: 1-2 ounces (slices) low-fat cheese (example: Alpine Lace Cheese) Fluids: 1c skim milk or water, sip slowly; 2oz every 15 minutes Lunch: 1-2 ounces turkey, 1/8-1/4 cup cooked carrots, and 1/8 cup fruit Fluids: 1 cup skim milk or water, sip slowly; 2oz every 15 minutes Snack: 1 cup protein supplement or ½ cup sugar-free instant pudding with protein powder Dinner: ¼ cup ground beef, ¼ cup mashed potatoes, and ¼ cup cooked broccoli Snack: ¼ cup cottage cheese and ¼ cup diced peaches (puree if needed) Fluids: 1 cup diluted juice, sip slowly; 2 oz every 15 minutes; take calcium supplement 107 STAGE IV – SOLIDS & MAINTENANCE Gradually advance to solids, usually about six to eight weeks after surgery. This step is now considered the maintenance level which will be followed for the rest of your life! It consists of high protein, low-fat, low-sugar foods. Slightly larger amounts of food are now able to be tolerated, but remember not watching portion sizes can lead to weight gain as the pouch will stretch. Gradually add whole grains, limit fat and sugar Pay attention to volume Remember to eat high protein foods first, focus on 3-4 ounces of protein per meal Drink constantly just before or after meals Strive for a high fiber diet by including non-starchy high fiber carbohydrates such as oatmeal or cracker type foods Gradually add fresh fruit and vegetables In general, each meal should be about 1-1/2 cups in size Eat slowly, taking 20-30 minutes for a meal Stage IV Sample Menu Breakfast o 1 egg o 1 slice wheat bread o 1 tsp butter Snack o 2-3 ounces low-fat cottage cheese o 3 strawberries Lunch o 3 ounces of turkey o 1 slice wheat bread o 1 tsp light mayonnaise o 1 slice tomato Dinner o 3 ounces of chicken o ¼ cup broccoli o ¼ cup potato Snack o 1 Tbsp peanut butter (creamy) o 3 crackers 108 109 110 REFERENCES 1. Center for Disease Control. Obesity among adults in the United States—no statistically significant change since 2003-2004.Center for Disease Control. Available at: http://www.cdc.gov/nchs/pressroom/07newsreleases/obesity.htm. Published November 2007. Accessed May 25, 2008. 2. World Health Organization. Fact sheet No. 311. Obesity and overweight. World Health Organization. Available at: http://www.who.int/mediacentre/factsheets/fs311/en/print.html. Published September 2006. Accessed May 25, 2008. 3. Pinna K, Rolfes SR, Whitney E. Understanding Normal and Clinical Nutrition, 7th edition. US: Thomson Wadsworth: 2006. 4. Burke, LE, Cartwritght MA. Obesity: an overview of assessment and treatment. In: Bridges, EJ, Froelicher ES, Motzer SA, Woods, S, ed. Cardiac Nursing, 5th Edition. Lippincott Williams & Wilkins; 2005:42. 5. Catenacci VA, Hill JO, Peters JC, Wyatt HR. International strategies to address obesity. Obesity Reviews. 2008;9:441-47. Accessed June 1, 2008. 6. US Department of Health and Human Services. The surgeon general’s call to action to prevent and decrease overweight and obesity 2001. Center for Disease Control. Available at: http://www.cdc.gov/nccdphp/dnpa/pdf/CalltoAction.pdf. Published 2001. Accessed May 25, 2008. 7. Mattar, SG. Lifting the unbearable weight of morbid obesity. Annals of Surgery. 2008;247:28-29. doi:10.1097/SLA.Ob013e318160c1fc. 8. Jones DB, Powers KA, Rehrig ST. Financial impact of obesity and bariatrics Surgery. The Medical Clinics of North America. 2007;91:321-338. doi:10.1016/j.mcna.2007.01.001. 9. Mathier MA, Ramanathan RC. Impact of obesity and Bariatric Surgery on Cardiovascular Disease. The Medical Clinics of North America. 2007;91:415-431. doi:10.1016/j.mcna.2007.02.002. 10. Buchwald, H. Consensus conference statement bariatric surgery for morbid obesity: health implications for patients, health professionals, and third-party payers. Surgery for Obesity and Related Diseases. 2005;1:371-381. 11. Blanc PM, Bomans B, de Seguin de Hons C, Fabre JM, Gagner M, Krawczykowsky D, Millat B, Monnier L, Nocca D, Picot MC. A prospective multicenter study of 163 sleeve gastrectomies: results at 1 and 2 years. Obesity Surgery. 