* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download MICROPATTERNED CELL CO-CULTURES USING LAYER
Cre-Lox recombination wikipedia , lookup
Community fingerprinting wikipedia , lookup
Gel electrophoresis of nucleic acids wikipedia , lookup
Cell-penetrating peptide wikipedia , lookup
Transformation (genetics) wikipedia , lookup
Vectors in gene therapy wikipedia , lookup
Cell culture wikipedia , lookup
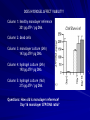
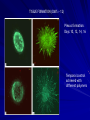
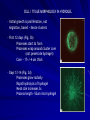
THREE-DIMENSIONAL GROWTH AND FUNCTION OF NEURAL TISSUE IN DEGRADABLE POLYETHYLENE GLYCOL HYDROGELS M.J. Mahoney, K.S. Anseth Department of Chemical and Biological Engineering and the Howard Hughes Medical Institute University of Colorado, Campus Box 424, Boulder, CO 80309, USA MARK HWANG POLYMER SCAFFOLDS - BACKGROUND Uses: - Model tissue ECM environment in vitro - Observation platform for: * cell-cell interaction * cell-ECM interaction - Testing platform for drug delivery * pre/post –encapsulation - Tissue replacement/grafting therapy Key Factors: - Mesh size - Scaffold chemistry POLYMER SCAFFOLDS - CENTRAL NERVOUS SYSTEM Grafting as disease treatment limited by: graft survival (viability) integration (retain functionality) Graft research focus for CNS disease Scaffolds support neural cell - growth - differentiation - function Cell line chosen for this study: undifferentiated (embryonic) murine neural precursor cells (NPC) STUDY GOALS Assess effect of 1) degradable hydrogel on 2) neural precursor cell (NPC) viability + 3) NPC differentiation + 4) 3D tissue morphology + given 5) mesh size that changes with time RATIONALE NPCs merits - in vitro expansion before transplantation unlimited NPC source - from previous studies successful transplantation into adult rats adequate chemical microenvironment in adult CNS ECM 3D v. 2D scaffold - directly implant 3D scaffold - cells must be dislodged from 2D substrate shear forces PEG hydrogel - non-immunogenic - tolerated in CNS - degradable - protein scaffolds (e.g. collagen) hard to control MATERIALS AND METHODS: BIG PICTURE Overall Goals 1) Construct gel determine degradation with mechanical tests Does hydrogel restrict cell growth? Does hydrogel affect viability? Incubate cells in gel 2) Cell imaging Stain for in gel viability Stain for in gel bioactivity (monitor calcium level) 3) Biochemical analysis after gel/cell lysed DNA levels ATP levels MATERIALS AND METHODS: CELLS and GEL Cell Culture -Embryonic forebrain removed + digested (rat) -Single cells cultured on: 1) poly ornithine coated cover slips + media (control) 2) 3-D hydrogel construct Hydrogel Preparation -Components PEG-Macromer (10wt%) cells + media photoinitiator (0.05wt%) -Exposed to UV light 10 min -Gel massed when dry/wet, calculate compressive modulus over time (degradation rate) MATERIALS AND METHODS: STAINING Confocal Imaging: Gels vibrotome sectioned a) Stained for live/dead VIABILITY Calcein-AM Calcein-AM = membrane permeable Cleaved calcein fluoresces, membrane impermeable Ethidium bromide Fluoresces red after binding DNA FUNCTION b) Stained for calcium Fluo-3 = calcium indicator GABA applied to cells Laser excited Fluo-3 measures calcium (GABA response) MATERIALS AND METHODS: DNA, ATP Obtaining cytosolic material Hydrogel homogenized with lysis buffer disrupts polymer gel Biochemical Analysis a) DNA content quantified with PicoGreen Assay b) Protein content analyzed with Western Blot - glial fibrillary acidic protein (GFAP) - beta tubulin III c) Immunocytochemical identification with antibodies (directly on gel) - GFAP - beta tubulin III - nestin - fibronectin - synaptophysin DOES HYDROGEL RESTRICT CELL GROWTH? Differences between monolayer (plate) and hydrogel cultures - 2-D v 3-D access to nutrients - physical obstruction in 3-D gel Is the hydrogel a physical barrier to growth? Are nutrients directed toward replication or creating space? 