* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Document
Survey
Document related concepts
Transcript
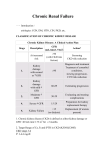
Diabetes, Anemia and Chronic Kidney Disease Josephine Carlos-Raboca,M.D., F.P.S.E.M., Endocrinology, Diabetes and Metabolism Makati Medical Center OUTLINE Defining Chronic Kidney Disease(Diabetic Renal Disease) Pathophysiology of Anemia in CKD Effects of Anemia of CKD Benefits of Early Treatment of Anemia Clinical Trials with Epoietin beta Conclusion What is Chronic Kidney Disease (CKD) ? Definition of Chronic Kidney Disease (CKD) CKD in early stages is characterised by kidney damage and level of kidney function CKD in later stages is defined as an estimated glomerular filtration rate (eGFR) for at least 3 months of – eGFR <60 mL/min/1.73m2 Stages of CKD are ranked by classifying severity of disease with declining eGFR and kidney damage CKD is a serious complication of diabetes mellitus NKF K/DOQI Clinical Practice Guidelines 2002: Am J Kidney Dis 2002; 39 (2 Suppl 1): S17-S31 Diagnosis of Kidney Function eGFR eGFR can be more accurately predicted from variables such as age, gender, race and body sizes with sCr – Commonly used prediction equations • Cockcroft-Gault uses sCr, age, weight and sex • MDRD (Modification of Diet in Renal Disease) in its simplest form uses sCr, age, sex and race eGFR is a better indicator of renal function than sCr alone eGFR easily determined from routine analyses Reviewed by Agarwal. Am J Kidney Dis 2005; 455:610-613 Glomerular Filtration Rate GFR in mg/dL GFR in umol/l Where Constant is 1.23 for men and 1.04 for women Stages of CKD by Glomerular Filtration Rate (GFR) Stage Description GFR (mL/min/1.73m2) 1 Kidney damage† with normal or GFR ≥90 2 Mild GFR 60−89 3 Moderate GFR 30−59 4 Severe GFR 15−29 5 Kidney failure <15 or dialysis †Kidney damage is defined by the National Kidney Foundation as ‘pathologic abnormalities or markers of damage, including abnormalities in blood or urine tests or imaging studies’ NKF-K/DOQI. Am J Kidney Dis. 2002;39(Suppl 1):S1-S266 Symptoms of CKD Diabetic kidney disease and anaemia Anaemia is prevalent in people with diabetic kidney disease and is largely unrecognised and untreated Anaemia occurs earlier in diabetic kidney disease than is commonly recognised Anaemia worsens with declining kidney function . Astor et al. Arch Intern Med. 2002;162:1401-1408 Anaemia develops early in CKD NHANES III – 15,419 non-institutionalised adults over the age of 20 – Prevalence of anaemia (KDOQI) increased from 1% at glomerular filtration rate of 60 ml/min to 9% at 30 ml/min and 33% at 15 ml/min 20 Hb (g/dL) Hb (men) Hb (women) 15 10 5 60 30 15 Glomerular filtration rate (ml/min) Astor et al, Arch Int Med 2002; 162: 1401-1408 Aetiology of anaemia Red Blood Cell (RBC) Production 2 000 000 cells/sec 120 000 000 cells/min 173 000 000 000 cells/day Normal values in adults RBC parameter Men Women Hb (g/dL) 15.7±1.7 13.8±1.5 Haematocrit (%) 46.0±4.0 40.0±4.0 RBC count (x1012/L) 5.2±0.7 4.6±0.5 Adapted from Williams et al. In: Williams’ Hematology. 5th ed. 1995;8-15 Defining Anemia Guideline Definition of Anemia European Best Practice Guidelines (EBPG) 2004 Anemia Guideline <12.0 g/d: in males and postmenopausal females; <11.0 g/dL in premenopausal females and prepubertal patients Kidney Disease Outcomes Quality Initiative (KDOQI) 2006 Anemia Guideline <13.5 g/dL males <12.0 g/dL females Potential causes of anaemia in chronic kidney disease Shortened red blood cell survival Iron deficiency Malnutrition and other deficiencies Chronic inflammation Decreased erythropoietin production Inhibition of erythropoiesis The Lifecycle of the RBC 120 days Globin Circulation Amino acids Erythropoiesis in bone marrow Fe3+ Transferrin Fe Heme Ferritin and haemosiderin Biliverdin Bilirubin Macrophage in spleen, liver or red bone marrow Bilirubin Liver EXCRETION Erythropoietin (EPO) Produced predominantly by peritubular fibroblasts in the kidneys and released in response to anaemia and hypoxia Release is modulated through the sympathetic nervous system (ß-adrenergic receptors) Anaemia associated with EPO deficiency usually occurs at a glomerular filtration rates below 35-40 ml/min but may occur at higher levels in diabetic kidney disease Regulation of Erythropoiesis Feedback loop Erythroid marrow Erythropoietin RBCs Kidney Circulating RBCs O2 Adapted from Erslev & Beutler. In: Williams’ Hematology. 