* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Document
Survey
Document related concepts
Transcript
Cancer, Infection &
Palliative Medicine
Dr Tim Collyns
Consultant Microbiologist
LTHT
Overview
“Febrile neutropenia”
Site specific infections
In era of increasing antimicrobial resistance
Urinary
tract
Respiratory tract
Skin / soft tissue
Clostridium difficile
Fungal
Overview
Presenter ignorance,…sucking eggs
Hospice: “between” hospital & community
Risk of “healthcare associated infections”
Meticillin
resistant Staphylococcus aureus (MRSA)
Clostridium difficile
{Meticillin
sensitive S aureus (MSSA)}
{Escherichia coli}
Multi-resistant Gram-negative bacilli
Extended spectrum β-lactamase (ESBL) producers
Plasmid: other antimicrobial classes.
Antimicrobial stewardship:
1o compared to 2o care
1o care:
Judicious
w.r.t starting antibiotics
“Viral” / (non-infectious) aetiology
“Simple”
(if likely to be effective), short courses
Resisting patient pressure for “positive” action
2o care
“Start
Smart – then Focus” (DH ARHAI, 2011)
..Right drug, right dose, right time, right
duration…every patient.
Start smart
Don’t start antibiotics in absence of clinical
evidence of bacterial infection.
If evidence: use local guidelines to initiate
prompt effective antibiotic therapy.
Document on drug chart & in medical notes:
clinical indication, duration or review date,
route & dose
Obtain cultures first
Then Focus:
Review clinical diagnosis & continuing need
for antibiotics by 48 hrs & make a clear plan
of action – the “Antimicrobial Prescribing
Decision” [APD]
The 5 APD options are
Stop
Switch
iv to oral
Change
Continue
Outpatient Parenteral Antibiotic Therapy (OPAT)
Clearly document the review & subsequent
APD in medical notes.
Hospice / palliative medicine setting
“Specific” challenges, include
Preceding
Acquisition of multi-resistant pathogens
Debilitated
of intravenous option
(Efficacy of enteral vs parenteral route)
Limited choice (if any) - MR GNBs.
Clinical
infection as the “terminal” event
Infection
/ immunocompromised
More prone to clinical infection post acquisition
Lack
/ ongoing hospital involvement
prevention in end-of-life care
Visitors, staff,
Guidelines
Source
“International”
National
– Yorkshire Cancer Network
LTHT / NHS Leeds – Leeds Health Pathways
Regional
What
Cancer-related
Site-specific
e.g. vascular catheter, urinary tract, pneumonia
Organism
specific
e.g. MRSA, C difficile, Candidiasis, Aspergillosis
(Setting:
1o or 2o care)
Guidelines:
NCCN: Prevention and treatment of cancer related
infections - V.1.2012
NCCN Clinical Practice Guidelines in Oncology
www.nccn.org
More traditional – “febrile neutropenia”
IDSA:
Clinical Practice Guideline for the use of
antimicrobial agents in neutropenic patients with
cancer: 2010 Update by [IDSA].
Freifeld A, Clin Inf Dis 2011:52:e56-e93.
NICE:
Neutropenic sepsis: prevention &
management…in cancer patients
guidance.nice.org.uk/cg 151 [issued Sep 2012]
1960s: Increased sepsis risk with falling neutrophil count – risk of
bacteraemia:
Gram negative bacilli, esp Pseudomonas aeruginosa
High mortality rate: > 50% < 48hrs
1970s: Empiric early iv therapy
Schimpf 1971: carbenicillin + gentamicin
1980s: Broader spectrum β - lactams
Option: Monotherapy vs “dual” therapy
1990s: Risk stratification w.r.t oral / out patient Mx
000s:
Emerging infections / new agents..