1 week Study: Culture both monolayer and 3-D hydrogel cultures Measure DNA content (reflects population size) Observations: No statistical difference in DNA content between both culture types (with p-value 0.05) Conclusion: Cells proliferate in early hydrogel equally well DOES HYDROGEL AFFECT VIABILITY? Study: Culture both monolayer and 3-D hydrogel cultures Measure DNA, ATP content (reflects population size) Observations: DEAD CELLS 1 pg ATP 8230 pg DNA 6 pg ATP / pg DNA Conclusion: MONOLAYER (24h) HYDROGEL (24h) 1486 ng ATP 7386 pg DNA 143 pg ATP / pg DNA 190 pg ATP / pg DNA Hydrogel viability significantly greater as measured by DNA content DOES HYDROGEL AFFECT VIABILITY? Column 1: healthy monolayer reference 201 pg ATP / pg DNA Column 2: dead cells Column 3: monolayer culture (24h) 143 pg ATP/ pg DNA Column 4: hydrogel culture (24h) 190 pg ATP/ pg DNA Column 5: hydrogel culture (16d) 215 pg ATP / pg DNA Questions: How old is monolayer reference? Day 16 monolayer ATP/DNA ratio? CELL AGGREGATION IN HYDROGEL (DAYS <12) Day 0: single cells distributed uniformly throughout gel Day 3: single cells and cell clusters (~ 20 um) Day 7: cell clusters (~ 30 um) Actively dividing CELL AGGREGATION IN HYDROGEL Mesh size increases 3x days 10-12 Gel completely hydrolyzed day 16 TISSUE FORMATION (DAYS > 12) Plexus formation: Days 10, 12, 14, 16 Temporal control achieved with different polymers CELL / TISSUE MORPHOLOGY IN HYDROGEL - Initial growth is proliferation, not migration, based ~ hence clusters - First 12 days (Fig. 3b) Processes start to form Processes wrap around cluster core (not penetrate hydrogel) Core ~ 17+/-4 um thick - Days 13-14 (Fig. 3d) Processes grow radially Rapid hydrolysis of hydrogel Mesh size increases 3x Process length ~52um into hydrogel CELL / TISSUE MORPHOLOGY IN HYDROGEL Fig. 1b - Mesh size inversely proportional to modulus - Processes penetrate hydrogel at 2 wks - PEG 2.5 glycolide decreases time to 1 wk - PEG 2 lactide increases time to 3 wks Possible to achieve temporal control of tissue growth in 3-D ECM IN HYDROGEL During development: Neurite receptors bind ECM ECM provides traction force for neurite extension ECM molecules - Laminin - Fibronectin - Collagen IV Normal Neurite ECM Y Y Y Hydrogel ECM N Y N CELL DIFFERENTIATION IN HYDROGEL - Neural precursors forms neurons or glia - Beta tubulin III neurons - GFAP glia Immunocytochemistry staining of hydrogel sections revealed: - Day 0 ~ 2.6*10^6 cells 66% cells beta tubulin III positive No GFAP - Day 16 ~ 7.3*10^6 cells (3.5x increase) 35% cells beta tubulin III positive (Fig. 4b) 38% cells GFAP positive (Fig. 4a) CELL DIFFERENTIATION IN HYDROGEL - Neural precursors forms neurons or glia - Beta tubulin III neurons - GFAP glia Immunocytochemistry staining Western Blot CELL FUNCTION IN HYDROGEL Method: observe cellular response to GABA to assay functionality 1) Fluo-3 tags calcium within cells 2) GABA transmitter applied to cells 3) Response cellular calcium influx visible with Fluo-3 Observations: Cells functional Fast and slow response types Did not mention proportion functional Typical calcium response SUMMARY - Achieved temporal control over gel degradation tissue formation - NPC generates limited (but sufficient) ECM - NPC proliferates AND differentiates - 3-D scaffold not physical obstacle - Proliferation/viability better than 2-D culture CELL / TISSUE MORPHOLOGY IN HYDROGEL Choice of graph? Does this mean at day 7: ~ 60% cells at 30um clusters and ~ 80% cells at 45um clusters? Or at day 16: ~ 20% cells at 20um clusters and ~ 80% cells at 70um clusters ?