* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download 11_lecture_microbes - New Learning Technologies website
Bacterial cell structure wikipedia , lookup
Phospholipid-derived fatty acids wikipedia , lookup
Triclocarban wikipedia , lookup
Bacterial morphological plasticity wikipedia , lookup
Human microbiota wikipedia , lookup
Marine microorganism wikipedia , lookup
Magnetotactic bacteria wikipedia , lookup
Neisseria meningitidis wikipedia , lookup
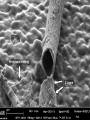
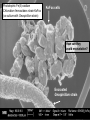
Metabolism Lectures Outline: Part I: Fermentations (Monday) Part II: Respiration (Wednesday) Part III: Metabolic Diversity (Friday) Learning objectives are: Learn about anaerobic respiratory metabolisms. How can an inorganic compound be use as an energy source. Bacteria and Archaea Agrobacterium species Alphaproteobacteria Gram negative rods Common in soil especially the root zone of plants Some are plant pathogens – – A. tumefaciens causes crown galls or plant tumors Only if A. tumefaciens has the Ti (tumor inducing) plasmid. Elements of Ti have been engineered to generate transgeneic plants using. Neisseria species Betaproteobacteria Gram negative, diplococcal Aerobic Most nonmotile N. gonorrhoeae – N. meningitidis – Spinal meningitis Other Neisseria spp. are present in respiratory tract of animals. – VD Most rarely cause disease. Cultivate on chocolate-blood agar with 3-10% CO2 www.textbookofbacteriology.net Pseudomonas species Gram negative, (Gammaproteobact.) Mostly obligate aerobes – Present in soil, water, plant surfaces Some can degrade pollutants – siderophores or iron binding molecules Some produce pigments: – TNT for example Produce secondary metabolites – Some can respire nitrate. Pyocyanin in P. aeruginosa Some fluoresce: – P. fluorescence From: www.bact.wisc.edu/Microtextbook Sulfate reducing bacteria Deltaproteobacteria Desulfovibrio speices Strict anaerobes Generate energy by respiration of sulfur compounds Some can use H2 for energy Many use lactate, acetate, and/or ethanol as carbon and energy sources. Abundant in anaerobic aquatic environments where sulfate is high – www.genomenewsnetwork.org Seawater Also abundant in anaerobic environments with lots of decomposing organic matter picasaweb.google.com/sd.gibson Campylobacter jejuni Gram -, (Epsilonproteobact.) Microaerophile Most prevalent food-borne pathogen in US – Under cooked poultry, pork, shellfish Prevalence of contamination: – 90% turkeys – 32% hogs – 89% chickens www.cdc.gov/ncidod/eid/vol5no1 Streptococcus Gram positive, Firmicutes Pathogenic and non-pathogenic kinds Non-pathogenic: – Oral Streptococcus – – S. lactis common dairy organism S. salivarius and mutans Grow on sugars in the mouth Pathogenic: – – – S. pyogenes: strains with hemolysins can cause scarlet fever S. pneumoniae: strains with capsules can cause disease. Some are “flesh eating” Bacteroides Rod shap, Gram Negative Strict anaerobe Dominant microbe in human feces 1010 per gram Purely fermentative organisms Normally commensal Most anaerobic infections are Bacteroides species. Big problem in GI tract surgeries from: microbewiki.kenyon.edu Pyrococcus “fireballs” furiosus Anaerobic, Crenarchaea Stetter isolated these from a solfatara field in Vulcano Italy (1986). Uses proteins, starch, sugars, maltose as electron donors for S0 reductions Also ferments sugars to H2 and CO2 Growth temps: – 70-106˚C – 100˚C is optimum www.microbeworld.org Thiobacillus ferrooxidans Gamma proteobacteria 4 Fe(II) + 4 H+ + O2 −−> 4 Fe(III) + 2 H2O Fe(II) is stable at acidic pH – Does not get oxidized in the presence of O2 T. ferrooxidans tolerates: – pH ~2.5 – It’s an acidiphile Can be found in acidic mine waters. Add water to pyrite: – FeS2 −> Fe(III) + H2SO4 – That’s sulfuric acid Iron mats made by iron oxidizing bacteria Anoxic photosynthetic iron(II) oxidizing bacteria O2 1 μm NO3−-dependent Fe(II)-oxidizer (Acidovorax sp. strain BoFeN1) Photos by Professor Andreas Kappler Fe2+ Phototrophic Fe(II)-oxidizer (R. ferrooxidans strain SW2) Fe-mineral coating 2 layers Phototrophic Fe(II)-oxidizer Chlorobium ferrooxidans strain KoFox (co-culture with Geospirillum strain) KoFox cells How can they avoid encrustation? Encrusted Geospirillum strain