* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download How KTP is making an impact at GAMA Healthcare
Survey
Document related concepts
Transcript
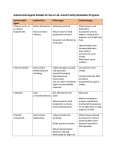
How KTP is making an impact at GAMA Healthcare Harsha Siani About Gama Healthcare GAMA Healthcare Ltd. develops and manufactures breakthrough antimicrobial products under the Clinell brand. Founded in 2004 the company focus is to develop infection control products with an emphasis on wet wipes. Encompassing a wide range of wipe products from skin decontamination and surface cleaning to catheter care and medical device disinfection. CLINELL PRODUCTS ARE SOLD TO OVER 450 NHS TRUSTS ACROSS THE UK, AND OVER 40 COUNTRIES WORLDWIDE Today the Clinell range is the number one brand of antimicrobial wipes used in the NHS 1 Cardiff University Expertise – Efficacy testing of biocidal products against micro-organisms (bacteria, spores, fungi) – Development of efficacy test protocols (3-stage wipe test) – Mechanisms of microbial resistance and cross-resistance to antimicrobials – Mechanisms of action of biocides 2 Cardiff – the home of ‘C. diff’ ! 3 Healthcare Associated Infections 4 Healthcare Associated Infections – 1 in 16 patients are diagnosed with an active HCAI. – HCAIs cost the NHS £1 billion annually (£7000 per patient). – 20-30% of HCAIs could be avoided. – Enhanced cleaning practices can reduce additional costs of HCAIs by 76% Types of HCAIs, HPA, 2011 5 What is C. difficile? – Gram-positive, spore forming anaerobic bacteria. – Patients excrete up to 107 C. difficile per g liquid faeces. – Primary virulence factors Toxin A and Toxin B. – Spores persist on surfaces for >5 months. – Infectious dose <7 spores/cm2. – Mode of transmission – Faecal oral route. Johnson and Gerding, 2012 6 Wipes and Healthcare Associated Infections – Pre-prepared wipes are increasingly being used for cleaning and disinfection. STAGE 1 Removal of bioburden from a surface – Wipes provide a convenient and quick means of cleaning and disinfection. STAGE 2 Bacterial transfer - Do the wipes transfer survivors to other surfaces – NHS market value for wet wipes is £15 million and growing by 10-20% per annum. STAGE 3 Antimicrobial activity – Current standard test methods are not representative of in-use application. – There is a poor level of evidence to support the efficacy of disinfectant wipes. 7 C. difficile Label Claims of Wipes WIPES INGREDIENT DISCLOSED ON LABEL CLAIM ON LABEL Wipe A Inorganic peroxygen generator, tetra acetyl ethylenediamine, surfactants Sporicidal Wipe B Impregnated with low-level biocides,’’ 5% cationic surfactant, amphoteric surfactant, EDTA Sporicidal Wipe C 1% cationic and nonionic surfactants, acidity modifiers Bactericidal claim and claim against C. difficile Wipe D 1% polymeric biguanide hypochloride, alkyl di-methyl benzyl ammonium chloride, didecyl dimethyl ammonium chloride Sporicidal Wipe E QAC, polymeric biguanide and bronopol Sporicidal Wipe F Didecyl dimethyl ammonium chloride 0.45% Sporicidal Wipe G Composition not disclosed Sporicidal Wipe H Didecyl ammonium chloride, laurakonium chloride, polyaminoporopyl biguanide, 2-bromo-2-nitro-para1-3-diol Sporicidal Wipe I Composition not disclosed Sporicidal Siani et al. AJIC 2011; 39(3):212-8. Siani et al. AJIC 2011; 39(3):212-8 Efficacy of ‘sporicidal’ wipes against C. difficile Wipes Bacterial Removal (log CFU/disc ± SD) Bacterial transfer following 10 s wiping 500 g surface pressure time at 500 g surface pressure Negative control 1.13 (± 0.36) 5 consecutive transfers. TNTC Hypochlorite soaked wipe 2.02 (± 0.21) 5 consecutive transfers. TNTC Wipe A 4.09 (± 0.79) No spore transferred Wipe B 0.22 (± 0.07) 5 consecutive transfers. From 0 to TNTC Wipe C 1.30 (± 0.33) 5 consecutive transfers. From 0 to TNTC Wipe D 0.57 (± 0.07) 5 consecutive transfers. From 1 to TNTC Wipe E +0.08 (± 0.08) 5 consecutive transfers. TNTC Wipe F 1.14 (± 0.65) 5 consecutive transfers. From 83 to TNTC Wipe G 0.67 (± 0.11) 5 consecutive transfers of ≤43 bacteria Wipe H 0.88 (± 0.13) 5 consecutive transfers. From 2 to TNTC Wipe I 0.84 (± 0.66) 5 consecutive transfers. From 40 to TNTC Siani et al. AJIC 2011; 39(3):212-8 9 KTP Aims – Transfer knowledge and understanding of biocide efficacy tests. – Provide scientific support to sales and marketing teams. – Develop in-use simulated tests for new and existing products. – Develop in-house R&D facility. – Assess efficacy of Clinell Sporicidal wipes in a phase 3 clinical field trial. 10 HOW HAS KTP BENEFITED GAMA HEALTHCARE? HOW HAS KTP BENEFITTED GAMA HEALTHCARE? 11 KTP and Gama Healthcare Provides scientific credibility to Gama’s products – Siani et al. AJIC 2011; 39(3):212-8 – Siani and Maillard, review to be submitted Export Team Conference presentations – Infection Control Africa Network, South Africa, 2012 (speaker) – Heamodialysis Technician Conference, Italy, 2012 (speaker) – Infection Prevention Society, London, 2013 (poster) Sales Team – Invited speaker for Local Infection Control study days – Scientific support for the dissemination of product data Marketing Team – Production of marketing material based on scientific literature ‘Protecting the Patient’ , ‘Benefits of Wipes’, ‘QACs are not Sporicidal.’ 12 Assessing the efficacy of Clinell® Sporicidal Wipes – A double cross over field trial – 9 months study at the University Hospital Wales across 2 wards. – Total aerobic and anaerobic counts. – ATP and UV monitoring. – Isolation of MRSA, VRE, CRE, ESBL and C. difficile. – Generating >12000 data points. Phase 1 Phase 2 Phase 3 Month 1 2 3 4 5 6 7 8 9 Ward 1 Cr Cr Cr Sp Sp Sp Cr Cr Cr Ward 2 Cr Cr Cr Cr Cr Cr Sp Sp Sp –Provides the first phase 3 clinical trial on the efficacy of wipes in in-use conditions. 13 KTP and Gama Healthcare Research and Development – Development of new products (Blood spill kit, Floor cleaner and 15 new formulas) – Efficacy testing of existing products – Review of competitor products and claims – Interaction of wipe materials and bacteria Cost Saving £25,000.00 Cost £20,000.00 £15,000.00 £10,000.00 £5,000.00 £0.00 Comercial Lab A Comercial Lab B KTP 14 KTP and Gama Healthcare Research and Development – Development of new products (Blood spill kit, Floor cleaner and 15 new formulas) – Efficacy testing of existing products – Review of competitor products and claims Net Profit (£) – Interaction of wipe materials and bacteria 400000 350000 300000 250000 200000 150000 100000 50000 0 During KTP Yr 1 Current customers Yr 2 New customers Yr 3 New products 15 What does it all mean? 16 ANY QUESTIONS? 23/05/2017 Clostridium Difficile 17