010s:
(..too early ?: more emergence…& fewer new agents)
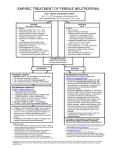
NICE (2012):
Treat neutropenic sepsis [NS] (2o / 3o care)
as acute medical emergency, offer empiric
antibiotic therapy immediately
Offer
β-lactam monotherapy with piperacillintazobactam initially to patients who need
intravenous treatment – unless there are patient
specific or local microbiological contraindications
Do
not offer an aminoglycoside, either as
monotherapy or dual therapy, for initial empiric
treatment of [NS] unless patient specific or local
microbiological indications.
Diagnosis of neutropenic sepsis in patients
having anticancer treatment with
neutrophil count < 0.5 x109 / L, &
Temperature > 380 C or
Other signs / symptoms consistent with
clinically significant sepsis.
…”Getting it right first time”
Kumar 2006: Septic shock patients,
duration of hypotension prior to initiation of
effective antimicrobial therapy – link to
survival.
Within first hour: Survival 79.9%
Each subsequent hour delay: average
drop in survival 7.6%
“Low risk” of complications
Oral therapy.
(Initial / sequential)
Oral vs intravenous
Meta-analysis: Vidal 2004
15 trials, mortality rate: 0 – 8.8%
RR: 0.91 (95% CI 0.51 – 1.62)
Treatment failure rates:
Initial; or sequential (iv then PO)
Overall: RR 0.94 (0.84-1.05)
Initial oral: 0.89 (0.77 – 1.03)
Sequential: 1.03 (0.86 – 1.24)
Adverse events
No death / permanent damage attributed to oral Rx.
Higher rate of GI side effects in oral regime.
MASCC risk index score (Klastersky 2000)
Characteristic
Extent of illness
No symptoms
Mild
Moderate
No hypotension
No COPD
Solid tumour or no IFI
No dehydration
Outpatient at onset of fever
Age < 60 [>16]
Score
5
5
3
5
4
4
3
3
2
>/ 21 = low risk complications / morbidity
PPV 91%, specificity 68%, sensitivity 71%
Commonest: Quinolone + co-amoxiclav
6 trials: quinolone alone
(No difference shown between above – ‘post –
protocol’ analysis).
Oral antibiotic therapy :
safely offered to neutropenic children / adults,
haemodynamically stable, have no organ failure, can
take PO medications,
Do not have pneumonia, central line infection, severe
SSTI
Not acute leukaemics
(Or use MASCC scoring system).
Prudent: FQ + 2nd drug active vs G+ve: eg co-amoxiclav.
Japanese guidelines: Quinolone alone
Unless mucositis / skin lesions: then eg with co-amoxiclav
Ciprofloxacin susceptibilities (bacteraemias, Haematology)
Organism
Ciprofloxacin resistant
(Total)
%
All Gram negative
bacilli
31 (132)
24
“Coliforms”
8 (70)
E coli 5
11
NLFs, incl P
aeruginosa
7 (45)
P aer: 2
16
S maltophilia
16 (17)
94
…potential fly in the ointment –
prophylactic strategy
Antibiotic prophylaxis
Afebrile neutropenic patients
Reduce frequency of febrile episodes by
administration of (broad spectrum) Ax:
But potential deleterious effects:
Toxicity
Emergence of antibiotic resistant bacteria
(FQ)
Fungal overgrowth
Bucaneve NEJM 2005 353:977-87
760 adult patients
500 mg Levofloxacin vs placebo for neutropenia
Febrile episode
All treated: 65 vs 85% (ADR -0.20; -0.26 to -0.14)
Acute leukaemia: 67 vs 85% (ADR -0.19; -0.27 to -0.10)
Solid tumours / lymphoma: 62 vs 84%, (-0.22, -0.29 to -0.12)
Death
All treated: 3 vs 5 % (ADR -0.02, -0.05 to 0.005)
Acute leukaemia: 5 vs 7% (ADR -0.02, -0.07 to 0.02)
Solid tumours / lymphoma: 1 vs 3%, (-0.02; -0.05 to 0.004)
Cullen 2005 NEJM 2005 353:988-98
1565 patients, cyclic chemotherapy for solid tumours /
lymphoma (13%)
500 mg Levofloxacin vs placebo for 7 days
Febrile episode first cycle: 3.5% vs 7.5% (p<0.001)
Over entire course: 10.8 % vs 15.2% (p=0.01)
Hospitalisation 15.7 vs 21.6% (p=0.004)
Severe infection 1 vs 2.0 % (NS)
Each group: four infection related deaths.