5th ed. 1995;425-441 The Role of Erythropoietin in Erythropoiesis Stage 1: CD-34 Stem cell pool Progenitor cells BFU-E, CFU-E Stage 2: Erythron Precursor cells erythroblasts Mature cells GM-CSF IL-3, IGF-1 SCF Erythropoietin Erslev & Besarab. Kidney Int. 1997;51:622-630 Erythropoietin Receptor 508 amino acids, 66–78 kDa glycoprotein Located on erythroid progenitor cell surface Approximately 1000 erythropoietin receptors per cell Expression – primarily on CFU-E – small numbers on BFU-E – no receptors present once cells become reticulocytes EPO Membrane P JAK2 P STAT P JAK2 P STAT P P Target genes Hb and Erythropoietin: the Anaemic Patient with CKD O2 Hb transport capacity peripheral hypoxia kidney peritubular cells DAMAGED serum EPO precursor cells O2 transport capacity erythroblasts ANAEMIA Hb erythrocytes reticulocytes INSUFFICIENT Diabetes and Anaemia Nephropathy (35%) CKD Neuropathy (50%) ↓Serum EPO level Diabetes Anaemia Hyperglycaemia ↓Serum EPO response RBC abnormalities ↓ RBC survival Anaemia in CKD: Summary The hormone erythropoietin is the physiological regulator of RBC production and lifespan In individuals with CKD, damage to the kidney compromises erythropoietin production Anaemia correlates with the severity of CKD Effects of anaemia in diabetic renal disease? The risk of coronary heart disease in people with diabetes is 2-4x higher than the general population and the risk of cerebrovascular disease up to 5x higher The risks of cardiovascular disease develop early in the course of chronic kidney disease and are increased by diabetes Combination of anaemia and chronic kidney disease substantially increases stroke risks Anaemia predicts left ventricular mass, left ventricular dilation, heart failure and death Reciprocal Relationship : Renal Anemia, Diabetes & CVD Cardiovascular disease in early chronic kidney disease HDFP study1 – subjects with serum creatinine >150 µmol/L vs. < 150 µmol/L OR for death after 8 years 2.2 Framingham study2 – increased incidence of cardiovascular disease in those with renal insufficiency Canadian multicenter cohort3 – incidence of cardiovascular disease already 35.2% in those with glomerular filtration rate >50 ml/min and rose to 45.3% in those with glomerular filtration rate <25 ml/min 1. Shulman et al Hypertension 1989; 13(5):I80-93 2. Culleton et al Kidney Int 1999; 56: 2214-2219 3. Levin et al, Am J Kidney Dis 1999; 34: 125 Anaemia in CKD Anaemia in CKD induces – increased cardiovascular (CV) workload leading to left ventricular hypertrophy (LVH) – reduced exercise capacity – fatigue Anaemia in CKD is linked with – increased CV morbidity and mortality Framingham study, N = 6223 8% mild CRF (males serum creatinine 136-265, females 120-265 µmol/L) Percentage (%) No renal insufficiency Chronic renal insufficiency 25 20 15 10 5 0 ECG LVH CHD CHF CVD ECG LVH=echocardiogram left ventricular hypertrophy CHD=coronary heart disease CHF=congestive heart failure CVD=cardiovascular disease Culleton et al Kidney Int 1999; 56: 2214-2219 CKD and Anaemia Increase the Risk of CHF Stage 5 CKD patients on dialysis (n=433) At start of dialysis – 31% had CHF – 19% had angina – 14% had coronary artery disease On dialysis, for each 1 g/dL fall in Hb – 42% increased risk of LVH – 18% increased risk of CHF – 14% increased risk of death 1. Foley et al. Kidney Int. 1995;47:186-192 2. Foley et al. Am J Kidney Dis. 1996;28:53-61 The Cardio-Renal Anaemia Syndrome A vicious circle Hypoxia CKD Anaemia Serum EPO production Apoptosis Renal vasoconstriction Fluid retention Cardiac output Hypoxia Sympathetic activity TNF-α Uraemia CHF CHF=congestive heart failure Adapted from Silverberg et al. Kidney Int Suppl. 