* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Nitrogen cycle
Survey
Document related concepts
Transcript
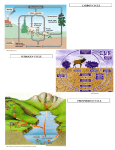
Environmental Chemistry: Nitrogen (N) Or….why we need healthy soils! N – So what??!! • What do plants need to live and grow? • “Nutrients” – what are they? • 17 Essential Elements – C, H, O – N-P-K – Ca, Mg, S – Micronutrients: B, Cu, Cl, Fe, Mn, Mo, Ni, Zn The Nitrogen Cycle • N2 makes up 78% of troposphere (inner-most layer of atmosphere)! • Nitrogen fixation: turning nitrogen in the atmosphere (N2) into a usable form for plants • N2 + 3H2 2NH3 (ammonia) • Bacteria do this (in soil, or living in nodules in special plants called legumes) • Industrial fertilizer factories do this • NH3 quickly dissolves into NH4 + (ammonium ions) GASEOUS NITROGEN (N2) IN ATMOSPHERE NITROGEN FIXATION by industry for agriculture FOOD WEBS ON LAND FERTILIZERS NITROGEN FIXATION bacteria convert to ammonia (NH3) ; this dissolves to form ammonium (NH4+) uptake by autotrophs excretion, death, decomposition uptake by autotrophs NITROGENOUS WASTES, REMAINS IN SOIL NO3IN SOIL AMMONIFICATION NH3, NH4+ IN SOIL loss by leaching by bacteria 2. NITRIFICATION bacteria, fungi convert the residues to NH3 , this dissolves to form NH4+ bacteria convert NO2- to nitrate (NO3-) 1. NITRIFICATION NO2IN SOIL bacteria convert NH4+ to nitrite (NO2-) DENTRIFICATION loss by leaching Fig. 4.30, p. 94 The Nitrogen Cycle • Nitrification: a two-step process • 1: Bacteria convert nitrogen in the soils (NH3 and NH4 + ) to two different forms – Nitrite ions (NO2-) BAD!! TOXIC! UNUSABLE! – Nitrate ions (NO3-) GOOD NUTRIENTS FOR PLANTS! • 2: Bacteria convert nitrite ions (bad, NO2) to nitrate ions (good, NO3-) that can be used by plants! GASEOUS NITROGEN (N2) IN ATMOSPHERE NITROGEN FIXATION by industry for agriculture FOOD WEBS ON LAND FERTILIZERS NITROGEN FIXATION bacteria convert to ammonia (NH3) ; this dissolves to form ammonium (NH4+) uptake by autotrophs excretion, death, decomposition uptake by autotrophs NITROGENOUS WASTES, REMAINS IN SOIL NO3IN SOIL AMMONIFICATION NH3, NH4+ IN SOIL loss by leaching by bacteria 2. NITRIFICATION bacteria, fungi convert the residues to NH3 , this dissolves to form NH4+ bacteria convert NO2- to nitrate (NO3-) 1. NITRIFICATION NO2IN SOIL bacteria convert NH4+ to nitrite (NO2-) DENTRIFICATION loss by leaching Fig. 4.30, p. 94 The Nitrogen Cycle • Denitrification: special bacteria in waterlogged soil or at the bottom of lakes, oceans, or wetlands change nitrogen back into nitrite and nitrate ions, then into nitrogen gas! • This is released into the air & atmosphere, to begin the cycle again. GASEOUS NITROGEN (N2) IN ATMOSPHERE NITROGEN FIXATION by industry for agriculture FOOD WEBS ON LAND FERTILIZERS NITROGEN FIXATION bacteria convert to ammonia (NH3) ; this dissolves to form ammonium (NH4+) uptake by autotrophs excretion, death, decomposition uptake by autotrophs NITROGENOUS WASTES, REMAINS IN SOIL NO3IN SOIL AMMONIFICATION NH3, NH4+ IN SOIL loss by leaching by bacteria 2. NITRIFICATION bacteria, fungi convert the residues to NH3 , this dissolves to form NH4+ bacteria convert NO2- to nitrate (NO3-) 1. NITRIFICATION NO2IN SOIL bacteria convert NH4+ to nitrite (NO2-) DENTRIFICATION loss by leaching Fig. 4.30, p. 94 The Nitrogen Cycle • More important processes! • Decomposition: process of breaking down organic material into its parts (nutrients) • Ammonification: process where bacteria and fungi convert nitrogen wastes into NH3 which dissolves into NH4• Assimilation: process where plant roots take up the useful forms of nitrogen to make DNA, amino acids and proteins in the plant tissue! The Nitrogen Cycle • When we “crop” the land, and take away plant material that was grown for crops, we remove N from this cycle! • If we deplete our soils of their nutrients too quickly, the sustainability of soil for our food sources and the health of our ecosystems are put in jeopardy! GASEOUS NITROGEN (N2) IN ATMOSPHERE NITROGEN FIXATION by industry for agriculture FOOD WEBS ON LAND FERTILIZERS NITROGEN FIXATION bacteria convert to ammonia (NH3) ; this dissolves to form ammonium (NH4+) uptake by autotrophs excretion, death, decomposition uptake by autotrophs NITROGENOUS WASTES, REMAINS IN SOIL NO3IN SOIL AMMONIFICATION NH3, NH4+ IN SOIL loss by leaching by bacteria 2. NITRIFICATION bacteria, fungi convert the residues to NH3 , this dissolves to form NH4+ bacteria convert NO2- to nitrate (NO3-) 1. NITRIFICATION NO2IN SOIL bacteria convert NH4+ to nitrite (NO2-) DENTRIFICATION loss by leaching Fig. 4.30, p. 94