Gafter-Gvili 2006, Cochrane review
101 trials, 12599 patients: 1973-2005
Infection related deaths
RR 0.59 (0.47-0.75)
Fever occurrence
RR 0.77 (0.74 – 0.81)
All cause mortality (quinolone)
RR 0.52 (0.37 – 0.74)
Antibiotic resistance
Infecting organisms
Individual patient
Unit / Hospital
Community
“Collateral”
MRSA, C difficile
(Reduced use of other antibiotics)
(Cost)
Treatment strategy: oral regimens
MRSA
Risk with fluoroquinolones.
MRSA usually resistant to fluoroquinolones
Good skin tissue penetration / excreted in
human sweat:
Loss
of colonisation resistance by normal skin flora
In vitro:
of fibronectin – binding proteins
Increased adhesion by quinolone resistant S
aureus
Induction
Bisognano 2000, Paterson 2004,
Multiple logistic regression analysis, factors associated with
MRSA infection
Graffunder 2002
Risk factor
OR
95% CIs
P value
Levofloxacin
8.01
3.15, 20.3
<0.001
Macrolides
4.06
1.15, 14.4
0.03
Enteral feeding
2.55
1.37, 4.72
0.003
Surgery
2.24
1.19, 4.22
0.01
Previous
hospitalisation
1.95
1.02, 3.76
0.04
LOS before
culture
1.03
1.0, 1.07
0.05
NCCN 2012:
Fever & Neutropenic risk category: LOW
Standard chemotherapy regimens for most
solid tumours
Anticipated neutropenia less than 7 days
Prophylaxis:
Bacterial: NONE
Fungal:
None
Viral:
None unless prior HSV episode
Fever & Neutropenic risk category: Intermediate
/ High
Prophylaxis:
Bacterial: Consider FQ prophylaxis
CG151:
Adult patients with acute leukaemias, stem
cell transplants or solid tumours in whom
significant neutropenia (\< 0.5x109 /L) is an
anticipated consequence of chemotherapy
Offer prophylaxis with fluoroquinolone during
expected period of neutropenia only.
[No mention of any different action if known
FQ resistant, or FQ contra-indicated].
[No mention of specific oral options for FNE
whilst on FQ prophylaxis – initial or s/down]
…CG151 still being assessed LTHT / YCN
β-lactam resistance in Enterobacteriaceae:
Enzyme mediated: β-lactamases
(Ancient
Serine residue active site; or metalloenzymes (Zinc ion)
Inherent
heritage: > 2 billion years old)
– gene carried on bacterial chromosome
“De-repressed”: e.g. Enterobacter, Citrobacter species
Acquired
– transmissible genetic elements:
plasmids
E.g. Klebsiella pneumoniae, E coli
Vary in ability to hydrolyse different β-lactams:
Some
drug structures more resilient than others.
Some blocked by β-lactamase inhibitors
clavulanic acid (co-amoxiclav), tazobactam (with
piperacillin)
Various classifications / name derivations
3 letter monikers for families:
SHV (>50): Variable response to sulfhydryl inhibitors
TEM (>130): After patient (Temoneira)
CTX-M (>40), OXA, IMP:
Ability to hydrolyse cefotaxime, oxacillin, imipenem
VIM: Verona integron encoded metallo-β-lactamase
KPC: Klebsiella pneumoniae carbapenemase
New York / US.
NDM: New Delhi metallo-β-lactamase
Jacoby 2005;
NDM-1:
First detected United Kingdom January 2008.