2003;(87):S40-S47 Anaemia, chronic kidney disease and risk of stroke – the ARIC study, n = 13,716 Community based cohort, 9 yr follow up – mean age 54.1 ± 5.7, mean Hb 13.9, 10.6% diabetic – 15 percent Cr Cl < 60 ml/min, mean blood pressure 120/71 – 85 percent Cr Cl ≥ 60 ml/min, mean blood pressure 121/74 – Use of anti-hypertensives 24.6% & 23.5% Lower Cr Cl associated with higher crude stroke rate – Cr Cl < 60 ml/min, stroke rate 3.7 – Cr Cl ≥ 60 ml/min, stroke rate 2.06 Abramson et al, Kidney Int 2003; 64: 610-615 ARIC study – influence of anaemia (WHO) 10.53 Stroke rate 12 10 Cr Cl ≥ 60 ml/min Cr Cl < 60 ml/min 8 6 4 3.7 2.06 2 1.52 2.12 2.85 0 Total sample Anaemic group Nonanaemic group Abramson et al, Kidney Int 2003; 64: 610-615 Hospitalisation Risk Increases with Hb <11 g/dL Dialysis patients RR of hospitalisation 2.0 n=7998 1.55 1.5 1.16 1.09 1.00 1.01 1.0 0.5 P<0.0001 P=0.001 P=0.05 <8 8−9.99 10−10.99 P=0.77 0.0 11−11.99 ≥12 Hb level (g/dL) Pisoni et al. Am J Kidney Dis. 2004;44:94-111 Summary : CKD and CVD The association between chronic kidney disease and all forms of cardiovascular disease begins early in the evolution of chronic kidney disease, anaemia significantly amplifies this association Cardiovascular events and mortality in chronic kidney disease are increased in patients with diabetic kidney disease and are closely related to anaemia Why do we treat renal anaemia? Subjective1 – – – – well-being life satisfaction happiness psychological affect Others – – – – cardiac status2 blood transfusions3 hospitalisation4 mortality5 Objective1 – – – – energy level functional ability activity level health status 1. Evans et al J Am Med Soc. 1990; 263:825-830 2. Winearls Nephrol Dial Transplant 1995; 10(suppl10):3-9 3. Fellner et al Kidney Int; 1993; 44:1309-1315 4 Churchill et al Clin Nephrol 1995; 43:184-188 5. US Renal Data System 1998 Epoetin improves quality of life in predialysis patients 83 predialysis patients entered into a parallel-group, open-label clinical trial and randomised to – epoetin – no treatment Epoetin treatment significantly improved anaemia and – – – – – energy physical function home management social activity cognitive function Revicki et al Am J Kidney Dis 1995; 25: 548-554 Correction of anaemia improves left ventricular hypertrophy in dialysis patients 22 dialysis patients studied by echocardiogram before and after correction of their anaemia with epoetin Hb increased at least 3.0 g/dL over baseline Correction of anaemia produced: – decrease in left ventricular mass (p = 0.0004) – decrease in left ventricular end-diastolic volume (p <0.0001) Adapted from Silverberg et al. Can J Cardiol 1990; 6: 1-4 Building the evidence: Mortality & hospitalisation Collins et al1 – Relative risk of death and/or hospitalisation lowest at Hct levels of 36-39% Fink et al2 – Pre-dialysis epoetin treatment leads to a relative risk of mortality of 0.8 (n=4866, 1107 epoetin) 1. Collins et al J Am Soc Nephrol November 2001 2. Fink et al Am J Kidney Dis 2001;37:348-355 Mortality and haematocrit level: First year follow up, all dialysis patients Diabetes mellitus No diabetes mellitus 400 350 300 Deaths per 1000 patient years 250 200 150 100 50 0 <30 30 - <33 33 - <36 Haematocrit 36+ USRDS prevalent patients 1994-1997 Duration of Hb <11 g/dL Increases Mortality Risk Dialysis patients Relative mortality risk 2 1 ** 1.82 n=41 919 1.00 ** ** 1.52 61−80 * 1.10 * 1.12 1.32 1−20 21−40 41−60 0 0 81−100 Time with Hb <11 g/dL over 2 years (%) *P<0.05; **P<0.001 Ofsthun et al. Nephrol Dial Transplant. 