2008;18:560-565. doi:10.1007/s11695-007-9288-7. 12. Dietel, M. A synopsis of the development of bariatric operations. Obesity Surgery. 2007;17:707710. 13. Davidson BK, Kurian MS, Thompson B. Weight Loss Surgery for Dummies. Indianapolis, IN: Wiley Publishing Inc.: 2005. 14. Rosenthal R, Szomstein S, Tucker O. Nutritional consequences of weight-loss surgery. The Medical Clinics of North America. 2007;19:499-514. doi:10.1016/j.mcna.2007.01.006. 15. Arar N, Beglaibter H, Givon-Madhala O, Lustigman H, Spector R, Stein M. Technical aspects of laparoscopic sleeve gastrectomy in 25 morbidly obese patients. Obesity Surgery. 2007;17:722727. 111 16. Kohan G, Oria As, P LTC, Quesada BM, Roff HR. Laparoscopic sleeve gastrectomy as an alternative to gastric bypass in patients with multiple intraabdominal adhesions. Obesity Surgery. 2008;18:566-568. doi:10.1007/s11695-007-9302-0. 17. Brownell Kd, Phelan S, Wadden TA. Kaplan & Sadock’s Comprehensive Textbook of Psychiatry, 8th Edition. Lippincott Williams & Wilkins; 2005:24.3. 18. Escott-Stump S, Mahan KL. Krause’s Food, Nutrition, & Diet Therapy, 11th edition. Philadelphia, PA: Saunders: 2004. 19. National Heart, Lung and Blood Institute (NHLB). NIH Publication No. 00-4084. The practical guide: identification, evaluation, and treatment of overweight and obesity in adults. Available at: http://www.nhlbi.nih.gov/guidelines/obesity/prctgd_c.pdf. Published October 2000. Accessed May 25, 2008. 20. National Institute of Diabetes and Digestive Kidney Disease. Statistics related to overweight and obesity. Weight-control Information Network. Available at: http://win.niddk.nih.gov/statistics/index.ht. Updated June 2007. Accessed May 25, 2008. 21. National Institute of Health. Publication No. 01-3680. Understanding adult obesity. Weightcontrol Information Network. Available at: http://win.niddk.nih.giv/understanding.htm. Published October 200l. Updated March 2006. Accessed May 25, 2008. 22. Obesity and Eating Disorders. In: Dudek S, ed. Nutrition Essentials for Nursing Practice, 5th Edition. Lippincott Williams & Wilkins;2006:14. 23. Escott-Stump S. Nutrition and Diagnosis-Related Care, 5ht edition. Baltmiore, MD: Lippincott Williams & Wilkins: 2002. 24. Janssen I, Mark AE. Elevated body mass index and mortality risk in the eldery. Obesity Reviews. 2007;8:41-59. doi:10.1111/j.1467-799X.2006.00248.x. 25. Center for Disease Control. Obesity by the Numbers. Obesity in America. Available at: http://www.obesityinamerica.org/trends.html. Accessed: June 5, 2008. 26. Bellisar A. Evolutionary origins of obesity. Obesity Reviews. 2008;9:165-180. doi:10.1111/j.1467789X.2007.00392.x. 27. Stunkard A, Wadden T. Handbook of Obesity Treatment. New York, NY: Guilford Press: 2002 28. Drent ML, Jakobsdottir S, Klok MD. The role of leptin and ghrelin in the regulation of food intake and body weight in humans: a review. Obesity Reviews. 2007;8:21-34. doi:10.1111/j.1467789X.2006.00270.x 29. Bray GA Popkin BM, Wolf A. A short history of beverages and how our body treats them. Obesity Reviews. 2008:9:151-164. doi:10.1111/j.1467-789x.2007.00389.x. 30. Approach to the patient with obesity. In: Alpers D, Kaplowitz N, Laine L, Owyang C, Powell D, ed. Textbook of Gastroenterology, 4th Edition;2003:36. 31. Astrup A, Dyerberg J, Selleck M, Stender S. Nutrition transition and its relationship to the development of obesity and related chronic diseases. Obesity Reviews. 2008;9:48-52. 32. RD411. The sleep /weight connection. RD411. Available at: http://www.rd411.com/article.php?ID=227. Accessed June 5, 2008. 33. Nutrition Care Manual. American Dietetic Association. Available at: http://www.nutritioncaremanual.org. Accessed June 25, 2008. 112 34. Office of Genetics and Disease Prevention Public Health Perspectives. Obesity and Genetics: what we know, what we don’t know and what it means. Center for Disease Control. Available at: http://www.cdc.gove/genomics/training/file/print/perspectives/obeseknow.pdf. Accessed June 5, 2008. 