* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Slide 1
Survey
Document related concepts
Transcript
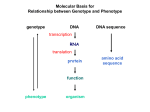
X-Ray Crystallographic Analyses of the Antimicrobial Resistant Enzymes: βlactamases and Aminoglycoside Phosphotransferases Matthew Au University of California: Merced Stanford Linear Accelerator Center August 16, 2012 Supervised by Clyde A. Smith Background (Infections) According to Centers for Disease Control (CDC): Nearly 2 million patients in the U.S. get an infection in the hospital each year About 90,000 of those patients die each year as a result of their infection, up from 13,300 patient deaths in 1992 This increase signifies that more and more bacteria are becoming resistant to an array of treatments Introduction In a U.S. News article about 5 months ago, doctors have identified bacteria that produce Klebsiella pneumoniae carbapenamse (KPC) Is an enzyme that makes bacteria resistant to most known treatments , even to the “last line of defense”: the carbapenem antibiotics Killed 50 people in Panama They have found traces in at least 37 U.S. states, Washington, D.C., and Puerto Rico The mortality rate is at around 50% They’re most commonly found on medical equipment Main Objective Have a basis of why these two bacterial enzymes have evolved to become resistant to these antibiotics Enzyme 1: Guiana Extended-Spectrum 1 (GES-1) Antibiotics attached: Doripenem, Ertapenem, Meropenem Enzyme 2: Aminoglycoside 3’-phosphotransferase (APH(2”)-IIa) and its mutant, R92H/D268N Antibiotics attached: Gentamicin and Isepamicin How? X-Ray Diffraction & Crystallography and Structural Construction and Refinement X- Ray Diffraction Molecules arranged in a regular, repeating pattern in crystals Electrons in the molecules bend an incident X-ray beam into thousands of diffracted beams Multiple copies of the same molecule in the same orientation amplify the diffraction peaks The diffraction pattern contains all of the information necessary to determine the structure X- Ray Diffraction Image Structural Construction The data is then run through a program called HKL2000, which measures the intensity of each diffraction spot These intensities are then converted into electron density (a probability function showing where the electrons are) The electron density gives us a guide as to where all the protein atoms are Electron Density 3 Carbapenem Antibiotics Carbapenem backbone Meropenem Ertapenem Doripenem GES-1 + Carbapenem GES-1 + Carbapenems Why GES-1? GES-1 is described as a weak carbapenemase It binds the carbapenem and deactivates the antibiotic-empowered beta-lactam ring but it doesn’t detach the drug This means the enzyme isn’t efficient for the bacteria because eventually all the enzyme gets tied up in an inactive form However GES-5, an evolved form of GES-1, deactivates and detaches the drug Leaves the active site open for infinite incoming antibiotic carbapenems This poses a big threat for the human population Now that we know how GES-1 binds these drugs we can start to understand how the GES enzymes are evolving into active carbapenemases APH(2”)-IIa Enzyme The aminoglycoside 3’-phosphotransferase [APH(2”)-IIa] enzyme is effective against an array of antibiotics: arbekacin, kanamycin, neomycin, streptomycin, gentamicin, and paromomycin It’s drug deactivation mechanism involves the phosphorylation of the antibiotics It derives an extra phosphate group from ATP APH(2”)-IIa + Gentamicin APH(2”)-IIa Mutant A new mutant of the APH(2”)-IIa, R92H/D268N, was recently discovered to have dominance over an additional antibiotic: isepamicin Isepamicin was unable to bind to the wild-type APH(2”)-IIa Isepamicin has a long tail which clashes with part of the enzyme structure Asparagine 196 (Asn196) APH(2”)-IIa + Isepamicin APH(2”)-IIa Mutant + Isepamicin APH(2”)-IIa Mutant Because of the mutation at arginine 92 (it is changed to histidine), a piece of structure containing Asn196 is made more flexible Asn196 is able to bend away from the tail of the isepamicin so that that drug can bind A phosphate group can now be attached and the drug is deactivated Acknowledgements Clyde Smith Stanford Linear Accelerator Center SULI Program Coordinators at SLAC Office of Science, Department of Energy