2005;20(Suppl 5):v261 (abstract MP204) Hb Levels Predict Survival Prior to Dialysis Initiation CKD patients not on dialysis Probability of survival 1.00 0.95 Hb ≥13.0 g/dL 0.90 12−12.9 g/dL 0.85 0.80 11−11.9 g/dL 0.75 <10 g/dL Log-rank test: P=0.0001 10−10.9 g/dL 0.70 0 3 6 9 12 15 18 21 24 27 31 33 37 Months from Hb result Levin et al. Nephrol Dial Transplant. 2006;21:370-377 Anaemia Treatment Greatly Reduces Blood Transfusions Dialysis patients Mean units per patient per 4 weeks 0.6 Commencement of anaemia therapy 0.5 0.4 0.3 0.2 0.1 * 0 Pre 4 *autologous blood donation ahead of elective hip surgery 12 20 28 36 44 52 Weeks Eschbach et al. Ann Intern Med. 1989:111:992-1000 Building the evidence: delaying progression of chronic renal failure • 63 patients (serum creatinine > 300 μmol/L, creatinine clearance < 15 mL/min/1.73 m2) • 20 with Hb < 10 g/dL = study group (epoetin+) 43 with Hb > 10 g/dL= control group • Significant reduction in rate of progression of chronic renal failure in study group, no change in control group Jungers et al Nephron Dial Transplant 2001; 16: 307-312 Trials in treatment of CKD anaemia CREATE trial (Cardiovascular risk Reduction by Early Anaemia Treatment with Epoetin beta) CHOIR trial (Correction of Haemoglobin and Outcomes In Renal Insufficiency) ACORD (Anaemia CORrection in Diabetes) Aims of the studies : to establish whether early intervention – – – – prevents development of left ventricular hypertrophy reduces cardiovascular mortality and morbidity delays progression of chronic renal failure reduces stroke and heart failure related hospitalisations The CHOIR and CREATE Studies: Overview CKD patients not on dialysis CHOIR (n=1432) CREATE (n=605) Patient Population Stage 3–4 patients with renal anaemia and not on renal ‡ replacement therapy (RRT) Stage 3–4 CKD patients with renal § anaemia not on RRT Duration 16 months 700 patients completed trial 48 months 476 patients completed trial Primary Endpoints Composite Composite (death, MI, HF, stroke) (sudden death, MI, acute HF, CVA, TIA, hosp for angina or arrhythmia, PVD complications) Group 1: 13.5 g/dL† Group 2: 11.3 g/dL† Hb Targets †Original targets before protocol amendment: • Group 1: 13.0–13.5 g/dL • Group 2: 10.5–11.0 g/dL ‡ 127 and 111 patients in groups 1 and 2, respectively, progressed to RRT during study Group 1: 13–15 g/dL Group 2: 10.5–11.5 g/dL § 127 and 111 patients in groups 1 and 2, respectively, progressed to RRT during study Singh et al. N Engl J Med. 2006;355:2085-2098 Drüeke et al. N Engl J Med. 2006;355:2071-2084 CHOIR: Increased Risk of Composite Event with Target Hb 13.5 g/dL Stage 3–4 CKD patients Time to the primary composite endpoint Probability of composite event 0.30 Hb target 13.5 g/dL Hb target 11.3 g/dL 0.25 0.20 0.15 0.10 Events: 125 vs 97 HR=1.34 (1.03–1.74) Log rank test P=0.03 0.05 0.00 0 3 6 9 12 15 18 21 24 27 30 33 36 39 Patients at risk Group 1 715 654 587 520 457 355 Group 2 717 660 594 539 499 397 Month 270 176 101 293 182 107 72 67 55 44 23 23 Singh et al. N Engl J Med. 2006;355:2085-2098 CREATE: No Significant Difference in Time to First CV Event CKD patients not on dialysis Time to the primary endpoint of a first cardiovascular event† Event-free Survival (%) 100 90 80 70 60 Hb target 13–15 g/dL Hb target 10.5–11.5 g/dL 50 40 Events: 58 vs 47 HR=0.78 (0.53–1.14) Log rank test P=0.20 30 20 10 0 0 Patients at risk Group 1 301 Group 2 302 6 12 18 24 30 36 42 48 158 177 97 121 56 61 2 2 Month 279 286 268 272 249 257 207 223 †Before censoring of data on patients at the time of initiation of dialysis Drüeke et al. N Engl J Med. 2006;355:2071-2084 The Anaemia CORrection in Diabetes (ACORD) study The ACORD study is investigating the effects of anaemia correction with subcutaneous epoetin beta on – cardiac structure – cardiac function In patients with early diabetic nephropathy Primary endpoint – effect of early anaemia treatment on left ventricular hypertrophy as a cardiovascular risk marker ACORD: Study