Now predominant carbapenem-hydrolysing
enzyme in Enterobacteriaceae in UK (44%
2009)
2008 – 2009: 37 isolates
K
pneumoniae (21), E coli (7), Enterobacter spp
(5), Citrobacter freundii (2), Morganella (1),
Providencia (1)
29 patients – 15 in urine
ESBLs widespread in India,
NDM-1 also in isolates in north & south India
Links between many of the UK patients and
India
Kumaraswamy 2010; HPA
ESBLs:
carbapenem
Carbapenemase producing
Tigecycline
Polymyxin (colistin)
?
Possible local choices outside the box
Yorkshire & Humber: E coli surveillance data, 2010 -2012
792 isolates, 14 hospital trusts (courtesy of HPA)
8% = ESβL producers
0.1% = carbapenemase producer
“Other” options
(Co-trimoxazole)
Intramuscular gentamicin
Urinary
catheter change
“usually” sensitive in vitro:
Nitrofurantoin
Pivmecillinam
(lower UTI, eGFR > 60ml/m)
Mecillinam
Beta lactam (6-β-amidinopenicillanic acid)
Pivmecillinam Pivaloyloxymethyl ester:
Much more active vs Gram negatives
(Enterococci resistant, S saprophyticus may be inhibited)
Enterobacteriaceae
Usual suspects more tricky:
P aeruginosa, Acinetobacter spp, anaerobes: resistant
Serratia marcescens: usually resistant
M morganii, Providencia spp may be sensitive
(Paradoxical effect with P stuartii)
P mirabilis, P vulgaris: usually sensitive
Uses: Urinary tract infections
Advantages:
Lower
(Upper – step down oral therapy).
((Other MDR coliforms: e.g. Biliary))
High % still susceptible (> 90% global)
Low C difficile propensity
“Avoid” if penicillin allergy
(tho’ hypersensitivity reactions uncommon)
…NB: Avoid treating “asymptomatic
bacteriuria” (catheterised or noncatheterised) in adults.
Increase resistance
Loss
of oral options.
“Good” bacteria
Enterococci
Respiratory tract bacterial pathogens
AWARE surveillance, US, 2008-10
Adapted from Pfaller 2012
% susceptible
S pneumoniae
H influenzae
M catarrhalis
Amoxicillin
83
73
Co-amoxiclav
83
99.9
100
Erythro/clarithro
60
76
99.5
Tetracycline
75
99
99.8
Co-trimoxazole
66
77
94.4
Levofloxacin
99
100
100
Linezolid
>99.9
N/A
N/A
..?.. Doxycycline may also protect against
development of C difficile infection
US study comparing ceftriaxone +/- doxycycline
> 2300 patients studied
1.67 / 10000 patient days vs 8.11 / 10000
Rate of CDI 27% lower for each day of receipt
HR
0.73, 95% CI 0.56 – 0-.96
Doernberg 2012
..NB these organisms (and S aureus et al)
can be normal upper respiratory tract
commensal flora.
Adjudge “positive” microbiology results in
conjunction with current clinical /
radiological findings.
S aureus, AWARE surveillance, US, 2008-10
Adapted from Farrell 2012
% susceptible
MSSA
MRSA
Penicillin (amoxicillin)
23
(0)
Flucloxacillin
(100)
(0)
Erythromycin
66
8
Clindamycin
94
66
Tetracycline
96
95
Co-trimoxazole
99
99
Levofloxacin
89
29
Linezolid
100
99.8
Long-term intravascular catheters
Long term catheters should be removed
for patients with CRBSI associated
with:
Severe sepsis
Suppurative thrombophlebitis
Endocarditis
BSI that continues despite > 72 hours
suitable antimicrobial therapy
Infections due to S. aureus, P aeruginosa,
fungi or mycobacteria
IDSA, Mermel 2009
S aureus ; removal, unless major contraindications; e.g.
No alternative venous access
Significant bleeding diathesis
Quality of life issues take priority over
need for re-insertion of a new catheter
at a different site
If retain: four weeks therapy, systemic +
lock therapy.