35. Bouchard C, Loos RJF. FTO: the first gene contributing to common forms of human obesity. Obesity Reviews. 2008;9:246-250. doi:10.1111/j.1467-789X.2008.00481.x. 36. World Health Organization. Fact Sheet No. 311. Obesity and overweight. World health Organization. Available at: http://www.who.int/mediacentre/factsheets/fs311/en/index.html. Published: September, 2006. Accessed May 25, 2008. 37. DeMaria EJ. Greenfield’s Surgery: Scientific Principles and Practice, 4th Edition. Lippincott Williams & Wilkins; 2006:47. 38. National Heart, Lung and Blood Institute (NHLB). NIH Publication No. 00-4084. The practical guide: identification, evaluation, and treatment of overweight and obesity in adults. Available at: http://www.nhlbi.nih.gov/guidelines/obesity/prctgd_c.pdf. Published October 2000. Accessed May 25, 2008. 39. National Institute of Health. Publication No. 07-4098. Do you know the health risks of being overweight? Weight-control Information Network. Available at: http://win.niddk.nih.gov/publications/health_risks.htm. Published November 2004. Updated October 2007. Accessed May 25, 2008 40. Approach to the patient with obesity. In: Alpers D, Kaplowitz N, Laine L, Owyang C, Powell D, ed. Textbook of Gastroenterology, 4th Edition;2003:36. 41. The Obesity Society. Obesity Statistics: Obesity Trends. The Obesity Society. Available at: http://www.obesity.org/stastistics/obesity trends.asp. Accessed June 1, 2008. 42. Davis G, Gagne DJ, Patel JA. Pulmonary consideration in Obesity and the bariatric surgical patient. The Medical Clinics of North America. 2007;91:433-442. doi:10.1016/j.mcna.2007.02.001. 43. The Obesity Society, Fact Sheets. Obesity and Cancer. The Obesity Society. Available at: http://www.obesity.org/information/cancer_obesity.asp. Accessed June 1, 2008. 44. The Surgeon General. Overweight and obesity: health consequences. United States Department of Health and Human Services. Available at: http://www.surgeongeneral.gov/topics/obesity/calltoaction/fact_consequences.htm. Updated January 1, 2007. Accessed May 25, 2008. 45. The Obesity Society, Fact Sheets. Obesity and Diabetes. The Obesity Society. Available at: http://www.obesity.org/info/diabetes_obesity.asp. Accessed June 1, 2008. 46. American Society for Metabolic & Bariatric Surgery. Metabolic surgery expected to play bigger role in treating type 2 diabetes and other metabolic disease. American Society for Metabolic & Bariatric Surgery. Available at: http://www.asbs.org/Newsite07/resources/press_release_8202007.pdf Published August 22, 2007. Accessed May 25, 2008. 113 47. The Obesity Society. Obesity, bias, and stigmatization. The Obesity Society. Available at: http://www.obesity.org/information/weight_bias.asp. Accessed June 5, 2008 48. National Institute of Health Publication No.07-4191. Prescription medications for the treatment of obesity. Weight-control Information Network. Available at: http://win.niddk.nih.gov/publications/prescription.htm. Published November 2004. Updated December 2007. Accessed May 25, 2008. 49. Atkins TJ. Central and peripheral neuroendocrine peptides and signaling in appetite regulation: considerations for obesity pharmacotherapy. Obesity Reviews. 2008;9:108-120. doi:10.1111/j.1467-789X.2007.00412.x 50. Elgamal MH, McLeod MK, Saber AA. Bariatric surgery: the past, present, and future. Obesity Surgery. 2008;18:121-128. Doi:10.1007/s11695-007-9308-7. 51. Folli F, Pontiroli AE, Schwesinger WH. Metabolic aspects of bariatric surgery. The Medical Clinics of North America. 2007;91:393-414. Doi:10.1016/j.mcna.2007.01.005. 52. National Institute of Health. Publication No. 08-4006. Bariatric surgery for severe Obesity. Weight-control Information Network. Available at: win.niddk.nih.gov/publications/gastric.htm. Accessed June 5, 2008. 