design Hb (g/dl) 16 14 16 m Randomisation Early intervention f 12 Target Hb: 13–15 g/dl 14 Standard treatment 12 Target Hb: 10.5–11.5 g/dl 10 8 Inclusion: Hb 10.5–13.0 g/dl Creatinine clearance ≥30 ml/min n = 160 6 10 8 6 Time Maintaining the Right Hb Range Summary Efficacy of SC epoetin beta confirmed by Hb response according to different Hb targets both in CREATE and ACORD Complete Hb correction improves QoL and was not associated with any consistent hazard Trend towards decrease in LVMI with complete anaemia correction among those with higher baseline LVMI – In CREATE and ACORD annual CV event rate lower than expected from previous studies Prospective international randomised trials (CREATE, ACORD) show no added benefit of complete Hb correction to 13-15 g/dL on CV outcome measures and CKD progression – But it may still be beneficial as CV event rates were low European recommendations for optimising treatment of renal anaemia Indication for start of epoetin therapy: – repeated Hb measurements <11g/dL – after exclusion of non-renal causes of anaemia (bleeding, nutritional deficiencies, hypothyroidism, iron deficiency, haemolysis) Target haemoglobin: – general: Hb >11 g/dL (no upper limit) – in CHD: Hb 11-12 g/dL Administration of epoetin: – SC dosing preferred; IV dosing also an option in HD patients – the goal is to increase Hb levels by 1–2 g/dL per month van Ypersele de Strihou Nephrol Dial Transplant 1999; 14 (suppl 2): 37-4 Iron stores Target – serum ferritin > 100 g/l (aim for 200-500) – hypochromic red blood cell count < 10%, TSAT > 20% (aim for < 2.5% & 30-40%) Level B Treatment strategies – predialysis and CAPD oral intravenous – HD will need intravenous Level B How should epoetin be administered to predialysis patients with diabetes and anaemia? EBPG & KDOQI recommend epoetin treatment for anaemia due to CKD when Hb < 11 g/dL Hb correction should be gradual to avoid: – exacerbation of hypertension – increased viscosity with adverse haemodynamics Guidelines recommend an increase of Hb 0.5 g/dL every 2 weeks Common practice suggest a target Hb of 11–12 g/dL (or ?12-13 g/dL) RECORMON DOSING GUIDE for CKD Patients with Renal Anemia: Correction Phase Subcutaneous Administration Intravenous Administration 60 IU/kg Body weight per week 120 IU/kg Body Weight per Week Dosage maybe increase every 4 weeks by 60IU/kg BW/wk if the increase in PCV is <0.5 volume % per week or <0.5 g/dL Hb every 2 weeks. Dosage maybe increase every 4 weeks by 120IU/kg BW/wk if the increase in PCV is <0.5 volume % per week or <0.5g/dL Hb every 2 weeks. Maintenance Phase: Dose can be reduced to 50% or adjusted at intervals of one or two weeks individually for the patients to maintain target Hb and Hct. The clear differences Epeotin Beta Recormon Epoetin alpha Half life SC 12-28 hours IV 4-12 hours SC 12-18 hours IV 4-6 hours Stabilizers Glycine Poylsorbate 20 Urea Calcium Chloride Complex of 5 amino acids Glycine Polysorbate 80 Storage and handling 2-8 degrees centigrade can be removed from refrigeration and store at 25 degrees centigrade for a single period of up to 3 days Can tolerate shaking 2-8 degrees at all times Can not tolerate shaking Tolerability Almost pain free at the injection site With pain at the injection site Dose Recommendation (Correction) 60 IU/kg Body weight/wk 100 IU/kg Body Weight /wk Shelf Life (PFS) 24 months 18 months Conclusions Anaemia is prevalent in diabetic kidney disease and occurs earlier than is commonly recognised Proven benefits of treatment of anaemia with epoetin (± intravenous iron) include – improved quality of life and performance status – regression of left ventricular hypertrophy – reduced transfusion requirements Potential benefits of early anaemia treatment include – reduced cardiovascular mortality and morbidity – delayed progression of chronic renal failure – reduced stroke and heart failure related hospitalisations Thank you