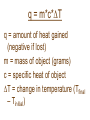* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Chapter 6
Survey
Document related concepts
Transcript
Chapter 6 Thermochemistry Energy • Is important macroscopically and microscopically • Def: the capacity to do work or produce heat Law of Conservation of Energy • Energy can neither be created nor destroyed, but can be converted from one form to another. • The energy of the universe is constant. • FIRST LAW OF THERMODYNAMICS Classifications of Energy • Potential Energy: energy due to position or composition – Ex: dam water, attractive/repulsive forces • Kinetic Energy: energy due to motion, depends on mass (m) and velocity (v) – KE = (1/2)mv2 • Energy can be converted between these Energy Transfer • Some energy can be lost as heat (ex: frictional heating), represented by q • Heat vs. Temperature: TEMPERATURE reflects movement of particles. HEAT deals with transfer of energy between two objects due to a temperature difference. • Energy can also be transferred through work (force activing over a distance) Important Vocabulary • Pathway determines how energy changes to heat/work – Includes condition of the surface • Total energy transferred will be constant, amounts of heat/work will differ State Function/Property • Property of the system that depends only on its present state (not past or future) • Changes in the state properties when switching from one state to another is independent of the particular pathway taken between the two states. • Example: energy is a state function, but work and heat are not. – Internal energy, pressure, volume, enthalpy Heat • Represented by “q” • Flows from warm to cold System vs. Surroundings • System is what you are focusing on, surroundings is everything else. • Energy lost/gained by the system = energy gained/lost by the surroundings Endothermic • System gains heat, surroundings cool • Heat INTO (“endo”) system • Example: Ice melting • “q” is positive • Heat is a reactant N2(g) + O2(g) + HEAT(kJ) --> 2NO(g) Exothermic • • • • • System loses heat to surrounding “Exo” like EXIT Example: fire “q” is negative Heat is a product CH4(g) + 2O2(g) --> CO2(g) + 2H2O(g) + HEAT(kJ) Potential Energy • Energy is stored in chemical bonds as potential energy. When bonds are broken (requires energy) and formed (releases energy), it changes the potential energy. Units for Energy Two common units: –The “calorie” (old school) –The “joule” (metric) 1 cal = 4.18 J J = kg•m2 s2 101.3 J = 1 L atm Energy Stoichiometry • Energy can be added into stoichiometry equations… C6H12O6 + 6O2 --> 6CO2 + 6H2O + 2800kJ • You can substitute it in as part of the mole to mole ratio! • How much heat is given off when 3.72 moles of oxygen react with glucose? • Answer: 1736 kJ Internal Energy • Represented by E • Sum of kinetic and potential energy in the system ∆E = q + w • ∆E = change in system’s internal energy • q = heat (usually in J) • w = work (usually units are L atm which can be converted to J) Example • Calculate the change in energy of the system if 38.9 J of work is done by the system with an associated heat loss of 16.2 J. • Answer: 55.1 J Expansion vs. Compression w = -P∆V • w = work • P = pressure • ∆V = change in volume Results… • If a gas is expanding, ∆V is positive • If a gas is compressed, ∆V is negative • When w is negative, work if flowing out of the system (into surroundings) • When w is positive, work is flowing into the system Example • A piston is compressed from a volume of 8.3 L to 2.8 L against a constant pressure of 1.9 atm. In the process, there is a heat gain by the system of 350 J. Calculate the change in energy of the system. • Answer: 1400 J Enthalpy • Represented by “H” H = E + PV • E = internal energy • P = pressure of the system • V = volume of the system This means at constant pressure… • ∆H = q • Negative ∆H is exothermic • Positive ∆H is endothermic ∆H = Hproducts - Hreactants Calorimetry • Energy can’t be created nor destroyed. • If one object loses heat, another object must gain that heat. • If a cool object is placed into a hot one, the hot object gives energy to the cool one until they arrive at the same final temp. Conservation of Energy • The amount of heat lost by the hotter object equals the amount of heat gained by the cooler object: qgained = -qlost (one is system, other surroundings) Specific Heat Capacity (J/g°C) • The amount of energy needed to raise the temp. of 1 gram of an object by 1°C. • A high specific heat means the object requires a lot of energy to change temp. (pg. 245) • If unit is J/mol°C or J/molK, then it’s called molar heat capacity Constant Pressure Calorimeter • Also have constant volume calorimeters q = m*c*DT q = amount of heat gained (negative if lost) m = mass of object (grams) c = specific heat of object DT = change in temperature (Tfinal – Tinitial) q = m*c*DT • This equation only works when the temperature is changing. • This is used in calorimetry! Example • A bar of iron at 21.0°C is heated to 83.5°C. If the iron’s mass is 551 grams, how much heat was added? • Answer: -1.5X104 J Calorimetry Example • A 36.9 g sample of metal is heated to 100.0ºC, and then added to a calorimeter containing 141.5 g of water at 23.1ºC. The temperature of the water rises to a maximum of 25.2ºC before cooling back down. What is the specific heat of the metal? • Answer: 0.45 J/gºC