53. Bloomberg RD, Fleishman A, Herron DM, Kini S, Nalle JE. Nutritional deficiencies following bariatric surgery: what have we learned? Obesity Surgery. 2005;15:145-154. 54. Marian M, Russell M, Shikora SA. Clinical Nutrition for Surgical Patients. Sadbury, Ma: Jones and Bartlett Publishers; 2008:6. 55. Laparoscopic Associates of San Francisco. Surgery Comparison Chart. Laparoscopic Associates of San Francisco. Available at: http://www.lapsf.com/weight-loss-surgeries.html. 56. Crosby RD, Deitel M, Gagner M. The first international consensus summit for sleeve gastrectomy (SG), New York City, October 25-27, 2007. Obesity Surgery. 2008;18:487-496. Doi:10.1007/s11695-008-9471-5. 57. Askoxylakis J, Daskalakis M, Karkavitsas N, Koukouraki S, Melissas J, Perisinakis K, Stathaki M. Sleeve gastrectomy – a restrictive procedure? Obesity Surgery. 2007;17:57-62. 58. Frangou C. Respect for sleeve gastrectomy grows among surgeons. Gastroenterology & Endoscopy News. 2007;58:12. 59. ObesityHelp.com. Vertical sleeve gastrectomy (VSG) – also known as sleeve gastrectomy, vertical gastrectomy. ObesityHelp.com. Available at: http://www.obesityhelp.com/forums/vsg/mode,pcontent/cmsID,8874/. 60. ObesityHelp.com. About Obesity. ObesityHelp.com. Available at: http://www.obesityhelp.com/content/aboutobese.html/mode.pcontent/cmsID,2409/aboutobe se. Published 2008. Accessed June 6, 2008. 61. Cho M, Kaidar-Person O, Pinto D, Roa PE, Rosenthal RJ, Szomstein S. Laparoscopic sleeve gastrectomy as treatment for morbid obesity: technique and short-term outcome. Obesity Surgery. 2006;16:1323-1326. 62. Laparoscopic Associates of San Francisco. Vertical sleeve gastrectomy (vsg) – frequently asked questions. Obesity Help.com. Available at: http://www.obesityhelp.com/forums/vsg/mode,pcontent/cmsID,8457/ 114 63. Ashar B, Corrado J, Fuller J, Scott W. Laparoscopic trocar injuries: a report from a U.S. Food and Drug Administration (FDA) Center for Devices and Radiological Health (CDRH) Systematic Technology Assessment of Medical Products (STAMP) Committee. U.S. Food and Drug Administration (FDA) Center for Devices and Radiological Health (CDRH) Systematic Technology Assessment of Medical Products (STAMP) Committee. Published: November 7, 2003. Updated: June 24, 2005. Available at: http://www.fda.gov/cdrh/medicaldevicesafety/stamp/trocar.html. Accessed June 13, 2008. 64. Medicine Net. Definition of bougie. Medicine net. Available at: http://www.medterms.com/script/main/art.asp?articlekey=12497. Accessed June 13, 2008. 65. Kosmix RightHealth. Trocars. RightHealth. Available at: http://www.righthealth.com/Health/trocars/-od-definition_wiki_Trocar-s. Accessed June 13, 2008. 66. Dapri G, Cadiere GB, Himpens J, Vax C. A prospective randomized study comparing two different techniques for laparoscopic sleeve gastrectomy. Obesity Surgery. 2007;17:1435-1441. 67. Formato A, Forestieri P, Monda A, Pilone V, Romano A, Tramontano S. Rhabdomyolysis after sleeve gastrectomy: Increase in muscle enzymes does not predict fatal outcome. Obesity Surgery. 2008;18:349-351. doi:10.1007/s11695-007-9356-z. 68. Saltxman E, Shikora S, Tarnoff M. Managing obesity – options for obese and morbidly obese patients. The Journal of Family Practice. Supplement May 2007. Available at: www.jfponline.com. Accessed May 25, 2008. 69. American Society for Metabolic & Bariatric Surgery. Position statement on sleeve gastrectomy as a bariatric procedure. American Society for Metabolic & Bariatric Surgery. Available at: http://www.asbs.org/Newsite07/resources/sleeve_statement.pdf. Published June 2007. Accessed May 25, 2008. 70. Laparoscopic Associates of San Francisco. About vertical gastrectomy. Laparoscopic Associates of San Francisco. Available at: http://www.lapsf.com/vertical-gastrectomy-weight-losssurgery.php. Accessed June 1, 2008 71. Pronsky ZM. Food Medication Interactions, 13th Edition. Birchrunville, PA:Food-Medications Interactions:2004. 115