Uncomplicated CRBSI involving longterm catheters due to [other] pathogens
Attempt treatment without catheter
removal:
Systemic and antimicrobial lock therapy
Administer both for 7 – 14 days
..If multiple positive catheter-drawn
blood cultures with coagulase –
negative staphylococci or Gram –
negative bacilli, but concurrent negative
peripheral blood cultures: can give lock
therapy without systemic therapy for 10
– 14 days.
Vancomycin: at least 1000x higher than
organism MIC (e.g. 5 mg/ml)
Other locks
Gentamicin
Taurolidine (TauroLock®)
Broad
spectrum antimicrobial
Unique site of action / no cross resistance
Spontaneously breaks down.
Lock therapy:
14 days in total
?Alternating lumens 24 hourly in hospital &
access needed
Dwell time up to one week
Repeat luminal blood cultures post completion
48
– 72 hours
Clostridium difficile
Diagnosis
Now two stage testing process:
Initial: GDH
If positive: toxin test.
Treatment (..if indicated; also review PPIs)
Metronidazole: “non-severe”
Vancomycin: “severe”: colitis; WCC >15, AKI
(Fidaxomycin)
Infection Prevention:
Hand washing with soap & water
..Improve speeds of other diagnoses
Aetiology; in vitro susceptibilities
Bacteriological methods long-standing,
but:
Direct to specimens: PCR / NAAT
blood,
sterile sites
Organism identification: MALDI-TOF
Matrix-Assisted
Laser Desorption Ionisation Time
of Flight Mass Spectroscopy: bacteria / yeasts
…expect more “unheard of” species names
Rapid automated sensitivity testing (< 12
hours), [EUCAST].
Fungal:
Prophylaxis
Empiric
Fever
in neutropenic patient unresponsive to broad
spectrum antibiotics
“Pre-emptive”: suspicion of IFI
Targeted treatment – proven / probable
Candida
Candida albicans
Non C albicans: C krusei
C glabrata
Aspergillus spp
Aspergillus fumigatus
Zygomycetes
Mucor, Rhizopus,
Amphotericin B
Azoles
Lipid formulations – ambisome, (abelcet, amphocil)
Fluconazole
Itraconazole
Voriconazole
Posaconazole
Echinocandins
Caspofungin
Micafungin
Anidulafungin
Changing criteria for diagnosis
De Pauw 2008: EORTC / MSG revised definitions of Invasive
Fungal Disease [for trial use]
Host factors:
Recent neutropaenia (<0.5 for > 10 days)
Allogeneic stem cell transplant receipt
Prolonged use corticosteroid (mean minimum: equivalent 0.3mg/kg/d, >
3 weeks)
Other recognised T cell immunosuppressants:
Clinical criteria
Lower respiratory tract disease: 1 out of 3 defined HRCT signs.
Sinonasal infection
Sinusitis on imaging + > 1 sign of eg acute localised pain, nasal ulcer + black
eshar
Mycological criteria:
eg cyclosporin, alemtuzumab (CamPath)
Direct tests: microscopy / culture,
Indirect tests: Aspergillus antigen
“Proven”, “probable”, “possible”
Pre-emptive
(“Suggestive”) Evidence of IFI:
Galactomannan
Blood
BAL.
Β – glucan
High
- Aspergillus
Resolution chest CT
Dense, well-circumscribed lesion(s) +/- halo sign
Air-crescent sign
Cavity.
Halo sign
Air crescent sign
Aspergillus microscopy
Colonies of Aspergillus fumigatus
Treatment – ongoing – azoles
Aspergillosis: voriconazole
More exotic moulds: posaconazole
…trough levels.
Conclusions:
Antimicrobial stewardship
Start Smart – then Focus.
Increasing antimicrobial resistance – Gram
negatives.
Review Microbiology results in conjunction
with other current information.
Antimicrobial guidelines on intranet individual
cases - Microbiology advice.